CRISPR Nanodelivery: Next-Gen Vectors for Precision Gene Therapy and Editing
This article provides a comprehensive, current analysis of nanotechnology-based delivery systems for CRISPR-Cas gene-editing tools.
CRISPR Nanodelivery: Next-Gen Vectors for Precision Gene Therapy and Editing
Abstract
This article provides a comprehensive, current analysis of nanotechnology-based delivery systems for CRISPR-Cas gene-editing tools. Tailored for researchers and drug development professionals, we explore the foundational principles of viral and non-viral nanocarriers, detail the latest methodological advancements and therapeutic applications, address key challenges in targeting, efficiency, and safety, and offer a comparative validation of leading platforms. The synthesis guides the selection and optimization of delivery strategies for advancing CRISPR from bench to bedside.
The Engine of CRISPR Delivery: Decoding Viral and Non-Viral Nanocarriers
Within the broader thesis on CRISPR nanotechnology delivery systems, the central challenge is unequivocal: the therapeutic potential of CRISPR-Cas genome editing is bottlenecked by delivery. The CRISPR machinery—comprising Cas nuclease and guide RNA (gRNA)—is a large, negatively charged, and unstable macromolecular complex that must traverse multiple biological barriers to reach its intracellular target. Conventional delivery methods (e.g., viral vectors, electroporation) face limitations in immunogenicity, packaging size, tissue specificity, and manufacturability. Nanotechnology provides a rational engineering platform to overcome these barriers through tailored design of non-viral delivery vehicles, enabling targeted, efficient, and safe in vivo genome editing. This application note details the quantitative rationale and provides protocols for key nanomaterial-based CRISPR delivery systems under investigation.
Quantitative Data: Barriers and Nanocarrier Performance
Table 1: Key Physical and Biological Barriers to CRISPR-Cas9 Delivery
| Barrier | Quantitative Challenge | Consequence |
|---|---|---|
| Serum Stability | Naked siRNA t½ < 10 min; Cas9-gRNA complex degrades rapidly. | Rapid clearance, requires stabilization. |
| Cell Membrane | Negative charge repulsion; Cas9 RNP size > ~150 kDa. | Low cellular uptake (<1% without carrier). |
| Endosomal Escape | ~99% of internalized material remains trapped and degrades. | Major bottleneck for efficacy. |
| Nuclear Entry | Nuclear pore cutoff: ~40 kDa for passive diffusion. | Requires active import signals (NLS). |
| Off-Target Editing | Can occur at sites with 1-5 base mismatches to gRNA. | Safety risk requiring controlled delivery. |
Table 2: Performance Metrics of Leading Nanocarrier Platforms for CRISPR Delivery (Recent Data)
| Nanocarrier Type | Typical Loading Method | Average Size (nm) | Zeta Potential (mV) | Reported In Vivo Editing Efficiency | Key Advantage |
|---|---|---|---|---|---|
| Lipid Nanoparticles (LNPs) | Electrostatic/complexation | 70-120 | +10 to -10 | 40-60% in liver (mouse) | Clinical validation, high payload. |
| Polymeric Nanoparticles (e.g., PEI) | Complexation/encapsulation | 80-200 | +20 to +40 | 10-30% in various tissues | Tunable polymer chemistry. |
| Gold Nanoparticles (AuNPs) | Covalent conjugation/adsorption | 10-50 | Variable | 5-15% in vitro | Precise surface functionalization. |
| Mesoporous Silica Nanoparticles (MSNs) | Physical adsorption/packing | 50-150 | Variable | ~10% in vitro | High surface area, rigid structure. |
| DNA Nanoclews | Rolling circle amplification | 50-100 | Negative | 20-35% in vitro | Sequence-specific loading. |
Experimental Protocols
Protocol 3.1: Formulation of CRISPR-Cas9 RNP Loaded Lipid Nanoparticles (LNPs)
Objective: To prepare ionizable lipid-based LNPs encapsulating Cas9 ribonucleoprotein (RNP) for in vivo hepatic delivery. Materials: Cas9 protein, sgRNA, ionizable lipid (e.g., DLin-MC3-DMA), DSPC, cholesterol, PEG-lipid, ethanol, sodium acetate buffer (pH 4.0), PBS, dialysis tubing. Procedure:
- RNP Complexation: Incubate purified Cas9 protein with synthesized sgRNA at a 1:1.2 molar ratio in nuclease-free buffer for 10 min at 25°C to form the RNP complex.
