Albumin Nanoparticles in Oncology: A Comprehensive Guide to Next-Generation Cancer Drug Delivery
This article provides a comprehensive, state-of-the-art analysis of albumin-based nanoparticles for cancer therapy, tailored for researchers and drug development professionals.
Albumin Nanoparticles in Oncology: A Comprehensive Guide to Next-Generation Cancer Drug Delivery
Abstract
This article provides a comprehensive, state-of-the-art analysis of albumin-based nanoparticles for cancer therapy, tailored for researchers and drug development professionals. It systematically explores the foundational science of albumin as a versatile drug carrier, details advanced synthesis and targeting methodologies, addresses critical challenges in formulation and scale-up, and validates performance through comparative analyses with other nanoplatforms. The scope encompasses the full pipeline from rational design and preclinical optimization to clinical translation and future outlook.
Why Albumin? The Scientific Rationale for a Natural Drug Carrier
Application Notes
Biocompatibility and Safety Profile
Human serum albumin (HSA) is an endogenous protein, conferring high biocompatibility and low immunogenicity to nanoparticles (NPs). This underpins its clinical success as a carrier, as evidenced by the FDA-approved nab-paclitaxel (Abraxane). The safety profile is quantified by key parameters.
Table 1: Quantitative Biocompatibility Metrics of Albumin-Based Nanoparticles
| Parameter | Typical Value Range | Measurement Method | Significance |
|---|---|---|---|
| Hemolysis (%) | <5% (at 1 mg/mL) | Spectrophotometry (540 nm) | Indicates erythrocyte membrane integrity. |
| Plasma Protein Corona Thickness (nm) | ~5-15 nm | Dynamic Light Scattering (DLS) | Lower thickness than synthetic polymers reduces macrophage recognition. |
| Complement Activation (C3a elevation) | Minimal | ELISA | Predicts reduced infusion-related reactions. |
| In Vivo Half-life (Human, HSA) | ~19 days | Radiolabeling (¹³¹I) | Long endogenous circulation reduces clearance. |
| In Vivo Half-life (Murine, Albumin NPs) | 6-24 hours | NIR Fluorescence / HPLC | Depends on particle size and surface charge. |
| LD₅₀ (Rodent, HSA) | >20 g/kg | Acute toxicity study | Demonstrates exceptional intrinsic safety. |
Long Circulation and the EPR Effect
Albumin NPs exploit the Enhanced Permeability and Retention (EPR) effect. Their small size (<150 nm) and negative surface charge (approx. -10 to -25 mV) minimize renal clearance and reduce nonspecific uptake by the reticuloendothelial system (RES). The long circulation is mediated by albumin's interaction with the neonatal Fc receptor (FcRn), which recycles albumin from endosomes back to the bloodstream, prolonging half-life.
Table 2: Key Physicochemical Determinants for Long Circulation
| Property | Optimal Range for Long Circulation | Protocol Reference |
|---|---|---|
| Hydrodynamic Diameter | 80-150 nm | Protocol 2.1 |
| Polydispersity Index (PDI) | <0.2 | Protocol 2.1 |
| Zeta Potential | -10 to -30 mV | Protocol 2.2 |
| FcRn Binding Affinity (Kd) | ~0.3-1 µM (pH 6.0) | Surface Plasmon Resonance (SPR) |
SPARC-Mediated Tumor Targeting
Secreted Protein Acidic and Rich in Cysteine (SPARC) is an extracellular matrix glycoprotein overexpressed in many aggressive tumors (e.g., pancreatic, melanoma). It binds albumin with high affinity (Kd ~ 1 µM), facilitating the accumulation and retention of albumin-bound therapeutics in the tumor microenvironment. This active targeting complements passive EPR-driven delivery.
Table 3: SPARC Expression and Targeting Efficacy Across Cancers
| Cancer Type | SPARC Expression Level (IHC Score) | Correlation with Albumin-NP Accumulation (Fold Increase vs. Low SPARC) | Key Clinical Evidence |
|---|---|---|---|
| Pancreatic Ductal Adenocarcinoma | High (≥2+) | 2.5-4.0 fold | Abraxane + gemcitabine improves survival in metastatic PDAC. |
| Triple-Negative Breast Cancer | Moderate to High | 1.8-3.2 fold | Preclinical models show enhanced doxorubicin delivery. |
| Glioblastoma | High in tumor periphery | 2.0-3.5 fold (in murine models) | SPARC facilitates blood-brain barrier transcytosis. |
| Melanoma | High | 2.2-3.8 fold | Nab-paclitaxel shows activity in metastatic melanoma. |
Experimental Protocols
Protocol 2.1: Synthesis and Characterization of HSA Nanoparticles via Desolvation
Objective: To prepare monodisperse, drug-loaded HSA nanoparticles. Materials: HSA (≥96%), Anhydrous Ethanol, Glutaraldehyde (8% aqueous), Drug (e.g., Paclitaxel), Magnetic Stirrer, Ultrasonic Probe. Procedure:
- Dissolve 100 mg HSA in 2 mL deionized water (50 mg/mL). Adjust pH to 8.5-9.0 using 0.1M NaOH.
- Under constant stirring (500 rpm), slowly add 8 mL of anhydrous ethanol (1:4 v/v ratio) dropwise using a syringe pump (1 mL/min). A milky opalescence indicates nanoparticle formation.
- For drug loading, add 5-10 mg of hydrophobic drug (in DMSO or ethanol) to the HSA solution before desolvation.
- Crosslink the formed NPs by adding 50 µL of 8% glutaraldehyde and stir for 12 hours at room temperature.
- Purify NPs by centrifugation at 15,000 x g for 20 minutes. Wash pellet 3x with deionized water to remove ethanol and unreacted crosslinker.
- Resuspend the final NP pellet in 2 mL PBS (pH 7.4) and store at 4°C.
- Characterize size and PDI using Dynamic Light Scattering (DLS): dilute sample 1:50 in PBS, measure in triplicate at 25°C.
Protocol 2.2: Determination of Zeta Potential
Objective: To measure the surface charge of albumin nanoparticles. Materials: Zeta Potential Analyzer, Folded Capillary Cell, PBS (1x, pH 7.4). Procedure:
- Dilute the purified NP suspension from Protocol 2.1 in 1x PBS to a final conductivity of 0.5-1.5 mS/cm. Avoid using pure water.
- Inject the sample into a clean, folded capillary cell, ensuring no air bubbles.
- Insert the cell into the instrument and set the temperature to 25°C.
- Run the measurement in automatic mode (typically 10-100 runs). The Smoluchowski model is applicable for aqueous, moderate ionic strength solutions.
- Report the zeta potential as the mean ± standard deviation of at least three independent measurements.
Protocol 2.3: Evaluating SPARC-Dependent UptakeIn Vitro
Objective: To confirm SPARC-mediated endocytosis of albumin NPs. Materials: SPARC-high (e.g., Panc-1) and SPARC-low/null (e.g., MIA PaCa-2) cell lines, Fluorescently-labeled HSA NPs (e.g., FITC-HSA), Anti-SPARC neutralizing antibody, Flow Cytometer. Procedure:
- Seed cells in 12-well plates at 2x10⁵ cells/well and culture for 24 hours.
- Pre-treat one set of SPARC-high wells with 20 µg/mL anti-SPARC antibody for 1 hour.
- Replace medium with fresh medium containing FITC-HSA NPs (100 µg/mL equivalent HSA). Incubate for 2-4 hours at 37°C.
- Wash cells 3x with cold PBS, trypsinize, and resuspend in PBS containing 1% FBS.
- Analyze cellular fluorescence intensity (FITC channel) using flow cytometry for 10,000 events per sample.
- Compare mean fluorescence intensity (MFI) of: (i) SPARC-high cells + NPs, (ii) SPARC-high cells + antibody + NPs, (iii) SPARC-low cells + NPs. A significant reduction in MFI in group (ii) confirms SPARC-mediated uptake.
Diagrams
Diagram 1: SPARC-Mediated Tumor Cell Targeting Pathway
Diagram 2: FcRn-Mediated Recycling vs. Degradation
Diagram 3: HSA Nanoparticle Synthesis and Analysis Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
Table 4: Essential Materials for Albumin-Based Cancer Drug Delivery Research
| Item / Reagent | Function / Purpose | Example Vendor / Catalog Consideration |
|---|---|---|
| Human Serum Albumin (Fatty Acid-Free) | Core material for NP formulation; ensures reproducibility. | Sigma-Aldrich (A3782), Octapharma (clinical grade). |
| SPARC Recombinant Protein & Antibodies | For binding/blocking studies to validate targeting mechanism. | R&D Systems (941-SP), Abcam (anti-SPARC ab[#]). |
| FcRn Recombinant Protein | For in vitro studies of recycling kinetics and binding affinity. | Sino Biological (10249-H08H), AcroBiosystems. |
| Near-Infrared (NIR) Dyes (e.g., DIR, Cy7) | For labeling albumin to track biodistribution and tumor accumulation in vivo. | LI-COR, Lumiprobe. |
| DLS/Zeta Potential Analyzer | Critical instrument for characterizing NP size, PDI, and surface charge. | Malvern Panalytical Zetasizer Nano series. |
| Dialysis Membranes (MWCO 10-100 kDa) | For purification and drug release studies. | Spectrum Labs, Float-A-Lyzer G2. |
| Matrigel | For establishing 3D tumor spheroid models to better mimic EPR. | Corning (356231). |
| IVIS Imaging System | For non-invasive, longitudinal in vivo imaging of fluorescent NP distribution. | PerkinElmer. |
Within the broader research on albumin-based nanoparticles (ABNs) for oncology, a principal objective is to engineer delivery platforms that surmount the intrinsic pharmaceutical limitations of potent chemotherapeutic and targeted payloads. Many promising anti-cancer agents suffer from poor aqueous solubility, chemical instability in physiological environments, and dose-limiting systemic toxicities. ABNs, leveraging endogenous albumin pathways, provide a versatile and biocompatible strategy to encapsulate, protect, and selectively deliver these challenging payloads, thereby enhancing their therapeutic index.
Application Notes: Quantitative Advantages of Albumin Nanotechnology
Table 1: Impact of Albumin-Based Formulations on Payload Properties
| Payload Challenge | Conventional Formulation | Albumin-Based Nanoparticle (ABN) Solution | Quantifiable Improvement (Example) | Key Mechanism |
|---|---|---|---|---|
| Aqueous Solubility | < 0.1 mg/mL (e.g., Paclitaxel) | > 10 mg/mL in final injectable | >100-fold increase in deliverable concentration | Hydrophobic core encapsulation; surface stabilization by albumin. |
| Plasma Stability (t½) | Rapid degradation (<10 min) for some oligonucleotides | Significant prolongation (e.g., >2 hours) | >12-fold increase in circulation half-life | Albumin shielding against nucleases; reduced renal clearance. |
| Off-Target Toxicity | High incidence of severe neutropenia, neuropathy | Reduced severity and frequency | ~50% reduction in severe neutropenia (nab-paclitaxel vs. solvent-based) | Enhanced tumor targeting via SPARC & EPR effect; avoidance of toxic solvents. |
| Tumor Accumulation (%ID/g) | Low (< 2% ID/g at 24h) | Enhanced accumulation (e.g., 5-10% ID/g) | 2-5 fold increase in tumor AUC | Passive (EPR) and active (gp60/SPARC-mediated) targeting. |
| Therapeutic Index (TI) | Narrow TI limits dose | Expanded TI allows higher, more effective dosing | MTD increased by up to 70% for some payloads | Improved pharmacokinetics and biodistribution. |
Experimental Protocols
Protocol: Preparation of Drug-Loaded Albumin Nanoparticles via Desolvation
Objective: To prepare stable, homogeneous albumin nanoparticles loaded with a hydrophobic drug (e.g., paclitaxel, curcumin).
Materials & Reagents:
- Bovine Serum Albumin (BSA) or Human Serum Albumin (HSA)
- Anhydrous ethanol (desolvating agent)
- Glutaraldehyde (8% aqueous solution, cross-linker)
- Drug payload (hydrophobic)
- Phosphate Buffered Saline (PBS, pH 7.4)
- Magnetic stirrer, syringe pump, ultracentrifuge.
Procedure:
- Dissolve 100 mg of albumin in 2 mL of 10 mM NaCl solution under constant magnetic stirring (500 rpm).
- Adjust the solution pH to 8.5-9.0 using 0.1M NaOH.
- Dissolve the hydrophobic drug (e.g., 10 mg paclitaxel) in 8 mL of anhydrous ethanol.
- Using a syringe pump, add the ethanolic drug solution dropwise (1 mL/min) to the albumin solution under continuous stirring. The solution will become opalescent due to nanoparticle formation.
- After complete addition, continue stirring for 1 hour to allow for drug trapping.
- For cross-linking, add 50 µL of 8% glutaraldehyde and stir for 12-24 hours at room temperature.
- Purify the nanoparticles by centrifugation at 15,000 x g for 20 minutes, discard the supernatant, and resuspend the pellet in PBS. Repeat 3x.
- Characterize particle size (DLS), zeta potential, and drug loading efficiency (HPLC).
Protocol: Assessing Plasma Stability and Drug Release
Objective: To evaluate the stability of ABNs in plasma and the kinetics of drug release.
Procedure:
- Incubate 1 mL of purified ABN suspension (1 mg/mL drug equivalent) with 4 mL of human or fetal bovine serum (37°C, gentle shaking).
- At predetermined time points (0, 0.5, 1, 2, 4, 8, 24 h), withdraw 500 µL aliquots.
- Immediately centrifuge aliquots at 15,000 x g for 10 min to separate nanoparticles from free drug/protein.
- Analyze the supernatant for free drug concentration using HPLC to determine premature release.
- To assess nanoparticle integrity, measure the size and PDI of the pellet resuspended in PBS via DLS at each major time point.
- Generate a drug release profile and compare to release in PBS (control).
Protocol: In Vitro Cytotoxicity and Therapeutic Index Assessment
Objective: To compare the cytotoxicity and selectivity of free drug vs. ABN-formulated drug.
Procedure:
- Culture cancer cell lines (e.g., MCF-7, MDA-MB-231) and a non-cancerous cell line (e.g., MCF-10A) in appropriate media.
- Seed cells in 96-well plates at 5,000 cells/well and allow to adhere for 24 h.
- Treat cells with a concentration range (e.g., 0.1 nM - 100 µM) of free drug, empty ABNs, and drug-loaded ABNs. Include vehicle controls.
- Incubate for 48-72 hours.
- Perform an MTT or similar viability assay. Measure absorbance and calculate % viability.
- Determine IC50 values for each formulation in each cell line.
- Calculate a Selectivity Index (SI) as: SI = IC50 (non-cancerous cells) / IC50 (cancerous cells). A higher SI for ABNs indicates an improved therapeutic index.
Visualizations
Title: ABNs Overcome Drug Delivery Hurdles
Title: ABN Synthesis by Desolvation
Title: ABN Tumor Targeting Pathways
The Scientist's Toolkit
Table 2: Essential Research Reagents & Materials for ABN Development
| Item | Function in Research | Key Consideration |
|---|---|---|
| Human Serum Albumin (HSA) | The primary building block for clinically relevant ABNs; ensures biocompatibility and functional receptor binding. | Use endotoxin-free, fatty-acid poor grade for reproducible formulation. |
| Desolvating Agent (Ethanol/ Acetone) | Induces albumin conformational change and aggregation to form the nanoparticle matrix. | Must be anhydrous; addition rate and pH critically control particle size. |
| Cross-linker (Glutaraldehyde) | Stabilizes albumin nanoparticles via amine group cross-linking, preventing dissolution in vivo. | Concentration and reaction time determine degree of cross-linking & drug release rate. |
| Model Hydrophobic Payload (e.g., Paclitaxel, Curcumin) | A challenging compound used to develop and optimize the encapsulation protocol. | High purity; establish validated HPLC-UV/FL method for loading & release analysis. |
| Dynamic Light Scattering (DLS) Instrument | Measures hydrodynamic diameter, polydispersity index (PDI), and zeta potential of ABNs. | Essential for quality control; sample must be adequately filtered and diluted. |
| Dialysis Membranes (MWCO 10-100 kDa) | Used for purification or in drug release studies to separate free drug from nanoparticles. | Choose MWCO well below albumin molecular weight but above drug's. |
| SPARC Protein / Anti-gp60 Antibody | Tools to validate and study active targeting pathways in cellular and in vivo models. | Confirm activity and specificity for functional binding/uptake inhibition assays. |
| Simulated Biological Fluids (PBS, Serum) | Used for stability, drug release, and protein corona studies under physiologically relevant conditions. | Use fresh or properly stored serum to avoid degradation products affecting results. |
Application Notes
Human Serum Albumin (HSA) nanoparticles represent a versatile, biodegradable, and non-immunogenic platform for cancer drug delivery. Recent advancements focus on creating hybrid and surface-engineered systems to overcome biological barriers, enhance tumor targeting, and improve therapeutic efficacy. The core applications are driven by HSA's inherent ability to bind diverse ligands, its accumulation in tumors via the Enhanced Permeability and Retention (EPR) effect, and its interaction with the gp60 receptor (albondin) and SPARC protein in the tumor microenvironment.