- Lipid Solution Preparation: Dissolve ionizable lipid, DSPC, cholesterol, and PEG-lipid at a molar ratio (e.g., 50:10:38.5:1.5) in ethanol.
- Aqueous Phase Preparation: Dilute the RNP complex in 25 mM sodium acetate buffer (pH 4.0).
- Microfluidic Mixing: Using a staggered herringbone micromixer or equivalent, mix the ethanol lipid phase and the aqueous RNP phase at a 1:3 volumetric flow rate ratio (total flow rate 12 mL/min). The RNP is protonated at low pH, promoting encapsulation.
- Buffer Exchange & Dialysis: Immediately dilute the formed LNP suspension in 1x PBS (pH 7.4). Dialyze against 1x PBS for 2 hours using a 20kDa MWCO membrane to remove ethanol and adjust pH.
- Characterization: Measure particle size and PDI via DLS, zeta potential via electrophoretic light scattering, and encapsulation efficiency using a Ribogreen assay post-Triton X-100 disruption.
Protocol 3.2: Synthesis and Functionalization of CRISPR-Gold Nanoconjugates
Objective: To covalently conjugate Cas9 RNP to polyethyleneimine (PEI)-coated gold nanoparticles for localized delivery. Materials: 15nm citrate-capped AuNPs, branched PEI (25 kDa), Cas9 RNP, EDC/NHS coupling reagents, MES buffer. Procedure:
- AuNP Coating: Add 10 µg of PEI per 1 mL of AuNP solution (OD₅₂₀ ~1). Incubate with gentle shaking for 30 min. Purify via centrifugation (14,000 rpm, 20 min) and resuspend in 0.1 M MES buffer (pH 6.0).
- RNP Conjugation: Activate carboxyl groups on the Cas9 protein surface by incubating RNP with 5 mM EDC and 10 mM NHS in MES buffer for 15 min. Remove excess reagents using a desalting column.
- Conjugation Reaction: Mix the activated RNP with the PEI-coated AuNPs (aiming for a molar ratio of ~5 RNP per AuNP). React overnight at 4°C on a rotator.
- Quenching & Purification: Quench the reaction by adding 100 mM glycine for 30 min. Purify the conjugates by centrifugation (5,000 rpm, 10 min) to remove unbound RNP. Resuspend in sterile PBS.
- Characterization: Confirm conjugation via UV-Vis spectroscopy (shift in plasmon peak), agarose gel electrophoresis (reduced RNP mobility), and TEM imaging.
Diagrams
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for CRISPR Nanocarrier Research
| Item | Function/Application | Example Product/Catalog |
|---|---|---|
| Recombinant Cas9 Protein | Core nuclease component for RNP assembly. | Thermo Fisher Scientific, A36498. |
| Ionizable Cationic Lipid | Key LNP component for nucleic acid/complex encapsulation and endosomal escape. | MedChemExpress, HY-112366 (DLin-MC3-DMA). |
| Branched Polyethylenimine (PEI) | Cationic polymer for forming polyplexes or coating nanoparticles; promotes endosomal escape. | Sigma-Aldrich, 408727 (25 kDa). |
| Microfluidic Mixer Device | For reproducible, scalable nanoparticle formulation via rapid mixing. | Precision NanoSystems, Ignite mixer. |
| Dynamic Light Scattering (DLS) System | Measures nanoparticle hydrodynamic size, PDI, and zeta potential. | Malvern Panalytical, Zetasizer Ultra. |
| Ribogreen/Quant-iT Assay Kit | Quantifies encapsulation efficiency of nucleic acid payloads. | Invitrogen, R11490. |
| Endosomal Escape Assay Dye | Fluorescent probe (e.g., LysoTracker) to assess endosomal disruption. | Invitrogen, L7528. |
| In Vivo Edit-R Detection Kit | NGS-based or T7E1 assay for quantifying on- and off-target editing efficiency. | Integrated DNA Technologies. |
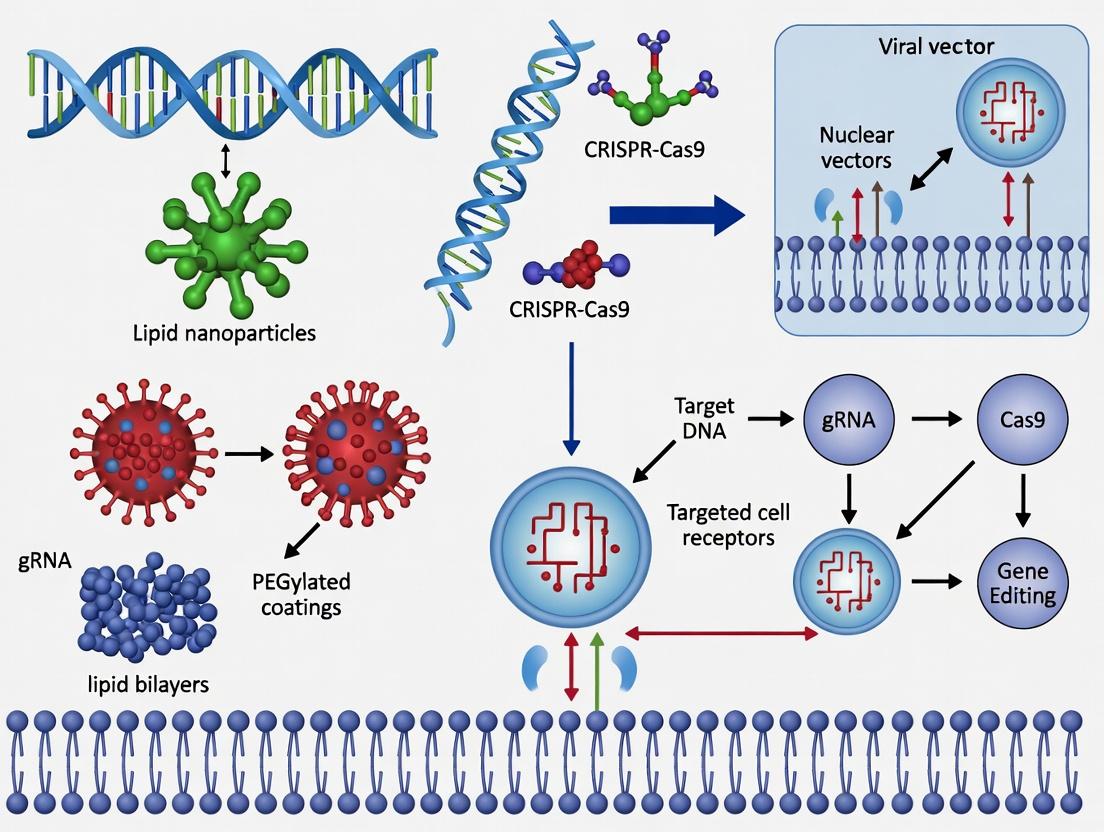
The integration of CRISPR-Cas systems into therapeutic and research applications is critically dependent on delivery vectors that ensure efficient, specific, and safe transfer of genetic cargo. While non-viral nanotechnology (e.g., lipid nanoparticles, polymeric NPs) is advancing, engineered viral vectors remain the gold standard for in vivo delivery due to their innate ability to traverse cellular membranes. This Application Notes document details re-engineered Adeno-Associated Virus (AAV), Lentivirus (LV), and Adenovirus (AdV) platforms, framed within the thesis that rational vector engineering is essential to overcome delivery bottlenecks—such as immunogenicity, cargo capacity, tropism, and manufacturing—in CRISPR nanotechnology research.