1.1 Core HSA Nanoparticle Systems Standard HSA nanoparticles are primarily fabricated via desolvation or emulsion methods, encapsulating hydrophobic chemotherapeutics (e.g., paclitaxel, in the commercial formulation Abraxane). These systems provide improved solubility and reduced systemic toxicity compared to conventional solvent-based formulations.
1.2 Hybrid HSA Systems Hybrid systems integrate HSA with other organic or inorganic materials to create multifunctional carriers.
- HSA-Inorganic Hybrids: Combination with mesoporous silica, gold nanoparticles, or iron oxide. Silica provides high drug loading and stability, gold enables photothermal therapy, and iron oxide allows for magnetic targeting and MRI contrast.
- HSA-Polymer/Lipid Hybrids: Conjugation or co-assembly with polymers (e.g., PEG, poly(lactide-co-glycolide)) or lipids enhances circulatory half-life, modifies drug release kinetics, and enables combination therapy.
1.3 Surface-Engineered HSA Systems Surface modification is a critical strategy for active targeting and stealth properties.
- Ligand Attachment: Covalent conjugation or affinity-based binding of targeting moieties (e.g., folic acid, peptides like RGD, monoclonal antibodies) to HSA nanoparticles directs them to overexpressed receptors on cancer cells.
- PEGylation: Grafting polyethylene glycol (PEG) chains shields nanoparticles from opsonization and clearance by the mononuclear phagocyte system (MPS), prolonging circulation.
- Cell Membrane Coating: Coating HSA nanoparticles with membranes derived from red blood cells or cancer cells imparts immune evasion and homologous targeting capabilities.
The quantitative performance of these systems is summarized in Table 1.
Table 1: Comparative Performance of HSA Nanoparticle Systems
| System Type | Example Composition | Avg. Size (nm) | Avg. PDI | Drug Loading (%) | Key Functional Outcome |
|---|---|---|---|---|---|
| Core HSA | HSA-Paclitaxel (Abraxane) | 130 | 0.1 | ~10 | Improved solubility & reduced toxicity vs. Cremophor EL |
| Hybrid: Inorganic | HSA-Coated Mesoporous Silica + Doxorubicin | 180 | 0.15 | 15-25 | Sustained release, high loading capacity |
| Hybrid: Polymer | HSA-PEG-PLGA Core-Shell + Curcumin | 150 | 0.12 | 8-12 | Prolonged circulation (t1/2 > 12h in mice), enhanced stability |
| Surface-Engineered: Targeted | HSA-Folic Acid + Methotrexate | 110 | 0.18 | ~9 | 3-5x increased cellular uptake in FR+ cells vs. non-targeted |
| Surface-Engineered: Stealth | PEGylated HSA + SN-38 | 120 | 0.1 | 7-10 | Reduced MPS uptake (<15% vs. 60% for unmodified) |
Detailed Experimental Protocols
Protocol: Preparation of Doxorubicin-Loaded HSA Nanoparticles via Desolvation
Objective: To fabricate stable, drug-loaded core HSA nanoparticles.
Materials:
- Human Serum Albumin (HSA, ≥96%, fatty acid-free)
- Doxorubicin hydrochloride (DOX·HCl)
- Ethanol (absolute, desolvating agent)
- Glutaraldehyde (8% aqueous solution, crosslinker)
- Sodium chloride (NaCl)
- ʟ-Histidine (stabilizer)
- pH meter, magnetic stirrer, syringe pump, centrifuge.
Procedure:
- Dissolve 100 mg HSA in 2.0 mL of 10 mM NaCl solution. Adjust pH to 8.5 using 0.1M NaOH.
- Dissolve 10 mg DOX·HCl in 1.0 mL deionized water. Add dropwise to the HSA solution under constant stirring (500 rpm) at room temperature (RT). Incubate for 30 min in the dark to allow drug binding.
- Using a syringe pump, gradually add 8.0 mL of ethanol (desolvating agent) to the stirred HSA-DOX solution at a constant rate of 1.0 mL/min. The solution will become opalescent, indicating nanoparticle formation.
- To stabilize the nanoparticles, add 52 µL of 8% glutaraldehyde solution (to achieve a 50% molar ratio to HSA lysine residues). Stir for 12 hours at RT.
- Purify nanoparticles by centrifugation at 15,000 × g for 15 minutes. Wash the pellet twice with 70% ethanol followed by water to remove unreacted crosslinker and free drug.
- Re-disperse the final nanoparticle pellet in 5 mL of 1% ʟ-histidine solution (pH 6.5) via gentle sonication (30 sec, 30% amplitude). Filter through a 0.22 µm membrane.
- Characterization: Determine particle size and PDI by dynamic light scattering (DLS). Quantify drug loading via HPLC after dissolving nanoparticles in 0.1M NaOH with 1% SDS.
Protocol: Surface Engineering of HSA Nanoparticles with Folic Acid for Targeting
Objective: To conjugate folic acid (FA) to pre-formed HSA nanoparticles for folate receptor-mediated targeting.
Materials:
- Pre-formed, purified HSA nanoparticles (from Protocol 2.1, unloaded or drug-loaded)
- Folic Acid (FA)
- N-Hydroxysuccinimide (NHS)
- N-(3-Dimethylaminopropyl)-N′-ethylcarbodiimide hydrochloride (EDC)
- Dimethyl sulfoxide (DMSO), anhydrous
- 0.1 M MES buffer (pH 5.5), PBS (pH 7.4)
Procedure:
- Activation of Folic Acid: Dissolve 5 mg FA and 10 mg NHS in 2 mL anhydrous DMSO. Add 15 mg EDC. React for 3 hours at RT in the dark with stirring to form FA-NHS ester.
- Conjugation: Resuspend 50 mg of purified HSA nanoparticles in 10 mL of 0.1 M MES buffer (pH 5.5). Add the activated FA solution dropwise (10:1 molar ratio of FA:HSA). React for 6 hours at 4°C with gentle stirring.
- Purification: Centrifuge the reaction mixture at 15,000 × g for 15 min. Wash the pellet three times with PBS (pH 7.4) to remove unconjugated FA and reaction by-products.
- Characterization: Resuspend FA-HSA nanoparticles in PBS. Confirm conjugation via UV-Vis spectroscopy (characteristic peak of FA at ~365 nm) or by measuring the change in zeta potential (typically becomes more negative). Validate targeting efficacy via cellular uptake studies in folate receptor-positive (e.g., KB) and negative cell lines.
Visualizations
Evolution of HSA Nanoparticle Platforms
Active Targeting & Intracellular Pathway of FA-HSA NPs
Research Reagent Solutions Toolkit
Table 2: Essential Materials for HSA Nanoparticle Research
| Item | Function/Application | Example Product/Specification |
|---|---|---|
| Fatty Acid-Free HSA | Core nanoparticle matrix; ensures reproducible formulation without lipid interference. | Sigma-Aldrich, A3782, ≥96% (agarose gel electrophoresis). |
| Crosslinker (Glutaraldehyde) | Stabilizes HSA nanoparticles by inter- and intra-molecular crosslinking of lysine residues. | Thermo Fisher, G5882, 8% aqueous solution, EM grade. |
| Desolvating Agent (Ethanol) | Induces conformational changes in HSA, leading to nanoparticle aggregation/formation. | Sigma-Aldrich, 459836, absolute for molecular biology, ≥99.8%. |
| NHS/EDC Coupling Kit | Standard chemistry for covalent conjugation of targeting ligands (e.g., FA, peptides) to HSA surface. | Thermo Fisher, Pierce EDC Crosslinking Kit. |
| Functionalized PEG | Imparts "stealth" properties, prolonging circulation half-life (e.g., NHS-PEG-Mal for conjugation). | Creative PEGWorks, PG2-MLNS-5k, Maleimide-PEG-NHS, 5 kDa. |
| Targeting Ligand (Folic Acid) | Model active targeting agent for folate receptor-positive cancers. | Sigma-Aldrich, F7876, ≥97%. |
| Characterization Standards | For accurate DLS measurement of nanoparticle size and zeta potential. | Malvern Panalytical, Zeta Potential Transfer Standard, DTS1235. |
| Dialysis Membrane | Purification of nanoparticles and assessment of drug release kinetics. | Spectrum Labs, 132676, Float-A-Lyzer G2, 100 kDa MWCO. |
| SPARC Protein | Used in in vitro studies to validate SPARC-mediated targeting and uptake of HSA nanoparticles. | R&D Systems, 941-SP-050, Human SPARC, HEK293-derived. |
Application Notes: Marketed & Clinical-Stage Albumin-Nanoparticle Formulations
Note 1.1: Abraxane (Paclitaxel-Albumin Nanoparticles) The first FDA-approved (2005) albumin-bound nanoparticle, utilizing the 130 nm nab technology platform. It leverages endogenous albumin pathways (gp60/SPARC) for tumor targeting, enabling solvent-free administration with higher paclitaxel doses and improved safety over Cremophor-EL formulations.
Note 1.2: Emerging Clinical Candidates Post-Abraxane, the field has expanded to include novel chemotherapeutics, combination agents, and biologics. These candidates aim to improve drug stability, tumor penetration, and targeting specificity, often incorporating surface modifications or co-loaded agents.
Table 1: Marketed & Clinical-Stage Albumin-Based Nanoparticle Formulations for Oncology
| Formulation (Generic Name) | Drug Payload | Indication(s) (Phase) | Key Clinical Outcome/Status | Average Particle Size (nm) | Key Differentiator vs. Abraxane |
|---|---|---|---|---|---|
| Abraxane (nab-paclitaxel) | Paclitaxel | NSCLC, Pancreatic Cancer, Breast Cancer (Marketed) | Improved response rate & safety vs. solvent-based paclitaxel. | 130 | First-in-class, SPARC-mediated targeting. |
| Nab-rapamycin (ABI-009) | Sirolimus | Malignant PEComa, Solid Tumors (Marketed/Phase II) | ~39% ORR in advanced PEComa (NCT02494570). | ~100 | mTOR inhibitor delivery, orphan drug designation. |
| Aldoxorubicin (INNO-206) | Doxorubicin (acid-sensitive linker) | Soft Tissue Sarcoma (Phase III) | Improved PFS vs. investigator's choice therapy. | ~100 | Acid-triggered release in tumor microenvironment. |
| Nab-docetaxel (CPC634) | Docetaxel | Advanced Solid Tumors (Phase I) | Reduced systemic toxicity vs. Taxotere. | 120-140 | Solvent-free docetaxel, improved tolerability. |
| Anti-PD-L1 nab-paclitaxel | Paclitaxel + aPD-L1 | Triple-Negative Breast Cancer (Preclinical/Phase I) | Enhanced antitumor immunity in murine models. | ~150 | Co-delivery of chemo- & immunotherapy. |
Table 2: Key In Vitro & Preclinical Efficacy Parameters for Select Candidates
| Candidate (Model) | Cell Line/Animal Model | Key Metric | Reported Value | Reference Year (est.) |
|---|---|---|---|---|
| Aldoxorubicin (In Vivo) | MX-1 breast cancer xenograft | Tumor Growth Inhibition (TGI) | 98% TGI | 2023 |
| Nab-rapamycin (In Vitro) | TSC2-null mouse cell line | IC50 (Proliferation) | 0.5 nM | 2022 |
| Anti-PD-L1 nab-paclitaxel (In Vivo) | 4T1 syngeneic mouse model | Tumor Volume Reduction (Day 21) | ~80% reduction vs. control | 2023 |
| Nab-docetaxel (CPC634) (In Vivo) | PC3 prostate cancer xenograft | Tumor Drug Concentration (24h) | 3.5-fold > conventional docetaxel | 2021 |
Detailed Experimental Protocols
Protocol 3.1: Preparation of Albumin-Based Nanoparticles via High-Pressure Homogenization (nab-Technology Mimic)
- Objective: To fabricate drug-loaded albumin nanoparticles.
- Materials: Bovine Serum Albumin (BSA) or HSA, Drug (e.g., Paclitaxel), Chloroform, Phosphate Buffered Saline (PBS), High-pressure homogenizer (e.g., Avestin).
- Procedure:
- Emulsification: Dissolve 50 mg of drug and 250 mg of albumin in 10 mL of chloroform and 50 mL of aqueous PBS (pH 7.4), respectively. Pre-homogenize the mixture using a high-shear mixer for 2 min at 10,000 rpm to form a coarse emulsion.
- High-Pressure Homogenization: Process the coarse emulsion through a high-pressure homogenizer for 10 cycles at 15,000 psi, maintaining the temperature below 25°C using a cooling jacket.
- Solvent Evaporation: Stir the resulting nanoemulsion overnight at room temperature in a fume hood to evaporate chloroform completely.
- Purification: Centrifuge the suspension at 20,000 x g for 30 min, wash the pellet with PBS twice, and re-suspend in an appropriate buffer.
- Sterilization: Filter the final nanoparticle suspension through a 0.22 µm sterile syringe filter. Store at 4°C for short-term use.
Protocol 3.2: In Vitro Cytotoxicity Assessment (MTT Assay)
- Objective: To evaluate the cytotoxic potency of albumin-nanoparticle formulations.
- Materials: Cancer cell line (e.g., MCF-7, PC-3), Nanoparticle formulations, MTT reagent (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide), DMSO, Cell culture medium, 96-well plate, Microplate reader.
- Procedure:
- Cell Seeding: Seed cells in a 96-well plate at 5x10³ cells/well in 100 µL medium. Incubate for 24 h (37°C, 5% CO₂).
- Treatment: Prepare serial dilutions of the nanoparticle drug and its free drug equivalent. Replace medium with 100 µL of treatment medium per well. Include untreated control wells (medium only). Incubate for 48-72 h.
- MTT Incubation: Add 20 µL of MTT solution (5 mg/mL in PBS) to each well. Incubate for 4 h.
- Solubilization: Carefully aspirate the medium and add 150 µL of DMSO to each well to dissolve the formed formazan crystals.
- Absorbance Measurement: Shake the plate gently and measure the absorbance at 570 nm (reference 630 nm) using a microplate reader.
- Analysis: Calculate cell viability: % Viability = (Abssample/Abscontrol) x 100. Determine IC50 values using non-linear regression analysis (e.g., GraphPad Prism).