Table 1: Quantitative & Qualitative Comparison of Engineered Viral Vectors for CRISPR Delivery
| Parameter | Re-engineered AAV | Re-engineered Lentivirus | Re-engineered Adenovirus |
|---|---|---|---|
| Packaging Capacity | ~4.7 kb (Theoretical limit for efficient packaging) | ~8-10 kb | ~8-10 kb (ΔE1/E3 vectors); Up to ~36 kb (HDAd) |
| Integration Profile | Predominantly episomal; rare genotoxicity concerns. | Stable integration into host genome (for dividing cells). | Transient, episomal expression. |
| In Vivo Immunogenicity | Generally low; pre-existing humoral immunity common. | Moderate; potential for insertional mutagenesis concerns. | High; strong innate & adaptive immune responses. |
| CRISPR Payload Suitability | SaCas9, compact editors; dual-vector systems for SpCas9-gRNA. | Large Cas9 variants, base editors, prime editors, sgRNA. | Large Cas9, multiplexed gRNAs, or Cas9 with repair templates. |
| Tropism Modification | Extensive via capsid engineering (directed evolution, rational design). | Via pseudotyping (e.g., VSV-G, Rabies-G, LCMV-G for broad/cell-specific). | Via fiber knob or hexon modifications (peptide insertions, genetic ablations). |
| Production Titer | ~10¹³ – 10¹⁴ vg/mL (HEK293 suspension) | ~10⁸ – 10⁹ TU/mL (concentrated) | ~10¹¹ – 10¹² VP/mL (ΔE1/E3, HEK293) |
| Key Engineering Focus | Capsid diversification, immune evasion, enhanced CNS/ tissue tropism. | Safety (self-inactivating), targeting (pseudotypes), biosafety. | Detargeting & retargeting, "gutless" HDAd vectors, immune suppression. |
Application Notes & Protocols
Protocol: Production of Tropism-Modified AAV for CNS-Directed CRISPR Delivery
Aim: Generate AAV9-PHP.eB capsid pseudotyped vectors encoding SaCas9 and a gRNA expression cassette for in vivo murine brain targeting.
Research Reagent Solutions:
- pAAV-SaCas9-U6-sgRNA: AAV ITR-flanked plasmid expressing Staphylococcus aureus Cas9 and a U6-driven single guide RNA (sgRNA). Function: Provides the CRISPR genetic cargo.
- pAAV2/9-PHP.eB Rep-Cap Plasmid: Plasmid expressing AAV2 replication proteins and the engineered AAV9-PHP.eB capsid proteins. Function: Supplies replication machinery and determines tissue tropism.
- pAdDeltaF6 Helper Plasmid: Adenoviral helper plasmid providing essential genes (E2A, E4, VA RNA) for AAV replication. Function: Provides helper functions without adenovirus contamination.
- Polyethylenimine (PEI MAX), 40 kDa: Transfection reagent. Function: Facilitates plasmid co-transfection into HEK293T/HEK293 cells.
- Iodixanol Density Gradient Media (15%, 25%, 40%, 60%): Used for ultracentrifugation-based AAV purification. Function: Separates intact AAV particles from cellular debris and empty capsids based on buoyant density.
- Anti-AAV9 Monoclonal Antibody (Clone ADK9): Used for ELISA titering. Function: Specifically quantifies viral genome titer of AAV9-based capsids.