Diagrams
Diagram 1: Albumin Nanoparticle Tumor Targeting Pathways
Diagram 2: Workflow for Albumin-Nanoparticle R&D
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Albumin-Based Nanoparticle Cancer Research
| Item | Function in Research | Example/Catalog Consideration |
|---|---|---|
| Human Serum Albumin (HSA) | The core protein component for nanoparticle formation; preferred for translational studies. | Sigma-Aldrich A9731 (Fatty acid-free, ≥96%). |
| nab-Technology Mimic Kits | Provides standardized reagents and protocols for bench-scale nanoparticle preparation. | Particle Works HSA-NP Starter Kit (Research-grade). |
| SPARC Recombinant Protein | Used in binding studies to validate the active targeting mechanism of formulations. | R&D Systems 941-SP-050. |
| gp60/Caveolin-1 Antibody | For IHC or Western Blot to assess target expression in cell/animal models. | Cell Signaling Technology 3267S. |
| Simulated Body Fluid (SBF) | To test nanoparticle stability and drug release kinetics under physiological conditions. | Biorelevant.com FaSSIF/FeSSIF media. |
| Transwell Co-culture Systems | To model endothelial transport and tumor penetration in vitro. | Corning 3460 (24-well, 0.4 µm pore). |
| Near-IR Fluorescent Dye (e.g., DIR) | For in vivo imaging to track nanoparticle biodistribution and tumor accumulation. | Invitrogen D12731. |
| Size Exclusion Chromatography (SEC) Columns | For purification and aggregate analysis of albumin nanoparticle formulations. | Tosoh Bioscience TSKgel G3000SWxl. |
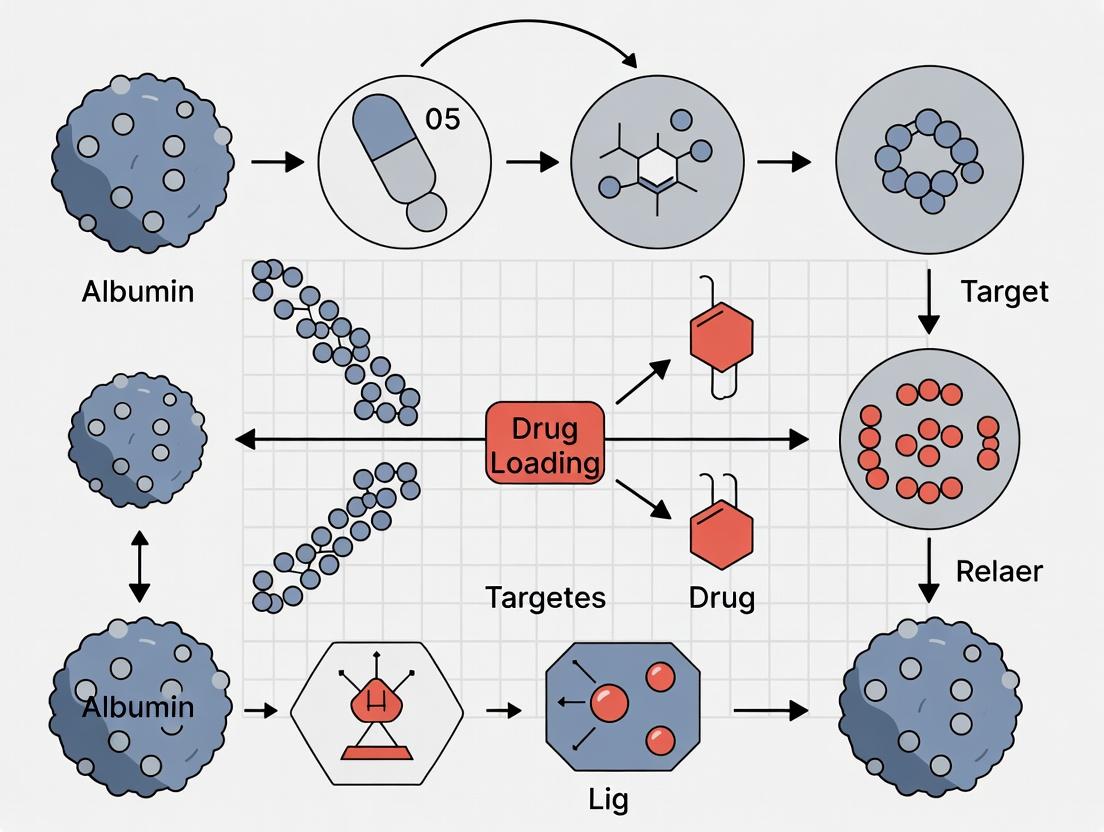
From Bench to Bedside: Synthesis, Loading, and Targeted Delivery Strategies
Within the thesis framework of developing albumin-based nanoparticles for targeted cancer drug delivery, the selection and optimization of a fabrication technique is paramount. This application note details three core methodologies—desolvation, emulsification, and self-assembly—that are fundamental to producing albumin nanoparticles with controlled size, stability, and drug release profiles. Each protocol offers distinct advantages for encapsulating chemotherapeutics, from hydrophobic small molecules to biologics.
Desolvation Protocol
Desolvation is a widely used, straightforward method for fabricating protein nanoparticles by inducing conformational changes through solvent displacement.
Key Application in Cancer Research
Ideal for the encapsulation of chemotherapeutics like doxorubicin, methotrexate, and gemcitabine. The mild, aqueous-based process helps maintain drug stability.
Detailed Experimental Protocol
- Dissolution: Dissolve 100 mg of human serum albumin (HSA) in 1 mL of deionized water under mild magnetic stirring (300 rpm).
- pH Adjustment: Adjust the solution to pH 7.0-9.0 using 0.1M NaOH.
- Desolvation: Under continuous stirring (500 rpm), slowly add a desolvating agent (e.g., ethanol or acetone) at a controlled rate of 0.5 mL/min until the solution becomes opalescent, indicating nanoparticle formation. Typical volume ratio is 1:1.5 (Albumin solution:Ethanol).
- Cross-linking: Add 5-20 µL of 8% glutaraldehyde (cross-linker) and stir for 12-24 hours to stabilize particles.
- Purification: Centrifuge at 15,000 x g for 20 minutes. Wash the pellet 3x with ethanol/water mixture and resuspend in phosphate-buffered saline (PBS) or water.
- Characterization: Analyze particle size (DLS), zeta potential, and drug loading efficiency (HPLC/UV-Vis).
| Parameter | Typical Range | Impact on Nanoparticle Characteristics |
|---|---|---|
| Albumin Concentration | 50-200 mg/mL | Influences final particle size and yield. |
| pH of Albumin Solution | 7.0 - 9.0 | Higher pH promotes smaller, more uniform particles. |
| Desolvating Agent | Ethanol, Acetone | Ethanol yields more homogeneous particles. |
| Agent Addition Rate | 0.25 - 1.0 mL/min | Slower rate improves size uniformity. |
| Cross-linker (Glutaraldehyde) Amount | 5-20 µL per 100 mg HSA | Determines particle stability and drug release kinetics. |
| Average Particle Size (DLS) | 100 - 300 nm | Tunable based on process parameters. |
| Drug Loading Efficiency (Doxorubicin) | 60 - 85% | Depends on drug-albumin incubation step. |
Diagram: Desolvation Nanoparticle Formation Workflow
Emulsification (High-Pressure Homogenization) Protocol
This technique uses high shear forces to form a water-in-oil (w/o) emulsion, ideal for encapsulating hydrophobic drugs.
Key Application in Cancer Research
Suited for potent, water-insoluble drugs like paclitaxel, docetaxel, and curcumin. Forms the basis for Abraxane-like nanoparticle albumin-bound (nab) technology.
Detailed Experimental Protocol
- Organic Phase Preparation: Dissolve 10 mg of hydrophobic drug (e.g., paclitaxel) and 100 mg of HSA in 5 mL of dichloromethane (DCM) or chloroform.
- Aqueous Phase Preparation: Prepare 20 mL of a stabilizing surfactant solution (e.g., 1% w/v polyvinyl alcohol) in water.
- Primary Emulsion: Under high-speed homogenization (15,000 rpm, 2 min), add the organic phase to the aqueous phase to form a coarse w/o emulsion.
- High-Pressure Homogenization: Process the coarse emulsion through a high-pressure homogenizer for 5-10 cycles at 10,000-20,000 psi.
- Solvent Evaporation: Stir the fine emulsion overnight at room temperature to evaporate the organic solvent.
- Purification: Centrifuge at 12,000 x g for 15 min. Wash pellets and resuspend in buffer. Sterile filter (0.22 µm).
- Characterization: Determine particle size, PDI, drug content (HPLC), and in vitro drug release.
| Parameter | Typical Range | Impact on Nanoparticle Characteristics |
|---|---|---|
| Albumin:Drug Ratio (w/w) | 5:1 - 15:1 | Critical for achieving high drug loading and stability. |
| Homogenization Pressure | 10,000 - 20,000 psi | Higher pressure reduces particle size and PDI. |
| Number of Homogenization Cycles | 3 - 10 | More cycles reduce size but may denature albumin. |
| Surfactant Concentration (PVA) | 0.5 - 2.0 % w/v | Stabilizes emulsion, prevents aggregation. |
| Average Particle Size (DLS) | 120 - 200 nm | Target for EPR effect. |
| Drug Loading Efficiency (Paclitaxel) | 70 - 95% | Typically high due to hydrophobic interaction. |
| Final Drug Payload | 5 - 15 % w/w | Key efficacy parameter. |
Diagram: High-Pressure Homogenization Process
Self-Assembly Protocol
Self-assembly exploits non-covalent interactions (electrostatic, hydrophobic) to form nanoparticles upon specific triggers like pH change or solvent displacement.
Key Application in Cancer Research
Excellent for co-delivery of multiple drugs (e.g., doxorubicin + siRNA) or creating stimuli-responsive (pH, redox) nanoparticles for tumor microenvironment targeting.
Detailed Experimental Protocol
- Drug-Albumin Conjugate/Complex Formation:
- For electrostatic co-assembly: Mix a cationic HSA derivative (pre-modified) with anionic siRNA in nuclease-free water at a specific N/P ratio (e.g., 10:1). Incubate 30 min at room temperature.
- For hydrophobic drug loading: Incubate HSA with drug in a water-miscible organic solvent (e.g., DMSO). Dilute rapidly into 10x volume of aqueous buffer under vortexing.
- Induction of Self-Assembly: For pH-triggered assembly, dialyze or adjust the mixture against a low-pH buffer (e.g., pH 5.0-6.0) to induce nanoparticle formation.
- Stabilization: Optionally, add trace amounts of cross-linker (e.g., glutaraldehyde) or use disulfide-reducible cross-linking for intracellular drug release.
- Purification: Dialyze against buffer or use tangential flow filtration to remove organic solvents/unbound components.
- Characterization: Analyze size, zeta potential, complexation efficiency (gel retardation assay for nucleic acids), and drug release under simulated physiological vs. tumor conditions.
| Parameter | Typical Range | Impact on Nanoparticle Characteristics |
|---|---|---|
| N/P Ratio (for siRNA) | 5:1 - 20:1 | Balances complexation efficiency and cytotoxicity. |
| Solvent Shift Rate | Rapid (Vortex) vs. Slow (Dialysis) | Faster mixing creates smaller, more uniform particles. |
| pH Trigger Point | 5.0 - 6.5 | Mimics tumor microenvironment or endosomal pH. |
| Cross-linking Type | None, Glutaraldehyde, Disulfide | Controls stability and triggered release profile. |
| Average Particle Size (DLS) | 80 - 250 nm | Highly tunable based on assembly conditions. |
| siRNA Complexation Efficiency | >90% (at N/P 10:1) | Measured by gel electrophoresis. |
| Drug Co-Loading Efficiency | Variable (50-85%) | Depends on drug interaction with albumin. |
Diagram: Self-Assembly Pathways for Albumin Nanoparticles
The Scientist's Toolkit: Essential Research Reagent Solutions
| Reagent/Material | Function in Albumin Nanoparticle Fabrication |
|---|---|
| Human Serum Albumin (HSA) or Bovine Serum Albumin (BSA) | The core biopolymer matrix; biocompatible, biodegradable, and offers multiple binding sites for drugs. |
| Glutaraldehyde (8% aqueous solution) | Cross-linking agent for desolvated particles; stabilizes structure but must be carefully optimized to avoid cytotoxicity. |
| Ethanol (Absolute, HPLC Grade) | Common desolvating agent; induces protein conformational change and nanoparticle precipitation. |
| Dichloromethane (DCM) or Chloroform | Organic solvent for emulsification methods; dissolves hydrophobic drugs and albumin. |
| Polyvinyl Alcohol (PVA, Mw 30-70 kDa) | Surfactant/stabilizer in emulsification; forms a protective layer around nanoparticles during formation. |
| High-Pressure Homogenizer (e.g., Microfluidizer) | Equipment to apply extreme shear forces, reducing emulsion droplet size to the nanoscale. |
| Dialysis Tubing (MWCO 12-14 kDa) | For purifying self-assembled nanoparticles and removing organic solvents or unbound molecules. |
| Dynamic Light Scattering (DLS) / Zeta Potential Analyzer | Essential instrument for characterizing nanoparticle hydrodynamic diameter, PDI, and surface charge. |
This application note details three principal methodologies for loading therapeutic agents into albumin-based nanoparticles, a cornerstone technology in modern cancer drug delivery research. Albumin's biocompatibility, biodegradability, and inherent tumor-targeting via the gp60-SPARC pathway make it an ideal carrier. The choice of loading method—covalent conjugation, hydrophobic entrapment, or electrostatic binding—directly impacts critical parameters such as drug loading capacity (DLC), encapsulation efficiency (EE), release kinetics, and in vivo biodistribution. Within the context of a doctoral thesis on optimizing albumin nanocarriers, this document provides detailed protocols and comparative analysis to guide experimental design.
Covalent Conjugation
Covalent conjugation involves forming stable chemical bonds (e.g., amide, ester, disulfide) between drug molecules and functional groups (e.g., lysine ε-amines, cysteine thiols) on the albumin carrier. This method offers precise control over the drug-to-carrier ratio and high serum stability, but requires chemical modification of the drug and may necessitate a cleavable linker for intracellular drug release.
Protocol: Conjugation of Doxorubicin via pH-Sensitive Hydrazone Linker to Human Serum Albumin (HSA) Nanoparticles
Objective: To synthesize HSA-doxorubicin conjugates with a linker cleavable in the acidic tumor microenvironment.
Materials:
- HSA (≥99%, fatty acid-free)
- Doxorubicin hydrochloride (DOX·HCl)
- Traut's Reagent (2-Iminothiolane)
- 6-Maleimidocaproic acid hydrazide (EMCH) linker
- Dimethyl sulfoxide (DMSO), anhydrous
- Phosphate Buffered Saline (PBS), pH 7.4
- Sephadex G-25 PD-10 desalting columns
- Dialysis membrane (MWCO 10 kDa)
Procedure:
- Thiolation of HSA: Dissolve HSA (50 mg) in 5 mL of PBS (pH 8.0). Add a 20-fold molar excess of Traut's Reagent (from a fresh 10 mg/mL stock in PBS) and react for 1 hour at room temperature under gentle stirring. Purify thiolated HSA (HSA-SH) using a PD-10 column equilibrated with PBS (pH 7.0). Determine thiol concentration using Ellman's assay.
- Activation of DOX: Dissolve DOX·HCl (5 mg) in 1 mL anhydrous DMSO. Add a 1.2 molar equivalent of EMCH linker and a catalytic amount of triethylamine. React in the dark for 6 hours at room temperature.
- Conjugation: Slowly add the activated DOX solution to the purified HSA-SH solution under constant vortexing. Adjust the mixture to pH 6.5-7.0. Allow the reaction to proceed in the dark for 24 hours at 4°C.
- Purification: Dialyze the reaction mixture against distilled water (4 L, changed 3 times over 24 hours) using a 10 kDa MWCO membrane to remove unreacted DOX and linker byproducts. Lyophilize the purified conjugate.
- Nanoparticle Formation: Reconstitute the lyophilized HSA-DOX conjugate in PBS and form nanoparticles via desolvation (using ethanol) or high-pressure homogenization, followed by stabilization with glutaraldehyde or thermal crosslinking.
Key Evaluation Parameters: DLC (% w/w), conjugation efficiency (%), particle size (DLS), zeta potential, in vitro drug release at pH 7.4 and 5.0.
Diagram: Covalent Conjugation & Intracellular Release Workflow
Diagram Title: Covalent Conjugation and Intracellular Drug Release Pathway
Hydrophobic Entrapment
This method exploits the hydrophobic binding pockets of albumin (e.g., Sudlow sites I and II) to incorporate lipophilic drugs. It is the foundational mechanism for the clinically approved nanoparticle albumin-bound (nab) technology (e.g., Abraxane). The process typically involves forced co-assembly of drug and albumin under controlled conditions.
Protocol: Preparation of Paclitaxel-Loaded Albumin Nanoparticles via High-Pressure Homogenization (nab-Technology Mimic)
Objective: To fabricate albumin nanoparticles with high payloads of hydrophobic paclitaxel (PTX).
Materials:
- HSA (≥99%)
- Paclitaxel (PTX)
- Chloroform (or other volatile organic solvent)
- High-pressure homogenizer (e.g., Microfluidizer)
- Rotary evaporator
- Ultrapure water (18.2 MΩ·cm)
- 0.22 μm sterile syringe filters
Procedure:
- Drug Solution: Dissolve PTX (10 mg) in 2 mL of chloroform.
- Albumin Solution: Dissolve HSA (100 mg) in 10 mL of ultrapure water.
- Emulsification: Slowly add the PTX/chloroform solution to the HSA solution under high-speed probe sonication (e.g., 70% amplitude, 2 min on ice) to form a crude oil-in-water emulsion.
- Homogenization: Pass the coarse emulsion through a high-pressure homogenizer for 5-10 cycles at a pressure of 15,000-20,000 psi. This step reduces particle size and uniformly disperses the drug within the albumin matrix.
- Solvent Removal: Transfer the homogenized emulsion to a rotary evaporator to gently remove the organic solvent at 30°C (or under a stream of nitrogen if scale is small).
- Sterilization & Collection: Filter the resulting nanoparticle suspension through a 0.22 μm syringe filter. The nanoparticles can be used directly or lyophilized with a suitable cryoprotectant (e.g., 2% sucrose) for storage.
Key Evaluation Parameters: DLC (%), EE (%), particle size and PDI (DLS), morphology (TEM), sterility, in vitro release profile in PBS with surfactants (e.g., 0.1% Tween 80).
Electrostatic Binding
Electrostatic loading relies on the ionic interaction between charged drug molecules and oppositely charged amino acid residues on the albumin surface. This method is suitable for hydrophilic, ionizable drugs (e.g., nucleic acids, certain antibiotics, positively charged chemotherapeutics like doxorubicin at acidic pH).
Protocol: Electrostatic Complexation of siRNA with Cationized Albumin Nanoparticles
Objective: To formulate and characterize nanoparticles for siRNA delivery by exploiting electrostatic interactions.
Materials:
- HSA
- Ethylenediamine (EDA) or Polyethylenimine (PEI, 2 kDa) for cationization
- 1-Ethyl-3-(3-dimethylaminopropyl)carbodiimide (EDC)
- N-Hydroxysuccinimide (NHS)
- siRNA (e.g., against GFP or a target oncogene)
- Nuclease-free water and buffers
- Agarose gel electrophoresis system
Procedure:
- Cationization of HSA: Dissolve HSA (100 mg) and EDA (500 mM) in MES buffer (0.1 M, pH 5.5). Add EDC (50 mM) and NHS (25 mM) to activate carboxyl groups on HSA. React for 3-4 hours at room temperature. Dialyze extensively against distilled water (MWCO 10 kDa) and lyophilize to obtain cationized HSA (cHSA).
- Nanoparticle Formation: Prepare cHSA nanoparticles via desolvation. Dissolve cHSA (20 mg) in 2 mL of 10 mM NaCl. Adjust pH to 8.0. Add ethanol dropwise (4 mL, 1 mL/min) under constant stirring (550 rpm) until the solution becomes opalescent. Crosslink with 5 μL of 8% glutaraldehyde solution for 12 hours. Purify by centrifugation and resuspend in nuclease-free PBS.
- siRNA Complexation: Prepare varying weight ratios of cHSA nanoparticles to siRNA (e.g., 10:1, 20:1, 40:1). Mix the siRNA (0.1 mg/mL in nuclease-free water) with the nanoparticle suspension by gentle pipetting. Incubate at room temperature for 30 minutes to allow complex formation.
- Gel Retardation Assay: Analyze complex formation using agarose gel electrophoresis (2% gel, 100 V, 30 min). Free siRNA migrates, while fully complexed siRNA is retained in the loading well.
Key Evaluation Parameters: Complexation efficiency (gel assay), particle size & zeta potential before/after complexation, protection from RNase degradation, in vitro gene silencing efficacy.
Diagram: Electrostatic Loading & Cellular Uptake Pathway
Diagram Title: Electrostatic siRNA Loading and Cellular Uptake Mechanism
Table 1: Quantitative Comparison of Drug Loading Methodologies for Albumin Nanoparticles
| Parameter | Covalent Conjugation | Hydrophobic Entrapment (nab-like) | Electrostatic Binding |
|---|---|---|---|
| Typical Drug Load (wt%) | 5 - 15% | 10 - 20% (Up to 50% reported for PTX) | 1 - 10% (highly dependent on drug/nanocarrier MW) |
| Encapsulation Efficiency | 60 - 90% (Depends on reaction yield) | > 90% (for optimized homogenization) | 70 - 99% (at optimal +/- charge ratio) |
| Drug Stability | High (Covalent bond) | High (Kinetically trapped) | Moderate (Subject to ionic strength & competition) |
| Release Trigger | Chemical/enzymatic linker cleavage (pH, redox, enzymes) | Diffusion & matrix erosion | Desorption & ion exchange |
| Suitable Drug Classes | Must have modifiable functional group (-OH, -NH2, -COOH) | Highly hydrophobic (log P > 5) | Charged molecules (siRNA, peptides, ionizable drugs) |
| Clinical Example | Aldoxorubicin (in trials) | Abraxane (Paclitaxel) | Various in preclinical development |
| Key Challenge | Synthetic complexity, potential altered pharmacology | Limited to hydrophobic drugs | Stability in physiological ionic strength |
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 2: Key Reagents and Their Functions in Albumin Nanoparticle Drug Loading Research
| Reagent/Material | Primary Function | Typical Application |
|---|---|---|
| Human Serum Albumin (Fatty Acid-Free) | Primary carrier biomaterial; provides biocompatibility and active targeting potential. | Base material for all nanoparticle formulations. |
| Traut's Reagent (2-Iminothiolane) | Introduces sulfhydryl (-SH) groups onto primary amines for thiol-based conjugation. | Covalent Conjugation (Step 1: Thiolation of albumin). |
| Heterobifunctional Crosslinkers (e.g., EMCH, SMCC) | Spacer molecules with two different reactive ends (e.g., NHS-ester & maleimide). | Covalent Conjugation (Step 2: Linking drug to albumin). |
| High-Pressure Homogenizer (Microfluidizer) | Applies intense shear forces to produce uniform, sub-micron emulsions and dispersions. | Hydrophobic Entrapment (Key for nab-technology mimic). |
| Desolvating Agent (Anhydrous Ethanol) | Reduces solubility of albumin in water, inducing protein aggregation and nanoprecipitation. | Nanoparticle formation for Covalent & Electrostatic methods. |
| Glutaraldehyde (or Genipin) | Crosslinking agent that stabilizes albumin nanoparticles via amine crosslinks. | Particle hardening post-desolvation or homogenization. |
| Cationizing Agent (e.g., Ethylenediamine + EDC/NHS) | Converts albumin surface carboxyls to primary amines, imparting a positive charge. | Electrostatic Binding (Preparation of cationic carrier). |
| Dynamic Light Scattering (DLS) / Zeta Potential Analyzer | Instrument for measuring nanoparticle hydrodynamic size, PDI, and surface charge. | Critical characterization for all three methodologies. |
Within the paradigm of albumin-based nanoparticle (AbNP) drug delivery, passive targeting via the Enhanced Permeability and Retention (EPR) effect offers limited tumor specificity. Active Targeting 2.0 represents the strategic conjugation of high-affinity, target-specific ligands—peptides, antibodies, and aptamers—to the surface of AbNPs. This ligand decoration enables precision oncology by facilitating receptor-mediated endocytosis into cancer cells, overcoming multidrug resistance, and modulating tumor microenvironment interactions. This document provides application notes and protocols for integrating Active Targeting 2.0 ligands into albumin-based nanoplatforms.
Key Ligand Classes & Quantitative Comparison
Table 1: Comparative Analysis of Targeting Ligands for Albumin Nanoparticle Decoration
| Ligand Class | Typical Size (kDa) | Binding Affinity (Kd) | Conjugation Chemistry to AbNP | Key Advantages | Major Limitations | Example Target (Cancer Type) |
|---|---|---|---|---|---|---|
| Monoclonal Antibodies (mAbs) | ~150 | 0.1-10 nM | NHS-ester, maleimide, click chemistry (DBCO-Azide) | Exceptional specificity, high affinity, well-characterized. | Large size may hinder penetration, immunogenicity, complex production. | HER2 (Breast), EGFR (Colorectal) |
| Antibody Fragments (e.g., scFv) | ~25 | 1-100 nM | Maleimide (via engineered cysteine), Click chemistry. | Improved penetration, reduced immunogenicity, retains specificity. | Lower avidity, potential stability issues, still requires genetic engineering. | CAIX (Renal Cell Carcinoma) |
| Peptides | 1-3 | 100 nM - 10 µM | NHS-ester, maleimide, SPDP. | Small size, good penetration, facile synthesis & modification, low immunogenicity. | Moderate affinity, susceptibility to proteolysis, rapid clearance. | RGD (αvβ3 Integrin, Glioblastoma), NGR (CD13, Tumor Vasculature) |
| Aptamers (DNA/RNA) | 10-30 | 1-100 nM | Thiol-maleimide, NHS-ester (amino-modified), Click chemistry. | Small size, chemical stability, in vitro selection, low immunogenicity. | Susceptible to nuclease degradation (RNA > DNA), potential renal clearance. | AS1411 (Nucleolin, Various), A10 (PSMA, Prostate) |
Experimental Protocols
Protocol 1: Maleimide-Thiol Conjugation of cRGDfK Peptide to Human Serum Albumin (HSA) Nanoparticles
Objective: To decorate pre-formed HSA nanoparticles (e.g., prepared by desolvation or nab-technology) with a cyclic RGD peptide for targeting αvβ3 integrin.
Materials (Research Reagent Solutions Toolkit):
- HSA Nanoparticles: Pre-formed, lyophilized, with surface-exposed free cysteine residues or introduced via Traut's reagent (2-Iminothiolane).
- cRGDfK Peptide: Cyclo(Arg-Gly-Asp-D-Phe-Lys), with a C-terminal cysteine residue (cRGDfK-Cys).
- Maleimide-PEG-NHS Ester (Mal-PEG-NHS, MW 3400): Heterobifunctional linker for stable conjugation.
- Traut's Reagent (2-Iminothiolane): Thiolation reagent for introducing -SH groups onto primary amines.
- PD-10 Desalting Columns: For buffer exchange and removal of unreacted small molecules.
- Ellman's Reagent (DTNB): For quantifying free thiol groups.
- Phosphate Buffered Saline (PBS), pH 7.4, and Degassed PBS: For reaction and purification.
- Zeta Potential & DLS Instrument: For characterizing nanoparticle size and surface charge post-conjugation.
Methodology:
- Thiol Introduction (if required): Resuspend 10 mg of HSA nanoparticles in 1 mL of degassed PBS (pH 7.4). Add a 50-fold molar excess of Traut's Reagent and incubate for 1 hour at 25°C under gentle agitation. Purify thiolated NPs using a PD-10 column equilibrated with degassed PBS. Quantify free thiols using Ellman's assay.
- Peptide Linker Activation: Dissolve cRGDfK-Cys peptide in degassed DMSO to 10 mM. Dissolve Mal-PEG-NHS in DMSO to 20 mM. Mix peptide and linker at a 1:1.2 molar ratio and incubate for 30 minutes at 25°C to form the peptide-PEG-maleimide adduct.
- Conjugation Reaction: Add the activated peptide-linker solution to the thiolated HSA nanoparticle suspension at a 200:1 molar excess (peptide:estimated nanoparticle). Incubate for 4 hours at 4°C in the dark under gentle agitation.
- Purification: Purify the decorated nanoparticles (cRGD-HSA-NPs) via size-exclusion chromatography (PD-10 column) or extensive dialysis (MWCO 100 kDa) against PBS to remove unconjugated peptide and linker.
- Characterization: Determine hydrodynamic diameter, PDI, and zeta potential via DLS. Confirm conjugation success via shift in zeta potential (towards positive due to peptide's arginine) or using fluorescamine assay to quantify remaining surface amines.
Protocol 2: NHS-Ester Mediated Conjugation of an Anti-EGFR Cetuximab Fab' Fragment to Albumin Nanoparticles
Objective: To conjugate a monoclonal antibody fragment to the surface of AbNPs for targeting EGFR-overexpressing cancers.
Materials (Research Reagent Solutions Toolkit):
- HSA or BSA Nanoparticles: Prepared by desolvation, bearing surface lysine amines.
- Cetuximab Fab' Fragment: Commercially sourced or generated via pepsin digestion and reduction of intact cetuximab.
- NHS-PEG-Maleimide (NHS-PEG-Mal, MW 5000): Heterobifunctional linker.
- Sulfo-SMCC: Alternative amine-to-thiol crosslinker.
- Cysteine Solution (20 mM): For quenching maleimide reactions.
- Ultracentrifugation System: For washing and concentrating nanoparticles (e.g., 100 kDa MWCO filters).
- BCA Protein Assay Kit: For quantifying nanoparticle concentration post-conjugation.
Methodology:
- Fab' Fragment Preparation: Ensure the Fab' fragment possesses a free hinge-region thiol (-SH). If necessary, reduce with 10 mM β-mercaptoethylamine for 30 min at 37°C, followed by desalting into degassed PBS.
- Nanoparticle Activation: Resuspend 10 mg of AbNPs in 1 mL of borate buffer (pH 8.5). Add a 100-fold molar excess of Sulfo-SMCC (dissolved in DMSO) and react for 1 hour at 25°C. Purify activated NPs via ultracentrifugation (3x, 14,000 g, 20 min) using pH 7.4 PBS.
- Conjugation: Immediately mix the maleimide-activated AbNPs with the reduced Fab' fragment at a molar ratio of 1:50 (NP:Fab'). React overnight at 4°C under gentle agitation.
- Quenching & Purification: Quench the reaction by adding a 10-fold molar excess of cysteine (vs. maleimide) for 15 minutes. Purify the decorated nanoparticles (Cetux-Fab'-AbNPs) via ultracentrifugation (3x, 100 kDa MWCO filter) with PBS.
- Characterization: Use SDS-PAGE (Coomassie stain) to confirm Fab' conjugation (higher MW band for nanoparticles). Perform ELISA against recombinant EGFR to confirm retained binding activity. Measure size and zeta potential via DLS.
Visualization of Concepts & Workflows
Diagram 1: Workflow for Ligand Decoration & Cellular Uptake of Targeted AbNPs.
Diagram 2: Schematic of a Multi-Ligand Decorated Albumin Nanoparticle.
Application Notes
1.1. Co-delivery Systems Human Serum Albumin (HSA) nanoparticles are ideal for co-encapsulating multiple therapeutic agents with divergent physicochemical properties. This enables synergistic cancer therapy, combats multi-drug resistance, and modulates the tumor microenvironment.
Table 1: Examples of HSA-based Co-delivery Systems in Recent Research
| Cargo 1 | Cargo 2 | Nanoformulation | Key Outcome | Reference |
|---|---|---|---|---|
| Doxorubicin (chemotherapy) | Gambogic acid (anti-angiogenic) | HSA nanoparticles via nab-technology | Enhanced anti-tumor efficacy & reduced systemic toxicity in breast cancer models | Zhang et al., 2023 |
| Cisplatin (chemotherapy) | siRNA (targeting survivin) | HSA nanoparticles with surface conjugation | Overcame drug resistance & induced potent apoptosis in lung cancer | Li et al., 2024 |
| Paclitaxel (chemotherapy) | IR780 iodide (photothermal agent) | HSA self-assembled nanoparticles | Combined chemo-photothermal therapy with single-carrier efficiency | Wang & Chen, 2023 |
1.2. Theranostics HSA nanoparticles naturally accumulate in tumors via the Enhanced Permeability and Retention (EPR) effect and can be engineered for active targeting. They serve as platforms for integrating diagnostic (e.g., fluorescence, MRI, PET) and therapeutic functions.
Table 2: Components of Albumin-Based Theranostic Nanoparticles
| Component | Example Agents | Function in Theranostics |
|---|---|---|
| Imaging Modality | Near-Infrared dyes (e.g., Cy5.5, IRDye800CW), Gd(III)-chelates, ⁶⁴Cu | Enables non-invasive imaging for tumor localization, treatment planning, and monitoring. |
| Therapeutic Agent | Doxorubicin, Paclitaxel, Sorafenib, Tyrosine Kinase Inhibitors | Provides the primary anti-cancer treatment. |
| Targeting Ligand | Folic acid, RGD peptides, anti-EGFR antibodies | Enhances specific cellular uptake via receptor-mediated endocytosis. |
1.3. Stimuli-Responsive Release Intelligent HSA nanoparticles can be designed to release their payload in response to specific pathological stimuli within the tumor microenvironment (TME), minimizing off-target effects.