Methodology:
- Cell Seeding: Seed HEK293T cells at 70% confluency in 15-layer CellSTACKs in DMEM + 10% FBS.
- Transfection: At confluency, co-transfect using PEI MAX (1:3 DNA:PEI ratio) with a plasmid mix of pAAV-SaCas9-sgRNA, pAAV2/9-PHP.eB, and pAdDeltaF6 at a molar ratio of 1:1:1.
- Harvest: 72 hours post-transfection, harvest cells and medium. Lyse cells via freeze-thaw and Benzonase treatment (250 U/mL, 37°C, 1h).
- Purification: Clarify lysate by centrifugation. Perform iodixanol step gradient ultracentrifugation (250,000 x g, 2h). Extract the opaque 40-60% interface containing virus.
- Concentration & Buffer Exchange: Concentrate using 100 kDa MWCO centrifugal filters and exchange into PBS + 0.001% Pluronic F-68.
- Titration: Quantify viral genome titer (vg/mL) via qPCR using ITR-specific primers and a standard curve.
Protocol: Generation of Integrase-Defective Lentivirus (IDLV) for Transient CRISPR-Cas9 Delivery
Aim: Produce high-titer, integration-deficient lentivirus for transient expression of SpCas9 and gRNA to minimize off-target genomic integration risks.
Research Reagent Solutions:
- pLVX-U6-sgRNA-EF1α-SpCas9-P2A-eGFP: Third-generation lentiviral transfer plasmid. Function: Expresses sgRNA and SpCas9-EGFP fusion protein from separate promoters.
- pMD2.G (VSV-G Envelope): Plasmid expressing the vesicular stomatitis virus G glycoprotein. Function: Provides broad tropism pseudotype.
- psPAX2 (D64V Integrase Mutant): Packaging plasmid with a point mutation (D64V) in the integrase gene. Function: Supplies all lentiviral proteins except envelope, and the mutation abolishes integration, creating IDLV.
- Lenti-X Concentrator (Takara Bio): Solution containing a proprietary polymer. Function: Precipitates lentiviral particles from culture supernatant for easy concentration.
- Polybrene (Hexadimethrine bromide): Cationic polymer. Function: Enhances viral transduction efficiency by neutralizing charge repulsion between virus and cell membrane.
- p24 HIV ELISA Kit: Immunoassay kit. Function: Quantifies lentiviral particle concentration based on the p24 capsid antigen.
Methodology:
- Transfection: Seed HEK293T cells in T225 flasks. At 80% confluency, co-transfect with pLVX-SpCas9-sgRNA, psPAX2 (D64V), and pMD2.G using PEI MAX.
- Virus Collection: Replace medium 6-8h post-transfection. Collect virus-containing supernatant at 48h and 72h, filter through a 0.45 μm PES membrane.
- Concentration: Combine supernatants. Add 1/3 volume Lenti-X Concentrator, incubate at 4°C overnight. Centrifuge at 1,500 x g for 45 min. Resuspend pellet in 1/100th original volume in cold PBS.
- Titration: Determine functional titer (Transducing Units/mL, TU/mL) via flow cytometry (EGFP+ cells) on HEK293T cells 72h post-transduction, or quantify via p24 ELISA.
Visualized Workflows & Signaling
Diagram Title: AAV Production and Lentiviral Engineering Workflow
Diagram Title: Adenovirus Cell Entry and Engineering Targets
Within CRISPR-Cas9 nanotechnology delivery research, the primary bottleneck remains the safe, efficient, and tissue-specific transport of ribonucleoprotein (RNP) or mRNA/gRNA complexes. Viral vectors, while efficient, pose significant immunogenicity and insertional mutagenesis risks. This has catalysed the development of sophisticated non-viral nanocarriers. Lipid Nanoparticles (LNPs), polymer-based nanoparticles, and inorganic nanoparticles constitute the core of this non-viral arsenal, each with distinct advantages in encapsulation efficiency, tunability, and endosomal escape mechanisms. This document provides application notes and detailed protocols for their use in CRISPR delivery.