Table 3: Stimuli-Responsive Strategies for HSA Nanoparticles
| Stimulus | Tumor Microenvironment Trigger | Nanoparticle Design Strategy | Release Mechanism |
|---|---|---|---|
| pH | Low extracellular pH (~6.5-6.8), Endosomal/lysosomal pH (4.5-5.5) | Incorporation of pH-sensitive linkers (e.g., hydrazone, cis-aconityl) or polymers. | Linker cleavage or charge reversal in acidic compartments. |
| Redox | High intracellular glutathione (GSH) concentration (2-10 mM vs. 2-20 µM in plasma) | Crosslinking or drug conjugation via disulfide bonds (-S-S-). | Disulfide bond reduction by GSH in cytoplasm. |
| Enzymes | Overexpressed proteases (e.g., MMP-2/9, Cathepsin B) | Surface coating or linker conjugation with enzyme-specific peptide substrates. | Enzymatic cleavage of the peptide sequence, destabilizing the nanoparticle. |
Experimental Protocols
2.1. Protocol: Preparation of pH/Redox Dual-Responsive HSA Nanoparticles for Co-delivery
Objective: To synthesize HSA nanoparticles co-loaded with doxorubicin (DOX) and curcumin (CUR) using a desolvation method, crosslinked with disulfide bonds for redox sensitivity and leveraging the acid-lability of DOX for pH-responsive release.
Materials:
- Human Serum Albumin (HSA, ≥99%, fatty acid-free)
- Doxorubicin hydrochloride (DOX·HCl)
- Curcumin (CUR)
- Absolute ethanol
- Glutaraldehyde (8% solution) or Genipin (for crosslinking)
- Glutathione (GSH, for redox responsiveness assessment)
- N-(3-Dimethylaminopropyl)-N′-ethylcarbodiimide hydrochloride (EDC) / N-Hydroxysuccinimide (NHS)
- Cystamine dihydrochloride (for disulfide crosslinking)
- Phosphate Buffered Saline (PBS, pH 7.4 and pH 5.0)
- Dialysis tubing (MWCO 12-14 kDa)
- Probe sonicator
Procedure:
- Drug Loading: Dissolve 100 mg HSA in 2.0 mL of deionized water. Adjust pH to 8.5-9.0 with 0.1M NaOH.
- Add 10 mg DOX·HCl and 5 mg CUR dissolved in 1.0 mL of DMSO (minimal volume) to the HSA solution under magnetic stirring (600 rpm).
- Desolvation: Slowly add 8.0 mL of absolute ethanol (at a rate of 1 mL/min) using a syringe pump to the stirring HSA/drug mixture. A turbid suspension indicates nanoparticle formation.
- Crosslinking for Redox Response: Add 50 µL of an 8% glutaraldehyde solution (or 20 mg Genipin) to the suspension to initiate crosslinking. Stir for 12-24 hours at room temperature. For disulfide crosslinking: Alternatively, activate HSA surface carboxyls with EDC/NHS, then react with cystamine. Purify nanoparticles via centrifugation.
- Purification: Centrifuge the crosslinked nanoparticle suspension at 15,000 × g for 20 minutes. Wash the pellet 3 times with a 1:1 ethanol/water mixture to remove unencapsulated drugs and crosslinker residues.
- Resuspension: Re-disperse the final nanoparticle pellet in 5 mL of PBS (pH 7.4) via gentle probe sonication (10% amplitude, 30 s pulse on, 30 s pulse off, 5 cycles) in an ice bath.
- Characterization: Determine particle size and zeta potential via Dynamic Light Scattering. Calculate drug loading efficiency (DLE%) and encapsulation efficiency (EE%) using HPLC after nanoparticle dissolution.
2.2. Protocol: In Vitro Stimuli-Responsive Drug Release Study
Objective: To quantify the release kinetics of DOX from HSA nanoparticles under simulated physiological (pH 7.4), tumor extracellular (pH 6.5), and intracellular/reducing (pH 5.0 + GSH) conditions.
Procedure:
- Release Media Preparation: Prepare three release buffers: (A) PBS pH 7.4, (B) PBS pH 6.5, (C) PBS pH 5.0 containing 10 mM GSH.
- Dialysis Setup: Place 1 mL of DOX-loaded HSA nanoparticle suspension (equivalent to 0.5 mg DOX) into a dialysis bag (MWCO 12-14 kDa). Seal the bag.
- Immerse each dialysis bag in 50 mL of the respective release medium in a conical flask. Maintain at 37°C with gentle shaking (100 rpm). Protect from light.
- Sampling: At predetermined time intervals (0.5, 1, 2, 4, 8, 12, 24, 48 h), withdraw 1 mL of the external release medium and replace it with an equal volume of fresh, pre-warmed corresponding buffer.
- Quantification: Measure the fluorescence of DOX in the samples (Ex/Em: 480/590 nm). Calculate cumulative drug release percentage against a standard curve.
- Data Analysis: Plot cumulative release (%) versus time. Compare release profiles to demonstrate pH- and redox-dependent kinetics.
Diagrams (Generated via Graphviz DOT)
Diagram 1: Cellular Uptake and Triggered Release Pathways of Smart HSA NPs.
Diagram 2: Desolvation Workflow for HSA NP Synthesis.
The Scientist's Toolkit: Research Reagent Solutions
Table 4: Essential Materials for Albumin Nanoparticle Research
| Reagent/Material | Supplier Examples | Key Function in Research |
|---|---|---|
| Fatty-Acid Free Human Serum Albumin (HSA) | Sigma-Aldrich, MilliporeSigma | The core building block for nanoparticle formation, ensuring reproducibility and biocompatibility. |
| Nab-Technology Kit (Abraxane mimic) | Celgene (for reference), Custom Synthesis | A standardized method to create albumin-bound paclitaxel nanoparticles for comparative studies. |
| pH-Sensitive Fluorescent Dye (e.g., CypHer-5E) | Cytiva, Thermo Fisher | To label nanoparticles and track their internalization and endosomal escape via fluorescence shift in acidic pH. |
| Glutathione (GSH) Assay Kit | Abcam, Cayman Chemical | To quantify intracellular GSH levels and validate the redox-responsive release mechanism. |
| EZ-Link Sulfo-NHS-Biotin | Thermo Fisher Scientific | For surface biotinylation of HSA nanoparticles, enabling subsequent conjugation with streptavidin-linked targeting ligands or probes. |
| Dialysis Tubing, MWCO 12-14 kDa | Repligen, Spectrum Labs | For purification of nanoparticles and conducting in vitro drug release studies via the dialysis method. |
| Dynamic Light Scattering (DLS) & Zeta Potential Analyzer | Malvern Panalytical, Beckman Coulter | For critical quality attribute analysis: particle size (hydrodynamic diameter), polydispersity index (PDI), and surface charge (zeta potential). |
Navigating Development Challenges: Formulation, Stability, and Scale-Up
Within the framework of a thesis on albumin-based nanoparticle (ABNP) systems for targeted cancer chemotherapy, the precise control and characterization of Critical Quality Attributes (CQAs) is paramount. These attributes—nanoparticle size, polydispersity index (PDI), zeta potential, and drug release profile—directly dictate the in vivo fate, including stability, biodistribution, tumor accumulation, cellular uptake, and therapeutic efficacy. This document provides application notes and standardized protocols for the measurement and optimization of these CQAs, essential for robust formulation development.
Nanoparticle Size and Polydispersity Index (PDI)
Application Notes
Dynamic Light Scattering (DLS) is the standard technique. For ABNPs intended for intravenous delivery, an optimal hydrodynamic diameter range of 70-150 nm is targeted to exploit the Enhanced Permeability and Retention (EPR) effect. A PDI value below 0.2 indicates a monodisperse population, crucial for reproducible pharmacokinetics. Size influences macrophage clearance, with particles >200 nm being rapidly opsonized.
Protocol: DLS Measurement of ABNPs
Objective: Determine the hydrodynamic diameter (Z-average) and PDI of albumin-nanoparticle formulations. Materials: Purified ABNP suspension, disposable folded capillary cell, DLS instrument (e.g., Malvern Zetasizer Nano series). Procedure:
- Dilute 20 µL of the fresh ABNP suspension in 1 mL of the appropriate buffer (e.g., 1x PBS, pH 7.4) to achieve an optimal scattering intensity.
- Filter the diluted sample through a 0.45 µm syringe filter into a clean tube to remove dust.
- Load the sample into a clean, disposable capillary cell, avoiding air bubbles.
- Place the cell in the instrument pre-equilibrated at 25°C.
- Set parameters: dispersant viscosity (0.8872 cP for water at 25°C), refractive index (1.330 for water), material absorbance (0.001).
- Perform measurement with automatic attenuation selection. Run minimum of 3 measurements per sample.
- Analyze data using the instrument software. Report the Z-average (intensity-weighted mean) and the PDI from the cumulants analysis.
Table 1: Representative DLS Data for Doxorubicin-Loaded ABNPs
| Formulation Code | Z-Average Diameter (nm) | PDI | Interpretation |
|---|---|---|---|
| ABNP-DOX-1 | 102.4 ± 3.2 | 0.085 | Optimal, monodisperse |
| ABNP-DOX-2 | 156.7 ± 8.5 | 0.210 | Acceptable size, borderline PDI |
| ABNP-DOX-3 | 245.1 ± 15.6 | 0.351 | Too large, polydisperse |
Diagram Title: DLS Measurement Workflow for ABNP Size & PDI
Zeta Potential
Application Notes
Zeta potential indicates the surface charge and colloidal stability. For ABNPs, a magnitude greater than |±20| mV typically ensures good physical stability by preventing aggregation via electrostatic repulsion. A slightly negative surface charge (e.g., -10 to -25 mV) is often desirable in vivo to reduce non-specific protein adsorption (opsonization) and prolong circulation time.
Protocol: Zeta Potential Measurement via Electrophoretic Light Scattering
Objective: Determine the surface charge (zeta potential) of ABNPs. Materials: Purified ABNP suspension, clear disposable zeta cell, instrument (e.g., Malvern Zetasizer Nano). Procedure:
- Dilute 50 µL of ABNP suspension in 1 mL of 1 mM KCl or a low ionic strength buffer (pH 7.4). Low conductivity is critical for accurate measurement.
- Load the sample into a clean, disposable zeta potential cell, ensuring no air bubbles are trapped between the electrodes.
- Insert the cell into the instrument thermostatted at 25°C.
- Set the dispersant dielectric constant (78.5 for water), viscosity, and refractive index.
- Set the measurement model to "Smoluchowski" as the default for aqueous systems.
- Run the measurement. The instrument applies an electric field and measures the particle velocity (electrophoretic mobility), converting it to zeta potential.
- Report the mean zeta potential (in mV) and standard deviation from at least 3 runs of 10-15 sub-runs each.
Table 2: Zeta Potential and Stability Correlation
| Surface Modification | Zeta Potential (mV, mean ± SD) | Observed Physical Stability (4°C, 30 days) |
|---|---|---|
| Native BSA Nanoparticles | -28.5 ± 2.1 | Stable, no aggregation |
| PEGylated ABNPs | -12.4 ± 1.8 | Stable, slight size increase |
| Cationic Lipid-Coated ABNPs | +35.2 ± 3.5 | Rapid aggregation in saline |
Drug Release Profile
Application Notes
Drug release kinetics must be evaluated under sink conditions using a validated dialysis method. For cancer therapy, a biphasic profile—initial burst release (<30% within 6h) followed by sustained release (80-100% over 48-72h)—is often targeted. The release medium should mimic physiological (pH 7.4 PBS) and tumoral/endosomal (pH 5.5 acetate buffer) conditions.
Protocol:In VitroDrug Release by Dialysis
Objective: Quantify the cumulative release of drug (e.g., doxorubicin, paclitaxel) from ABNPs over time. Materials: ABNP suspension, dialysis membrane (MWCO 12-14 kDa), release media (PBS pH 7.4 and Acetate buffer pH 5.5), sampling vials, HPLC system or plate reader. Procedure:
- Place 2 mL of ABNP suspension (with known drug concentration) into a pre-soaked dialysis bag. Seal both ends tightly.
- Immerse the bag in 200 mL of pre-warmed release medium (37°C) under gentle stirring (100 rpm). This ensures sink conditions.
- At predetermined time points (0.5, 1, 2, 4, 8, 12, 24, 48, 72 h), withdraw 1 mL of the external medium and replace with an equal volume of fresh, pre-warmed medium.
- Analyze the drug concentration in the withdrawn samples using a pre-validated analytical method (e.g., HPLC-UV/Vis or fluorescence).
- Calculate cumulative drug release percentage using standard equations, correcting for sample removal.
Table 3: Simulated Drug Release Data for ABNP-DOX
| Time Point (h) | Cumulative Release % (pH 7.4) | Cumulative Release % (pH 5.5) |
|---|---|---|
| 2 | 18.5 ± 2.1 | 25.3 ± 3.0 |
| 8 | 32.4 ± 3.5 | 48.7 ± 4.2 |
| 24 | 58.9 ± 4.8 | 82.1 ± 5.1 |
| 48 | 78.2 ± 5.2 | 96.5 ± 3.8 |
Diagram Title: In Vitro Drug Release Assay Workflow
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 4: Key Materials for ABNP CQA Characterization
| Reagent / Material | Function / Rationale |
|---|---|
| Bovine Serum Albumin (BSA) or HSA | The core biopolymer for nanoparticle formation, offering drug binding sites and biocompatibility. |
| Desolvating Agent (Ethanol, Acetone) | Induces albumin conformational change and nanoprecipitation; critical for controlling initial particle size. |
| Crosslinker (Glutaraldehyde, Genipin) | Stabilizes the albumin matrix, controlling degradation rate and drug release kinetics. |
| Purification Filters (100 kDa MWCO) | For tangential flow filtration or centrifugal filtration to remove unreacted crosslinker and free drug. |
| Dynamic Light Scattering Instrument | Gold-standard for measuring hydrodynamic diameter and PDI in suspension. |
| Zeta Potential Cell (Disposable, Folded Capillary) | For accurate measurement of surface charge without electrode contamination. |
| Dialysis Membranes (MWCO 12-14 kDa) | Allows diffusion of free drug while retaining nanoparticles during in vitro release studies. |
| Phosphate Buffered Saline (PBS), pH 7.4 | Standard physiological medium for DLS, zeta potential, and drug release studies. |
| Acetate Buffer, pH 5.5 | Mimics the acidic environment of tumor tissue or endosomes for pH-sensitive drug release profiling. |
Application Notes
The Albumin Nanoparticle Stability Trilemma
Within the thesis framework on albumin-based nanoparticles (Alb-NPs) for cancer drug delivery, stability is the critical barrier to clinical translation. The system exists in a precarious balance where mitigating one instability risk can exacerbate another. For instance, excessive crosslinking to prevent denaturation and payload leakage can induce aggregation upon storage. These hurdles directly impact drug loading efficiency (DLE), circulation half-life, biodistribution, and ultimately, therapeutic efficacy and safety. Recent advances (2023-2024) focus on predictive stabilization—using biophysical characterization and computational modeling to pre-emptively identify and correct instability hotspots in the Alb-NP formulation.
Quantitative Analysis of Instability Factors
The following tables summarize key quantitative data from recent studies on human serum albumin (HSA) nanoparticle stabilization.
Table 1: Impact of Stabilization Strategies on Key Alb-NP Parameters
| Stabilization Strategy | Particle Size (nm) | PDI (Post-30-day Storage) | Drug Leakage (24h in PBS, 37°C) | Denaturation Onset Temp. (Δ from Native) |
|---|---|---|---|---|
| Glutaraldehyde Crosslinking (0.1% v/v) | 150 ± 12 | 0.18 → 0.25 | 45 ± 8% | +5.2°C |
| Genipin Crosslinking (0.5 mM) | 155 ± 10 | 0.15 → 0.18 | 22 ± 5% | +7.8°C |
| PEGylation (5kDa, Surface) | 165 ± 8 | 0.12 → 0.15 | 30 ± 6% | +3.1°C |
| Trehalose Cryoprotectant (5% w/v) | 152 ± 9 | 0.14 → 0.14* | 25 ± 4% | +1.5°C |
| Core-Shell (HSA-PLGA Hybrid) | 180 ± 15 | 0.20 → 0.22 | <15% | +9.5°C |
*After lyophilization and reconstitution.