Application Notes & Quantitative Comparison
Table 1: Comparative Analysis of Non-Viral CRISPR Delivery Systems
| Property | Lipid Nanoparticles (LNPs) | Polymer-Based NPs (e.g., PEI) | Inorganic NPs (e.g., Gold NPs) |
|---|---|---|---|
| Typical Encapsulation Efficiency | >90% for mRNA | 70-85% for pDNA/RNP | 60-80% for RNP (surface conjugation) |
| Average Particle Size (nm) | 70-150 | 50-300 | 10-100 |
| Zeta Potential (mV) | Slightly negative to +20 | Highly positive (+20 to +50) | Variable (-30 to +30) |
| Key Mechanism of Endosomal Escape | pH-dependent ionizable lipid fusion | Proton sponge effect (buffering) | Photothermal/Photochemical disruption |
| Primary CRISPR Payload | mRNA/gRNA or sgRNA | pDNA, RNP, or sgRNA | RNP (covalent/non-covalent) |
| In Vivo Clearance | Hepatic (dominant), tunable | Renal & Hepatic | Renal & Reticuloendothelial System |
| Notable Toxicity Concern | Reactogenicity (acute) | Cytotoxicity at high N/P ratios | Long-term biodistribution & degradation |
Table 2: Recent In Vivo Efficacy Data (Representative Studies)
| Nanoparticle Type | Target Organ/Tissue | CRISPR Payload | Editing Efficiency (Reported) | Key Functional Group/Component |
|---|---|---|---|---|
| LNP (DLin-MC3-DMA) | Liver (Hepatocytes) | saCas9 mRNA + sgRNA | ~40% (serum Pcsk9 reduction) | Ionizable lipid (MC3) |
| Polymer (PBAE) | Lung Epithelium | Cas9 RNP | ~5% (airway epithelial cells) | Poly(beta-amino ester) |
| Gold Nanoparticle | Tumor (local) | Cas9 RNP | ~30% (EGFP knockout in tumor) | Thiolated PEG & Nuclear Targeting Peptide |
Experimental Protocols
Protocol 1: Formulation of CRISPR mRNA-LNPs via Microfluidics
Application: For hepatic delivery of Cas9 mRNA and sgRNA. Materials: See "The Scientist's Toolkit" (Table 3). Method:
- Prepare Aqueous Phase: Dissolve CRISPR-Cas9 mRNA and sgRNA in 10 mM citrate buffer (pH 4.0) at a total concentration of 0.1 mg/ml.
- Prepare Lipid Phase: Dissolve ionizable lipid (e.g., DLin-MC3-DMA), DSPC, Cholesterol, and DMG-PEG2000 at a molar ratio of 50:10:38.5:1.5 in absolute ethanol.
- Mixing: Using a microfluidic mixer (e.g., NanoAssemblr), set the total flow rate (TFR) to 12 ml/min and a flow rate ratio (aqueous:lipid) of 3:1. Combine streams to form nanoparticles.
- Buffer Exchange & Purification: Dialyze the formed LNP suspension against 1x PBS (pH 7.4) for 4 hours at 4°C using a 20kD MWCO dialysis membrane. Alternatively, use tangential flow filtration.
- Characterization: Measure particle size and PDI via DLS. Determine encapsulation efficiency using Ribogreen assay.
- Storage: Store LNPs in PBS at 4°C for short-term use (≤1 week) or flash-freeze in single-use aliquots at -80°C.
Protocol 2: Complexation of Cas9 RNP with Polymeric Nanoparticles (PEI)
Application: For in vitro delivery of pre-assembled Cas9 RNP. Method:
- RNP Preparation: Pre-complex purified Cas9 protein and sgRNA at a 1:1.2 molar ratio in nuclease-free duplex buffer. Incubate at 25°C for 10 minutes.