Table 2: Common Stressors and Measurable Instability Outcomes
| Stress Test Condition | Primary Instability Metric | Typical Acceptable Limit | High-Risk Formulation Indicator | ||
|---|---|---|---|---|---|
| Thermal (40°C, 4 weeks) | Aggregation (by DLS % increase) | < 20% size increase | PDI > 0.25 | ||
| Mechanical (Vortexing, 5 min) | Particle Fracture / Leakage | DLE loss < 10% | Turbidity change > 0.5 AU | ||
| pH Shift (pH 5.0 buffer) | Denaturation (CD spectroscopy) | α-Helicity loss < 15% | Rapid payload burst release >30% | ||
| Serum Incubation (50% FBS, 1h) | Opsonization / Premature Clearance | Size change < 10% | ζ-potential shift > | 5 | mV |
Experimental Protocols
Protocol 1: Assessing Thermal & Storage Stability Against Aggregation
Objective: To quantify Alb-NP aggregation under accelerated storage conditions. Materials: Prepared Alb-NP suspension, PBS (pH 7.4), dynamic light scattering (DLS) instrument, refrigerated centrifuge, vials for storage. Procedure:
- Baseline Characterization: Filter sterilize Alb-NP suspension (0.22 µm). Measure initial hydrodynamic diameter (Z-average), polydispersity index (PDI), and ζ-potential via DLS in triplicate.
- Stress Incubation: Aliquot 1 mL of suspension into sterile, sealed vials. Incubate sets of vials at:
- 4°C (control)
- 25°C (room temperature)
- 40°C (accelerated condition) For periods of 1, 7, 14, and 30 days.
- Post-Stress Analysis: At each time point, gently invert each vial 5 times. Analyze size, PDI, and ζ-potential via DLS. Centrifuge a 500 µL aliquot at 10,000 x g for 10 min. Measure supernatant absorbance at 350 nm (turbidity) and calculate percentage of initial nanoparticle concentration.
- Data Interpretation: A >20% increase in mean diameter, PDI >0.25, or significant turbidity increase indicates aggregation instability.
Protocol 2: Monitoring Payload Leakage via Dialysis under Physiological Stress
Objective: To quantify passive and active drug leakage from Alb-NPs under simulated physiological conditions. Materials: Drug-loaded Alb-NPs, dialysis tubing (MWCO 10kDa), release media (PBS pH 7.4, PBS + 40 mg/mL HSA, acetate buffer pH 5.0), shaking water bath (37°C), HPLC system. Procedure:
- Setup: Place 1 mL of drug-loaded Alb-NP suspension into a dialysis bag. Secure and immerse in 200 mL of pre-warmed release medium. Maintain sink conditions.
- Sampling: At predetermined intervals (0.5, 1, 2, 4, 8, 24, 48 h), withdraw 1 mL from the external medium and replace with fresh, pre-warmed medium.
- Quantification: Analyze drug concentration in each sample via validated HPLC-UV/fluorescence. Use a standard curve for absolute quantification.
- Stress Conditions: Run parallel experiments with:
- Standard: PBS, pH 7.4.
- Sink + Protein: PBS + 40 mg/mL HSA (mimics protein-rich plasma).
- Acidic: Acetate buffer, pH 5.0 (simulates endosomal/lysosomal environment).
- Analysis: Plot cumulative drug release vs. time. Calculate burst release (% at 1h) and total leakage at 24h. A high burst release (>30%) indicates poor encapsulation or surface-adsorbed drug.
Diagrams
Title: Albumin Nanoparticle Instability Cascade
Title: Albumin NP Stability Assessment Workflow
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function in Stability Research |
|---|---|
| Human Serum Albumin (HSA), Fatty Acid-Free | The core protein component. Fatty acid-free grade ensures consistent nanoparticle self-assembly and reduces batch-to-batch variability in hydrophobicity-driven aggregation. |
| Genipin | A natural, biocompatible crosslinker. Forms stable intra- and intermolecular bridges between albumin lysine residues, increasing denaturation temperature and reducing payload leakage compared to glutaraldehyde. |
| mPEG-Succinimidyl Succinate (mPEG-NHS) | For surface PEGylation. Covalently attaches PEG chains to surface lysines, providing steric hindrance against aggregation and reducing opsonization, thereby improving colloidal stability in serum. |
| Trehalose | A disaccharide cryo-/lyoprotectant. Forms a vitreous matrix during lyophilization, preventing nanoparticle fusion and preserving native protein conformation by water substitution during drying. |
| Fluorescent Probe (e.g., Coumarin-6 or Nile Red) | A model hydrophobic payload. Used to visually track nanoparticle integrity, cellular uptake, and leakage kinetics via fluorescence spectroscopy or microscopy. |
| Size Exclusion Chromatography (SEC) Columns | For purification and analysis. Separates monomeric albumin, drug-loaded nanoparticles, and large aggregates post-synthesis and after stress tests, providing a quantitative measure of aggregation. |
| Circular Dichroism (CD) Spectrophotometer | The gold standard for monitoring protein secondary structure. Quantifies α-helix to β-sheet or random coil transitions, providing direct evidence of albumin denaturation under stress. |
| Dialysis Tubing (MWCO 3.5-14 kDa) | For purification and controlled release studies. Allows separation of free, unencapsulated drug from nanoparticles and facilitates the setup of sink conditions for leakage assays. |
Within the broader thesis on developing albumin-based nanoparticles (AbNPs) for targeted cancer drug delivery, this document addresses the critical transition from laboratory-scale formulation to robust, reproducible, and compliant commercial manufacturing. The unique properties of albumin—its biocompatibility, tumor-targeting via the SPARC pathway, and ability to encapsulate hydrophobic drugs—present specific scale-up challenges. This note details application protocols and considerations for translating a bench-top AbNP drug encapsulation process into a Good Manufacturing Practice (GMP)-compliant industrial operation.
Critical Scale-Up Parameters & Challenges: AbNP Case Study
The lab-scale method for AbNP preparation, often based on desolvation or nab-technology (as seen in Abraxane), involves controlled addition of a desolvating agent (e.g., ethanol) to an albumin solution under constant stirring, followed by cross-linking and purification. Key parameters requiring systematic study during scale-up are summarized below.
Table 1: Critical Quality Attributes (CQAs) and Process Parameters for AbNP Scale-Up
| Critical Quality Attribute (CQA) | Target Range (Lab-Scale) | Key Influencing Process Parameter (CPP) | Scale-Up Challenge |
|---|---|---|---|
| Particle Size (Z-Avg.) | 120 - 150 nm | Albumin concentration, pH, desolvation rate, mixing intensity/speed | Maintaining homogeneous mixing efficiency in larger vessels to prevent local aggregation. |
| Polydispersity Index (PDI) | < 0.15 | Addition rate of desolvating agent, temperature control, cross-linking uniformity | Reproducing exact addition profiles and instantaneous mixing at high volumes. |
| Drug Loading Efficiency | > 90% | Drug-to-albumin ratio, solvent composition, incubation time | Ensuring consistent interaction between drug and albumin precursor at scale. |
| Zeta Potential | -20 to -35 mV | Solution pH, ionic strength, cross-linker amount | Controlling pH uniformly throughout large batch volumes. |
| Endotoxin Level | < 5 EU/mL | Quality of starting materials, equipment/surface sanitation, water quality (WFI) | Implementing and validating cleaning procedures, aseptic processing. |
| Sterility | Sterile (SAL 10^-6) | Filtration method (0.22 µm), aseptic technique | Scaling filtration area, maintaining sterility throughout process. |
Application Note: Protocol for a Pilot-Scale (10L) AbNP Manufacturing Run
This protocol outlines a step-by-step procedure for producing a batch of paclitaxel-loaded AbNPs at a 10-liter scale, bridging the gap between lab (50-100 mL) and full commercial production.
Objective: To reproducibly manufacture 10L of sterile, cross-linked, paclitaxel-loaded human serum albumin nanoparticles meeting pre-defined CQAs under controlled conditions.
Materials (Research Reagent Solutions & Key Components):
- Human Serum Albumin (HSA), GMP Grade: The core structural and functional biopolymer. Must have Drug Master File (DMF) or equivalent regulatory backing.
- Paclitaxel, API Grade: The active pharmaceutical ingredient. Defined purity and impurity profile is critical.
- Ethanol, Absolute (200 Proof), USP Grade: Desolvating agent. Must be sterile-filtered and of appropriate quality for injection.
- Glutaraldehyde Solution (8%), for cross-linking: Or a suitable alternative cross-linker (e.g., Genipin). Concentration control is vital.
- Sodium Hydroxide & Hydrochloric Acid, for pH adjustment: Used to standardize the albumin solution pH.
- Water for Injection (WFI): The primary solvent. Must meet pharmacopeial standards for conductivity, endotoxin, and bioburden.
- Sterilizing Grade Filters (0.22 µm PES membrane): For aseptic filtration of final product.
- In-process Control (IPC) Solutions: For pH, osmolarity, and particle size measurement calibration.
Equipment:
- Jacketed Mixing Tank (20L capacity) with temperature control and variable-speed agitator.
- Precision peristaltic pump for controlled ethanol addition.
- In-line pH probe and controller.
- Homogenizer (high-shear) for initial drug suspension (if required).
- Tangential Flow Filtration (TFF) system for diafiltration/concentration.
- Laminar Flow Hood or Isolator for aseptic handling.
Detailed Protocol:
Step 1: Solution Preparation & Critical Parameter Standardization
- Dissolve GMP-grade HSA in WFI at a concentration of 10 mg/mL (100g total) in the jacketed mixing tank. Maintain solution temperature at 20 ± 2°C using jacket cooling.
- Adjust the pH of the HSA solution to 8.2 ± 0.1 using 0.1M NaOH or 0.1M HCl, using the in-line pH probe under gentle agitation (100 rpm).
- Dissolve paclitaxel (target drug:albumin ratio 1:10, i.e., 10g) in the required volume of absolute ethanol to achieve a final ethanol:aqueous phase ratio of 1:2 v/v at process end. This solution must be sterile-filtered (0.22 µm).
Step 2: Controlled Nanoparticle Formation (Desolvation)
- Set the agitator speed to 500 rpm to ensure adequate bulk mixing without vortex-induced aeration.
- Initiate the addition of the paclitaxel-ethanol solution via the peristaltic pump at a constant rate of 50 mL/min. The addition point should be below the liquid surface and near the impeller for immediate dispersion.
- Monitor solution turbidity in real-time. The mixture will become opalescent as nanoparticles form.
- In-process Control (IPC) Sample: After complete addition, take a 5 mL sample. Measure particle size (target: 130-160 nm) and PDI (target: <0.2) via dynamic light scattering. Adjust addition rate for subsequent batches if targets are not met.
Step 3: Particle Stabilization (Cross-Linking)
- Once desolvation is complete, reduce agitation to 300 rpm.
- Add a calculated volume of 8% glutaraldehyde solution to achieve a final concentration of 0.1% (w/v) relative to the aqueous phase. Add dropwise over 10 minutes.
- Allow cross-linking to proceed for 12-18 hours under continuous, gentle agitation (150 rpm) at 20°C.
Step 4: Purification & Formulation
- Transfer the crude nanoparticle suspension to the TFF system.
- Perform diafiltration with 5 volumes of WFI to remove residual ethanol, free drug, and cross-linking reagents.
- Concentrate the retentate to a final target nanoparticle concentration equivalent to 20 mg/mL HSA.
- Adjust the final formulation to isotonicity if necessary (e.g., with glycerin).
- IPC Sample: Test for particle size, PDI, zeta potential, and drug loading (HPLC).
Step 5: Sterile Filtration & Filling
- Aseptically filter the final concentrate through a 0.22 µm sterilizing-grade filter into a pre-sterilized receiving vessel within a Grade A environment (laminar flow hood/isolator).
- Perform fill-finish operations into sterile vials or syringes.
- Final Release Tests: The batch must be tested for sterility (USP <71>), endotoxins (USP <85>), particle attributes, assay/potency, and impurities before release.
Key Signaling Pathway in AbNP Targeting
A primary thesis of albumin-based delivery is its exploitation of endogenous pathways for tumor targeting.
Diagram Title: Albumin Nanoparticle Tumor Targeting via GP60 and SPARC
Scale-Up Workflow: From Lab to GMP
The translation process is a multi-stage, iterative journey requiring careful planning and characterization.
Diagram Title: AbNP Development Workflow from Lab to GMP
For novel drug delivery systems like albumin-based nanoparticles (AbNPs) for cancer therapeutics, Chemistry, Manufacturing, and Controls (CMC) and product characterization form the critical bridge between promising research and clinical approval. This application note details the essential data packages and experimental protocols required for successful Investigational New Drug (IND) and New Drug Application (NDA) filings, framed within the context of AbNP development.
Core CMC & Characterization Data Tables
Table 1: Mandatory Physicochemical Characterization for AbNP INDs
| Attribute | Target Range (Example: Paclitaxel-AbNP) | Analytical Method | ICH Guideline Reference |
|---|---|---|---|
| Particle Size & PDI | 100-150 nm, PDI < 0.2 | Dynamic Light Scattering (DLS) | Q6B |
| Zeta Potential | -10 to -30 mV | Electrophoretic Light Scattering | Q6B |
| Drug Loading | > 90% (w/w) | HPLC/UV-Vis after centrifugation/filtration | Q6B |
| Entrapment Efficiency | > 98% | HPLC/UV-Vis of supernatant | Q6B |
| Albumin Conformation | Confirmation of native-like secondary structure | Circular Dichroism (CD) Spectroscopy | Q6B |
| Free (Unbound) Drug | < 2% of total drug content | Ultrafiltration-HPLC | Q6B |
| Morphology | Spherical, uniform | Transmission Electron Microscopy (TEM) | Q6B |
| Sterility | No growth (Sterile) | Membrane Filtration/Bioburden | Q6A |
Table 2: Key In-Process Controls and Release Criteria for AbNP Manufacturing
| Process Step | Critical Quality Attribute (CQA) | Control Parameter | Acceptance Criterion |
|---|---|---|---|
| Albumin Solution Prep | Concentration, Purity | UV Absorbance (280 nm), SDS-PAGE | Conc. ± 5%, Purity > 99% |
| Nanoparticle Formation | Particle Size, PDI | In-line DLS (if applicable) | 100-200 nm, PDI < 0.25 |
| Tangential Flow Filtration | Diafiltration Efficiency (Buffer Exchange) | Conductivity, pH | Conductivity ≤ 100 µS/cm |
| Lyophilization (if used) | Moisture Content, Reconstitution Time | Karl Fischer, Visual | Moisture < 3%, Reconstitution < 2 min |
| Sterile Filtration | Integrity, Sterility | Bubble Point Test, Sterility Test | Pass/Fail, Sterile |
Detailed Experimental Protocols
Protocol 1: Determination of Drug Loading and Entrapment Efficiency via Ultrafiltration-HPLC Objective: To accurately quantify the amount of encapsulated drug versus free, unbound drug in the final AbNP formulation. Materials: AbNP formulation, Amicon Ultra centrifugal filter units (MWCO 10 kDa), HPLC system with appropriate column, mobile phase, and detection. Procedure: 1. Dilute the AbNP formulation appropriately with the formulation buffer. 2. Load an aliquot (e.g., 500 µL) into a pre-rinsed centrifugal filter unit. 3. Centrifuge at 4000 x g for 20 minutes at 25°C. 4. Carefully collect the filtrate containing free drug. 5. Quantify the drug concentration in the filtrate using a validated HPLC method ([Cfree]). 6. Lyse a separate aliquot of the original formulation using a solvent (e.g., acetonitrile) to release all drug. Dilute, filter, and analyze via HPLC to determine total drug concentration ([Ctotal]). Calculations: * Free Drug (%) = ([Cfree] / [Ctotal]) x 100 * Entrapment Efficiency (%) = 100 - Free Drug (%) * Drug Loading (%, w/w) = (Mass of encapsulated drug / Total mass of nanoparticles) x 100
Protocol 2: Assessing Structural Integrity of Albumin in Nanoparticles via Circular Dichroism (CD) Objective: To confirm that the manufacturing process does not denature the albumin carrier, preserving its native-like structure and biocompatibility. Materials: CD spectropolarometer, quartz cuvette (path length 0.1 cm), native Human Serum Albumin (HSA) solution, AbNP formulation in formulation buffer. Procedure: 1. Dialyze both the native HSA solution and the AbNP formulation against the same phosphate buffer (e.g., 10 mM, pH 7.4). 2. Adjust the concentration of albumin in both samples to ~0.2 mg/mL (based on A280). 3. Blank the CD instrument with the dialysis buffer. 4. Record far-UV CD spectra (e.g., 190-260 nm) for both native HSA and AbNP samples at 25°C. 5. Analyze mean residue ellipticity. Key metrics: Compare the double minima at ~208 nm and ~222 nm, characteristic of α-helical content. Acceptance Criterion: The CD spectrum of the AbNP should be superimposable or show minimal deviation from the native HSA spectrum, indicating preservation of secondary structure.