- Polymer Solution: Dilute branched PEI (25 kDa) in sterile, nuclease-free 150 mM NaCl to a concentration of 0.1 mg/ml.
- Complexation: Add the PEI solution dropwise to the RNP solution under vortexing to achieve the desired N/P ratio (typically 5-10). Vortex for 30 seconds.
- Incubation: Allow the complexes to form for 30 minutes at room temperature.
- Characterization: Assess complex size and zeta potential via DLS. Confirm complex formation by gel retardation assay (agarose gel electrophoresis).
Protocol 3: Conjugation of Cas9 RNP to Gold Nanoparticles (AuNPs)
Application: For photothermally triggered cytosolic delivery. Method:
- AuNP Functionalization: Incubate 20 nm citrate-capped AuNPs (OD520 ~1) with 1 µM thiolated-PEG5000-COOH for 1 hour. Purify via centrifugation (14,000 x g, 20 min).
- Activation: Activate carboxyl groups on purified AuNPs with 10 mM EDC and 5 mM Sulfo-NHS in MES buffer (pH 6.0) for 20 minutes.
- RNP Conjugation: Incubate activated AuNPs with Cas9 RNP (pre-assembled with a nuclear localization signal) for 2 hours at 4°C in PBS. The RNP is engineered to contain a lysine-rich tag for amine-coupling.
- Quenching & Purification: Quench the reaction with 100 mM glycine for 15 minutes. Purify conjugated AuNPs via centrifugation (3,000 x g, 10 min) to remove unbound RNP.
- Validation: Use SDS-PAGE (staining for protein) and UV-Vis spectroscopy to confirm conjugation.
Diagrams
Title: LNP Formulation via Microfluidics
Title: LNP Endosomal Escape Mechanism
The Scientist's Toolkit
Table 3: Essential Research Reagents & Materials
| Item | Function/Application | Example Vendor/Product |
|---|---|---|
| Ionizable Lipid (e.g., DLin-MC3-DMA) | Core LNP component; enables encapsulation and pH-dependent endosomal escape. | MedChemExpress (HY-108678) |
| Branched Polyethylenimine (PEI), 25 kDa | Cationic polymer for nucleic acid/RNP complexation via "proton sponge" effect. | Sigma-Aldrich (408727) |
| Citrate-Capped Gold Nanoparticles (20 nm) | Core for inorganic NP; surface allows facile conjugation of CRISPR RNP. | Cytodiagnostics (C-20-150-CTAB-DIH) |
| Microfluidic Mixer | Precision instrument for reproducible, scalable LNP formulation. | Precision NanoSystems (NanoAssemblr) |
| Ribogreen Assay Kit | Quantifies encapsulation efficiency of nucleic acid payloads in LNPs. | Invitrogen (R11490) |
| DMG-PEG2000 | PEGylated lipid for LNP surface shielding, modulates pharmacokinetics. | Avanti Polar Lipids (880151) |
| EDC / Sulfo-NHS | Crosslinker chemistry for conjugating proteins/RNPs to functionalized NPs. | Thermo Fisher (A35391) |
| Zeta Potential Analyzer | Instrument for measuring nanoparticle surface charge (DLS/Zeta). | Malvern Panalytical (Zetasizer) |
This document provides Application Notes and Protocols for studying the mechanisms of cellular entry, specifically within the context of CRISPR nanotechnology delivery systems. Efficient intracellular delivery of CRISPR-Cas ribonucleoprotein (RNP) complexes or nucleic acids remains a pivotal challenge. Success hinges on navigating three critical, sequential barriers: (1) Cellular internalization via endocytosis, (2) potential direct entry via membrane fusion (for specific nanocarriers), and (3) the indispensable escape of cargo from endosomal compartments to avoid lysosomal degradation. Understanding and optimizing these steps is fundamental for developing robust, therapeutically viable non-viral CRISPR delivery platforms for gene editing, activation, or repression.