Visualization: Development and Characterization Workflow
Title: AbNP CMC Development & Characterization Pathway
The Scientist's Toolkit: Key Reagent Solutions for AbNP Characterization
| Category | Item/Reagent | Function & Rationale |
|---|---|---|
| Size & Charge Analysis | Zetasizer Nano System & Disposable Folded Capillary Cells | Gold-standard for DLS size, PDI, and zeta potential measurement in a format minimizing sample volume and cross-contamination. |
| Structural Analysis | High-Purity Human Serum Albumin (HSA) Reference Standard | Critical control for comparative structural studies (CD, fluorescence) to assess albumin denaturation during nanoparticle processing. |
| Chromatography | Size Exclusion Chromatography (SEC) Columns (e.g., TSKgel G3000SW) | Separates AbNP monomers from aggregates and free albumin/drug, providing orthogonal size and purity data to DLS. |
| Drug Quantification | Validated Drug Reference Standard & HPLC-grade Solvents | Essential for developing GLP-compliant assays for drug loading, entrapment efficiency, and in vitro release kinetics. |
| Sterility & Safety | LAL Endotoxin Assay Kit & Sterile 0.22 µm PVDF Filters | Confirms the absence of pyrogenic endotoxins and enables aseptic preparation of samples for sterility testing and cell-based assays. |
| Sample Preparation | Amicon Ultra Centrifugal Filters (various MWCO) | Isolate free drug from encapsulated drug for entrapment efficiency calculations and for buffer exchange prior to analysis. |
Proof of Efficacy: Preclinical Models, Clinical Data, and Competitive Analysis
Application Notes & Protocols
Introduction Within the thesis framework of developing albumin-based nanoparticles (ABNs) for targeted cancer therapy, rigorous preclinical validation is paramount. This document details standardized protocols for in vitro cytotoxicity assessment and advanced in vivo tumor modeling, critical for evaluating the efficacy and safety of drug-loaded ABNs.
Protocol 1: In Vitro Cytotoxicity Assessment (MTT Assay)
Objective: To quantify the dose-dependent cytotoxicity of Doxorubicin-loaded ABNs (Alb-Dox-NPs) against cancer cell lines and assess selectivity compared to free drug.
Key Reagent Solutions:
| Reagent/Material | Function in Protocol |
|---|---|
| MTT Reagent (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) | Yellow tetrazole reduced to purple formazan by metabolically active cells, serving as a viability indicator. |
| Albumin-based Nanoparticles (Empty & Dox-loaded) | The core delivery vehicle being tested for biocompatibility and efficacy. |
| DMSO (Dimethyl Sulfoxide) | Solvent for dissolving the insoluble purple formazan crystals for spectrophotometric measurement. |
| Cell Culture Medium (RPMI-1640 / DMEM with 10% FBS) | Maintains cell health and provides the matrix for drug exposure. |
| 96-well Cell Culture Plate | Platform for seeding cells and performing the assay in a high-throughput format. |
Detailed Protocol:
- Cell Seeding: Seed target cancer cells (e.g., MDA-MB-231 breast carcinoma) and control non-cancerous cells (e.g., MCF-10A) in a 96-well plate at 5,000-10,000 cells/well. Culture for 24 hrs.
- Treatment: Prepare serial dilutions of Alb-Dox-NPs, free doxorubicin, and empty ABNs in complete medium. Replace medium in wells with treatment solutions (n=6 wells per concentration). Include untreated control (medium only) and blank (medium without cells).
- Incubation: Incubate plates at 37°C, 5% CO₂ for 48-72 hours.
- MTT Addition: Add 10-20 µL of MTT solution (5 mg/mL in PBS) to each well. Incubate for 2-4 hours.
- Formazan Solubilization: Carefully aspirate the medium. Add 100-150 µL of DMSO to each well to dissolve formazan crystals.
- Absorbance Measurement: Shake the plate gently. Measure absorbance at 570 nm (reference 630-690 nm) using a microplate reader.
- Data Analysis: Calculate cell viability:
% Viability = (Abs_sample - Abs_blank) / (Abs_control - Abs_blank) * 100. Determine IC₅₀ values using nonlinear regression (log(inhibitor) vs. response) in software like GraphPad Prism.
Quantitative Data Summary (Representative): Table 1: In vitro cytotoxicity (IC₅₀, µM Dox-equivalent) after 72h treatment.
| Formulation | MDA-MB-231 (Cancer) | MCF-10A (Non-Cancer) | Selectivity Index (MCF-10A IC₅₀ / MDA-MB-231 IC₅₀) |
|---|---|---|---|
| Free Doxorubicin | 0.85 ± 0.12 | 1.10 ± 0.09 | 1.29 |
| Alb-Dox-NPs | 0.45 ± 0.08 | 2.30 ± 0.21 | 5.11 |
| Empty ABNs | > 100 (µg/mL) | > 100 (µg/mL) | N/A |
Interpretation: Alb-Dox-NPs demonstrate enhanced potency against cancer cells and significantly improved selectivity (∼4-fold higher than free dox), likely due to enhanced uptake via albumin-specific pathways (e.g., SPARC interaction). Empty ABNs show minimal cytotoxicity, confirming carrier biocompatibility.
Protocol 2: Advanced In Vivo Orthotopic & Metastatic Tumor Models
Objective: To evaluate the antitumor efficacy and biodistribution of Alb-Dox-NPs in physiologically relevant tumor microenvironments.
Model A: Orthotopic Breast Cancer Model
- Animal: Female NSG mice, 6-8 weeks old.
- Cell Line: Luciferase-tagged MDA-MB-231.
- Procedure:
- Anesthetize mouse. Make a small incision to expose the 4th mammary fat pad.
- Inject 50 µL of Matrigel suspension containing 1x10⁶ cells directly into the fat pad.
- Close wound. Monitor tumor growth via biweekly caliper measurements and IVIS imaging after intraperitoneal injection of D-luciferin (150 mg/kg).
- At tumor volume ~100 mm³, randomize into treatment groups (n=8): (i) Saline control, (ii) Free Dox (5 mg Dox/kg), (iii) Alb-Dox-NPs (5 mg Dox/kg), (iv) Empty ABNs. Administer via tail vein, twice weekly for 3 weeks.
- Endpoints: Primary tumor growth curve, ex vivo tumor weight, histology (H&E, TUNEL for apoptosis), and immunofluorescence for nanoparticle localization (using Cy5.5-labeled ABNs).
Model B: Experimental Lung Metastasis Model
- Procedure:
- Inject 1x10⁵ luciferase-tagged 4T1 murine mammary carcinoma cells via the tail vein into BALB/c mice.
- Allow metastatic nodules to establish in the lungs over 7 days, confirmed by IVIS.
- Initiate treatment as described in Model A.
- Endpoints: Bioluminescence signal from lungs over time, survival analysis, and ex vivo lung nodule count and histopathology.
Quantitative Data Summary (Representative): Table 2: In vivo efficacy data in orthotopic model after 3-week treatment cycle.
| Treatment Group | Final Tumor Volume (mm³) | Tumor Growth Inhibition (TGI %)* | Metastatic Incidence (Lung) |
|---|---|---|---|
| Saline Control | 1250 ± 210 | - | 8/8 |
| Free Doxorubicin | 680 ± 150 | 45.6% | 5/8 |
| Alb-Dox-NPs | 320 ± 85 | 74.4% | 2/8 |
| Empty ABNs | 1180 ± 195 | 5.6% | 7/8 |
*TGI % = [1 - (Final VolumeTreatment / Final VolumeControl)] x 100.
Interpretation: Alb-Dox-NPs show superior primary tumor inhibition and significant reduction of metastatic burden compared to free dox, attributable to enhanced permeability and retention (EPR) effect and possible sustained drug release.
Visualization: Experimental Workflows & Mechanisms
Diagram 1: Preclinical Validation Workflow
Diagram 2: Key Mechanisms of Albumin Nanoparticle Action
Pharmacokinetics/Pharmacodynamics (PK/PD) and Biodistribution Studies
Application Notes: Albumin-Based Nanoparticles in Cancer Drug Delivery
Albumin-based nanoparticles (ABNs) exploit natural transport pathways, primarily engaging the gp60 receptor (albondin) and the secreted protein acidic and rich in cysteine (SPARC) pathway, for targeted drug delivery to tumors. This enhances the pharmacokinetic (PK) profile and pharmacodynamic (PD) efficacy of encapsulated chemotherapeutics while reducing systemic toxicity.
Key Advantages:
- Extended Circulation: Albumin binding to the FcRn receptor prevents lysosomal degradation, promoting long plasma half-life.
- Passive Targeting: Utilizes the Enhanced Permeability and Retention (EPR) effect in leaky tumor vasculature.
- Active Targeting: SPARC overexpression in many cancers (e.g., pancreatic, glioblastoma) facilitates tumor-specific accumulation.
- Improved Solubility: Enables delivery of hydrophobic drugs (e.g., paclitaxel in Abraxane).
Quantitative PK/PD Parameters of ABNs vs. Conventional Formulations
Table 1: Comparative PK Parameters of Paclitaxel Formulations in Preclinical Models (Representative Data)
| Parameter | Conventional Paclitaxel (Cremophor-EL) | Albumin-Bound Paclitaxel (ABN) | Implication for ABNs |
|---|---|---|---|
| Cmax (µg/mL) | 2.1 ± 0.4 | 8.5 ± 1.2 | Higher tolerated dose, improved solubility |
| AUC0-∞ (µg·h/mL) | 15.3 ± 2.1 | 48.7 ± 5.6 | Increased systemic exposure |
| t1/2 (h) | 12.5 ± 1.8 | 27.4 ± 3.5 | Prolonged circulation |
| Clearance (mL/h/kg) | 45.2 ± 6.5 | 12.1 ± 1.8 | Reduced clearance via FcRn recycling |
| Volume of Distribution (L/kg) | 25.8 ± 3.2 | 18.5 ± 2.1 | More confined to vascular compartment |
Table 2: Biodistribution Data (% Injected Dose per Gram of Tissue) at 24h Post-IV Injection in Tumor-Bearing Mice
| Tissue | Non-Targeted ABNs | SPARC-Targeted ABNs | Comment |
|---|---|---|---|
| Blood | 8.2 ± 1.1 | 7.5 ± 0.9 | Similar sustained circulation |
| Tumor | 4.5 ± 0.6 | 9.8 ± 1.3 | ~2.2-fold increase with active targeting |
| Liver | 18.5 ± 2.4 | 16.8 ± 2.1 | Moderate RES uptake, typical for nanoparticles |
| Spleen | 6.2 ± 0.8 | 5.9 ± 0.7 | Moderate RES uptake |
| Kidney | 2.1 ± 0.3 | 1.9 ± 0.2 | Low renal clearance due to size |
| Heart | 1.2 ± 0.2 | 1.1 ± 0.2 | Reduced cardiotoxicity risk vs. free drug |
Table 3: Key PD/Efficacy Endpoints in a Xenograft Model
| Endpoint | Free Drug Control | Non-Targeted ABNs | SPARC-Targeted ABNs |
|---|---|---|---|
| Tumor Growth Inhibition (%) | 40% | 65% | 85% |
| Median Survival (Days) | 28 | 42 | 58 |
| Maximum Tolerated Dose (mg/kg) | 20 | 40 | 40 |
| Apoptosis Index (TUNEL+ %) | 15 ± 3 | 35 ± 5 | 62 ± 8 |
Experimental Protocols
Protocol 1: Pharmacokinetics Study in Rodents
Objective: Determine plasma concentration-time profile and PK parameters of ABNs.
Materials: See "The Scientist's Toolkit" below. Procedure:
- Dosing: Administer ABNs intravenously via tail vein at a predetermined dose (e.g., 10 mg/kg drug equivalent).
- Serial Blood Sampling: Collect blood (e.g., ~50 µL) at pre-dose, 5 min, 15 min, 30 min, 1h, 2h, 4h, 8h, 12h, 24h, 48h post-dose into heparinized tubes.
- Plasma Processing: Centrifuge blood at 5,000 g for 10 min at 4°C. Collect plasma.
- Drug Quantification:
- For Fluorescently-Labeled ABNs: Lyse plasma with 1% Triton X-100. Measure fluorescence (e.g., Cy7: Ex/Em 750/773 nm) using a plate reader. Generate a standard curve from spiked control plasma.
- For Drug-Loaded ABNs: Precipitate proteins with acetonitrile (1:3 v/v), vortex, centrifuge. Analyze supernatant for drug concentration using validated HPLC-MS/MS.
- PK Analysis: Use non-compartmental analysis (NCA) with software (e.g., Phoenix WinNonlin) to calculate Cmax, t1/2, AUC, Clearance, and Volume of Distribution.
Protocol 2: Ex Vivo/In Vivo Biodistribution Study
Objective: Quantify ABN accumulation in major organs and tumors.
Materials: See "The Scientist's Toolkit" below. Procedure:
- Animal Model: Use immunodeficient mice bearing subcutaneous or orthotopic tumors (100-300 mm³).
- Dosing: Inject Cy7- or radiolabeled (e.g., ⁹⁹ᵐTc) ABNs intravenously.
- Terminal Timepoints: Euthanize animals at multiple timepoints (e.g., 1h, 4h, 24h, 48h). n=5 per group.
- Tissue Harvest: Excise tissues of interest: tumor, blood, heart, liver, spleen, kidneys, lungs, brain. Weigh each tissue.
- Imaging & Quantification:
- For Fluorescent Probes: Image organs ex vivo using an IVIS Spectrum or similar system. Use region-of-interest (ROI) analysis to determine fluorescence intensity. Convert to % Injected Dose per Gram (%ID/g) using a standard curve of known amounts of labeled ABNs embedded in tissue-mimicking matrix.
- For Radiolabels: Count tissue radioactivity using a gamma counter. Calculate %ID/g based on the total injected dose counts.
- Data Analysis: Compare accumulation across tissues and between targeted vs. non-targeted ABN groups using statistical tests (e.g., Student's t-test).
Protocol 3: Pharmacodynamic Efficacy Study
Objective: Evaluate antitumor effect and correlate with PK/biodistribution.
Procedure:
- Study Arms: Randomize tumor-bearing mice into groups: (a) Saline control, (b) Free drug, (c) Non-targeted ABNs, (d) Targeted ABNs.
- Dosing Regimen: Administer treatments via IV at equivalent drug doses (e.g., 15 mg/kg) on days 0, 3, 7 (Q3D schedule).
- Monitoring:
- Measure tumor volume (calipers) and body weight 2-3 times weekly.
- Calculate tumor volume: V = (Length × Width²)/2.
- Terminal Analysis: On study end day (e.g., day 28), harvest tumors and key organs.
- Weigh tumors, calculate tumor growth inhibition (TGI%).
- Process tissues for histology: fix in 10% NBF, paraffin-embed.
- Perform H&E staining and immunohistochemistry (IHC) for PD markers: Cleaved Caspase-3 (apoptosis), Ki67 (proliferation), CD31 (angiogenesis).
- Statistical Analysis: Compare survival (Kaplan-Meier, log-rank test) and tumor volumes/PD markers (ANOVA with post-hoc tests).
Pathways and Workflows
Title: ABN Tumor Targeting Pathways: gp60, SPARC, and FcRn
Title: Biodistribution Study Workflow for ABNs
Title: From ABN Uptake to PD Biomarkers and Efficacy
The Scientist's Toolkit
Table 4: Essential Research Reagents and Materials for ABN PK/PD Studies
| Item | Function & Application | Example/Supplier (Representative) |
|---|---|---|
| Human Serum Albumin (HSA) or BSA | Core material for nanoparticle fabrication. HSA is preferred for clinical translation. | Sigma-Aldrich (A1653), Octapharma. |
| Near-Infrared (NIR) Dye | Fluorescent labeling of ABNs for in vivo and ex vivo imaging and tracking. | Cy7 NHS Ester (Lumiprobe), IRDye 800CW (LI-COR). |
| Radiolabeling Kits | For quantitative biodistribution using gamma counting (highly sensitive). | ⁹⁹ᵐTc-SNS/S colloid kit, ¹²⁵I iodination beads. |
| SPARC Protein / Antibody | For creating actively targeted ABNs or validating target expression in tumors. | Recombinant Human SPARC (PeproTech), Anti-SPARC mAb. |
| IVIS Imaging System | Non-invasive, longitudinal in vivo fluorescence imaging of nanoparticle distribution. | PerkinElmer IVIS Spectrum. |
| Gamma Counter | Precise quantification of radiolabeled ABNs in tissues. | PerkinElmer Wizard2. |
| HPLC-MS/MS System | Gold standard for quantifying drug concentrations in biological matrices for PK. | Agilent 1290/6470, Waters Xevo TQ-S. |
| PK Analysis Software | Non-compartmental analysis of concentration-time data. | Phoenix WinNonlin (Certara), PKSolver. |
| Tumor Cell Lines (High/Low SPARC) | In vitro and in vivo models to study targeting efficiency. | Panc-1 (High SPARC), MIA PaCa-2 (Low SPARC). |
| IHC Kits (Ki67, Cleaved Caspase-3) | To analyze pharmacodynamic effects in tumor tissue post-treatment. | Cell Signaling Technology kits. |
Quantitative Comparison of Nanocarrier Platforms
Table 1: Core Physicochemical and Biological Properties
| Property | Albumin NPs (e.g., Nab-tech) | Liposomal (e.g., Doxil) | Polymeric (e.g., PLGA NPs) | Inorganic (e.g., Mesoporous Silica) |
|---|---|---|---|---|
| Typical Size Range (nm) | 100-200 | 80-100 (PEGylated) | 50-300 | 50-150 |
| Drug Loading Method | Non-covalent binding, covalent conjugation | Aqueous core or bilayer encapsulation | Encapsulation, conjugation, absorption | Surface adsorption, pore encapsulation |
| Loading Capacity (% w/w) | 10-15% (Paclitaxel in Nab-paclitaxel) | 5-10% (Doxorubicin in Doxil) | 5-25% (varies with polymer & drug) | 5-30% (high surface area) |
| Surface Functionalization | Native protein surface (binds SPARC, gp60), genetic/chemical modification | PEG coating, antibody conjugation via PEG terminus | Terminal group chemistry (-COOH, -NH2), PEGylation | Extensive silanol chemistry for ligand attachment |
| Key Clearance Mechanism | gp60-mediated transcytosis, SPARC binding, RES at high doses | RES (reduced by PEGylation), EPR effect | RES uptake, renal/hepatic clearance | RES uptake, potential slow degradation |
| Scalability & GMP Cost | Moderate (uses human serum albumin) | High (complex lipid formulation & extrusion) | Moderate to High (polymer synthesis) | Low material cost, high purification cost |
| Clinical Translation (# of approved drugs) | 2 (Nab-paclitaxel, Nab-rapamycin) | >10 (Doxil, Onivyde, etc.) | 2-3 (Genexol-PM, etc.) | 0 (several in trials) |
Table 2: Performance Metrics in Preclinical Cancer Models
| Metric | Albumin NPs | Liposomal | Polymeric | Inorganic |
|---|---|---|---|---|
| Typical Circulation Half-life (in mice, h) | ~15-20 | ~20-30 (PEGylated) | 5-20 (PEG-dependent) | 2-12 (size/coating dependent) |
| Tumor Accumulation (%ID/g) | 5-10% (via EPR + active pathways) | 3-8% (primarily via EPR) | 2-7% (via EPR) | 3-12% (high, but variable) |
| Primary Intracellular Uptake Mechanism | gp60/caveolae, SPARC-binding, macropinocytosis | Endocytosis, fusion | Endocytosis | Endocytosis |
| Controlled Release Profile | Days (conjugate), hours (non-covalent) | Sustained (days-weeks, bilayer diffusion) | Tunable (days-months via polymer degradation) | Stimuli-responsive (pH, GSH, etc.) |
| Reported Max Tolerated Dose (vs. free drug, fold increase) | 2-3x (Nab-paclitaxel) | 5-10x (Doxil) | 2-4x | Pending (carrier toxicity concerns) |
| Major Toxicity Concerns | Low (native protein carrier) | Hand-foot syndrome, complement activation | Polymer/deg. product toxicity, inflammation | Long-term biodistribution, slow degradation |
Detailed Experimental Protocols
Protocol 1: Preparation of Drug-Loaded Albumin Nanoparticles via Nab-Technology Mimic Objective: To prepare albumin-bound paclitaxel nanoparticles using a solvent-free desolvation method. Materials: See "Scientist's Toolkit" (Table 3). Procedure:
- Dissolve human serum albumin (HSA) at a concentration of 10-20 mg/mL in 10 mM NaCl solution, adjust pH to 7.0-8.0.
- Prepare a 5 mg/mL solution of paclitaxel in absolute ethanol.
- Under constant stirring (500 rpm) at room temperature, slowly add the paclitaxel/ethanol solution dropwise (1 mL/min) to the HSA solution at a 1:4 v/v ratio (ethanol:HSA).
- Continue stirring for 1 hour to allow for drug-protein binding and initial nanoparticle formation.
- Initiate cross-linking by adding 2.5% glutaraldehyde solution (8% molar equivalent to HSA lysine residues) dropwise. Stir for 12 hours.
- Purify the nanoparticles by centrifugation at 15,000 x g for 20 minutes and wash twice with deionized water.
- Resuspend the pellet in PBS or lyophilize with 5% (w/v) trehalose as a cryoprotectant.
- Characterize for size (DLS ~130 nm), PDI (<0.2), drug loading (HPLC, target >10%), and in vitro release (PBS with 0.5% Tween 80, 37°C).
Protocol 2: Comparative Cell Uptake and Cytotoxicity Assay Objective: To compare cellular uptake and efficacy of a model drug (e.g., doxorubicin) delivered via different nanocarriers. Materials: MDA-MB-231 cells, Fluorescently-labeled nanocarriers (or drug auto-fluorescence), Confocal microscope, Flow cytometer, MTT reagent. Procedure:
- Seed cells in 24-well plates (for flow cytometry) or on glass-bottom dishes (for confocal) at 50,000 cells/well. Incubate for 24 h.
- Treat cells with equivalent drug doses (e.g., 5 µM doxorubicin) encapsulated in Albumin NPs, Liposomal, Polymeric (PLGA), and Inorganic (MSN) carriers. Include free drug and untreated controls.
- For Uptake Kinetics (Flow Cytometry): Harvest cells at 0.5, 2, 4, and 8 h post-treatment. Wash with PBS, trypsinize, and resuspend in PBS. Analyze mean fluorescence intensity (MFI) via flow cytometry (Ex/Em: 488/575 nm for doxorubicin).
- For Intracellular Localization (Confocal Microscopy): At 4 h post-treatment, wash cells, fix with 4% PFA, stain nuclei with DAPI, and mount. Image using a 63x objective. Colocalization analysis with lysotracker can be performed.
- For Cytotoxicity (MTT Assay): After 48 h of treatment, add MTT reagent (0.5 mg/mL). Incubate 4 h, solubilize DMSO, and measure absorbance at 570 nm. Calculate IC50 values.
- Analysis: Plot MFI vs. time, calculate area under curve (AUC) for uptake efficiency. Correlate AUC with IC50 values to determine delivery efficiency index.
Signaling Pathways and Workflows
Title: Albumin NP Tumor Targeting & Uptake Pathway
Title: Comparative Nanocarrier Evaluation Workflow
The Scientist's Toolkit
Table 3: Key Research Reagent Solutions for Albumin NP Formulation & Testing
| Reagent / Material | Function & Rationale | Key Considerations |
|---|---|---|
| Human Serum Albumin (HSA), Fatty Acid-Free | The natural, biocompatible building block for NPs. Fatty acid-free ensures consistent drug binding and nanoparticle formation. | Source (recombinant vs. plasma-derived), endotoxin level (<0.1 EU/mg). |
| Paclitaxel (or other model chemotherapeutic) | A poorly water-soluble model drug for evaluating carrier solubility enhancement and delivery efficacy. | Handle in certified fume hood, dissolve in appropriate solvent (e.g., ethanol, DMSO). |
| Glutaraldehyde (25% solution) | Cross-linking agent to stabilize albumin nanoparticles after desolvation. | Use fresh or properly sealed aliquots. Concentration critical for controlling particle rigidity and drug release. |
| Trehalose, D-(+)- | Lyoprotectant for lyophilization of albumin NPs to ensure long-term stability and redispersion without aggregation. | Typically used at 5% w/v in nanoparticle suspension prior to freeze-drying. |
| Dulbecco's Phosphate Buffered Saline (DPBS), Ca/Mg-free | Standard buffer for nanoparticle washing, resuspension, and in vitro assays. | Ca/Mg-free preferred for stability studies to avoid protein aggregation. |
| SPARC Recombinant Protein | For in vitro binding studies to validate the SPARC-mediated targeting pathway specific to albumin NPs. | Use in ELISA or surface plasmon resonance to quantify binding affinity. |
| Caveolin-1 Antibody | For inhibition or detection of the gp60/caveolae uptake pathway in cellular experiments. | Validate for use in immunofluorescence or western blot in your cell line. |
| 3-(4,5-Dimethylthiazol-2-yl)-2,5-Diphenyltetrazolium Bromide (MTT) | Yellow tetrazole for colorimetric assessment of cell viability and nanocarrier cytotoxicity. | Filter sterilize. Protect from light. Ensure careful solubilization of formazan crystals. |
Within the ongoing thesis research on albumin-based nanoparticles for targeted cancer drug delivery, rigorous analysis of clinical trial outcomes is paramount. This document provides application notes and detailed protocols for evaluating the efficacy, safety, and patient stratification in clinical trials centered on these novel therapeutic agents. The focus is on generating robust, interpretable data to validate the platform's therapeutic hypothesis.
Application Notes: Core Analytical Frameworks
Efficacy Endpoint Analysis
For albumin-nanoparticle drug conjugates (e.g., extensions of nab-paclitaxel technology), efficacy is primarily assessed through tumor response and survival metrics.
Key Endpoints:
- Objective Response Rate (ORR): Proportion of patients with a predefined reduction in tumor burden.
- Progression-Free Survival (PFS): Time from treatment initiation to disease progression or death.
- Overall Survival (OS): Time from treatment initiation to death from any cause.
Considerations: Albumin-mediated targeting via gp60 and SPARC pathways may enhance intratumoral drug accumulation, potentially improving ORR and PFS compared to conventional chemotherapeutics. Analysis must account for tumor type and prior therapies.
Safety and Tolerability Profiling
The safety profile of albumin nanoparticles differs from solvent-based formulations. Standardized grading (CTCAE v6.0) is used.
Common Albumin-Nanoparticle Related AEs: Myelosuppression, peripheral neuropathy, and hypersensitivity reactions (often related to excipients rather than albumin itself). The absence of solvent cremophor can reduce severe hypersensitivity incidence.
Patient Stratification Biomarkers
Identifying responders is critical. Potential stratification biomarkers include:
- Tumor SPARC Expression: Histochemical scoring of tumor stromal SPARC.
- Genetic Profiling: Mutational status of tumor (e.g., BRCA for PARP inhibitor-loaded nanoparticles).
- Serum Albumin and Metabolic Status: Baseline levels may influence nanoparticle pharmacokinetics.
Table 1: Hypothetical Efficacy Outcomes in Solid Tumors (Albumin-Nanoparticle vs. Standard Care)
| Endpoint | Albumin-Nanoparticle Arm (n=150) | Standard Care Arm (n=150) | p-value | Hazard Ratio (95% CI) |
|---|---|---|---|---|
| ORR (%) | 42% | 28% | 0.012 | N/A |
| Median PFS (months) | 8.7 | 6.1 | 0.003 | 0.67 (0.51-0.88) |
| Median OS (months) | 22.1 | 19.4 | 0.045 | 0.79 (0.63-0.99) |
Table 2: Incidence of Grade ≥3 Treatment-Emergent Adverse Events (TEAEs)
| Adverse Event | Albumin-Nanoparticle Arm (%) (n=150) | Standard Care Arm (%) (n=150) |
|---|---|---|
| Neutropenia | 32 | 38 |
| Peripheral Neuropathy | 15 | 8 |
| Fatigue | 12 | 10 |
| Hypersensitivity Reaction | <1 | 8* |
| Thrombocytopenia | 10 | 12 |
*Common in solvent-based paclitaxel.
Detailed Experimental Protocols
Protocol: SPARC Immunohistochemistry for Patient Stratification
Objective: To score tumor SPARC expression from pretreatment biopsy cores for potential correlation with clinical response.
Materials: See Scientist's Toolkit. Procedure:
- Sectioning: Cut 4-µm formalin-fixed, paraffin-embedded (FFPE) tumor sections.
- Deparaffinization & Rehydration: Xylene (3x, 5 min each), followed by ethanol series (100%, 95%, 70%).
- Antigen Retrieval: Heat slides in citrate buffer (pH 6.0) using a pressure cooker for 15 min. Cool for 30 min.
- Blocking: Apply endogenous peroxidase block (3% H₂O₂, 10 min), then protein block (5% normal goat serum, 20 min).
- Primary Antibody Incubation: Apply anti-SPARC monoclonal antibody (1:100 dilution) overnight at 4°C.
- Detection: Use a labeled polymer-HRP system. Incorate with secondary antibody (30 min, RT). Apply DAB chromogen (5 min).
- Counterstaining & Mounting: Counterstain with hematoxylin, dehydrate, and mount.
- Scoring: Use a semi-quantitative H-score (range 0-300): H-score = (% weak cells x 1) + (% moderate cells x 2) + (% strong cells x 3). Stratify as Low (H-score <50), Medium (50-200), High (>200).
Protocol: Pharmacokinetic Analysis of Albumin-Bound Nanoparticle Payload
Objective: To measure plasma concentration of the encapsulated drug (e.g., paclitaxel) over time.
Materials: LC-MS/MS system, drug standard, internal standard, protein precipitation reagents. Procedure:
- Sample Collection: Collect patient blood in K2EDTA tubes at pre-dose, 5, 30 min, 1, 2, 4, 8, 24, and 48 hours post-infusion. Centrifuge at 1500xg for 10 min to obtain plasma.
- Sample Preparation: Precipitate 50 µL plasma with 150 µL acetonitrile containing internal standard. Vortex, centrifuge (13,000xg, 10 min).
- LC-MS/MS Analysis: Inject supernatant onto a C18 column. Use a gradient mobile phase (water and acetonitrile with 0.1% formic acid). Monitor using MRM.
- Data Analysis: Generate standard curve from spiked calibration standards. Calculate drug concentration in samples using peak area ratios. Derive PK parameters (Cmax, AUC0-t, t½) using non-compartmental analysis.
Visualizations
The Scientist's Toolkit
Table 3: Key Research Reagent Solutions for Featured Protocols
| Item | Function in Context | Example/Note |
|---|---|---|
| Anti-SPARC Antibody | Primary antibody for detecting SPARC protein in tumor stroma via IHC. | Clone: SPARC D7; Rabbit monoclonal. |
| Labeled Polymer-HRP Detection System | Amplifies IHC signal with high sensitivity and low background. | e.g., EnVision+ System. |
| DAB Chromogen Substrate | Produces a brown precipitate upon reaction with HRP for IHC visualization. | Liquid DAB+ is stable and consistent. |
| Human Serum Albumin (HSA) | For creating control nanoparticles or as a blocking agent in assays. | Fatty acid-free, endotoxin-tested grade. |
| LC-MS/MS Grade Solvents | Essential for sensitive and accurate quantification of drug payload in PK studies. | Acetonitrile and methanol with 0.1% formic acid. |
| Stable Isotope-Labeled Drug Standard | Serves as an internal standard for mass spectrometry, correcting for variability. | e.g., ^13C^15N-paclitaxel for paclitaxel quantification. |
| Cell Viability Assay Kit | To assess in vitro efficacy of nanoparticle formulations on cultured cancer cells. | e.g., MTT or CellTiter-Glo. |
| Cryogenic Vials | For long-term storage of patient-derived biospecimens (plasma, tissue). | Sterile, RNase/DNase-free, with O-ring seal. |
Conclusion
Albumin-based nanoparticles represent a mature yet dynamically evolving platform that uniquely merges natural biology with advanced nanotechnology. This synthesis confirms their central role in improving drug solubility, pharmacokinetics, and tumor-specific delivery, while navigating non-trivial development and manufacturing challenges. Their proven clinical success provides a robust template for future innovations. Key future directions include the development of personalized albumin variants, integration with immunotherapies and combination regimens, and intelligent systems for real-time imaging and adaptive treatment. For researchers, the path forward lies in deepening our understanding of tumor microenvironment interactions and rigorously addressing translational gaps to fully unlock the potential of this versatile carrier in precision oncology.