Quantitative Data on Cellular Entry Pathways
Table 1: Characteristics of Major Endocytic Pathways for Nanocarrier Uptake
| Pathway | Size Range (nm) | Key Machinery/Receptor | Dynamin-Dependent? | Common Cargo Example | Relevance to CRISPR Nano-Delivery |
|---|---|---|---|---|---|
| Clathrin-Mediated Endocytosis (CME) | ~100-120 | Clathrin, AP2, LDLR, TransferrinR | Yes | Transferrin, viruses | Primary route for many ligand-targeted lipid nanoparticles (LNPs) and polymeric NPs. |
| Caveolae-Mediated Endocytosis | ~60-80 | Caveolin-1, Cholesterol | Yes | Albumin, SV40 virus | Can avoid lysosomal trafficking; relevant for some lipid-based systems. |
| Macropinocytosis | >200 | Actin, Rac1, Pak1 | No | Fluid, large particles, pathogens | Significant for large or aggregating polyplexes and charge-mediated uptake. |
| CLIC/GEEC Pathway | ~90-200 | GRAF1, Cdc42, Actin | No | Fluid, glycosylphosphatidylinositol (GPI)-anchored proteins | Less characterized for delivery; contributes to non-clathrin uptake of some NPs. |
Table 2: Endosomal Escape Efficiency of Various Nanocarrier Formulations (Illustrative Data)
| Nanocarrier Type | Escape Mechanism | Typical Efficiency Range* | Key Determinants |
|---|---|---|---|
| Cationic Lipid NPs (LNPs) | pH-dependent "fusogenic" disruption | 2-10% | Lipid pKa, fusogenic lipid (e.g., DOPE) content, membrane charge. |
| Polymeric NPs (e.g., PEI) | "Proton Sponge" effect & membrane disruption | 5-20% | Polymer molecular weight, branching, N/P ratio, buffering capacity. |
| Cell-Penetrating Peptide (CPP) Conjugates | Direct translocation or endosomal leakage | 0.1-5% | Peptide sequence (e.g., TAT, Penetratin), charge, hydrophobicity, cargo linkage. |
| pH-Sensitive Polymers (e.g., PBAE) | Conformational change & membrane destabilization at low pH | 5-15% | Polymer backbone chemistry, hydrophobicity, pH-trigger transition point. |
* Efficiency is highly formulation and cell-type dependent. Values represent approximate percentages of internalized cargo that reach the cytosol.
Experimental Protocols
Protocol 3.1: Distinguishing Endocytic Pathways via Pharmacological Inhibition
Objective: To identify the primary endocytic pathway(s) responsible for the cellular uptake of a CRISPR nanocarrier. Key Materials: HeLa or HEK293T cells, CRISPR nanocarrier (e.g., Cas9 RNP-loaded LNP), specific pathway inhibitors (see Reagent Toolkit), serum-free media, fluorescence plate reader or flow cytometer. Workflow:
- Seed cells in a 96-well plate (~2x10⁴ cells/well) and culture for 24h.
- Pre-treat cells with pathway-specific inhibitors for 30-60 min in serum-free media:
- CME: 80 µM Dynasore (dynamin inhibitor) or 10 µg/mL Chlorpromazine.
- Caveolae: 5 µM Filipin III (cholesterol disruptor).
- Macropinocytosis: 50 µM EIPA (Na+/H+ exchanger inhibitor).
- Control: DMSO vehicle only.
- Add nanocarrier (fluorescently labeled, e.g., with Cy5) to inhibitor-containing media. Incubate for 2-4h at 37°C/5% CO₂.
- Wash cells 3x with cold PBS + 0.1% heparin to remove surface-bound particles.
- Lyse cells with 1% Triton X-100. Measure fluorescence intensity (Ex/Em for Cy5) using a plate reader.
- Calculate percentage uptake relative to DMSO control. >50% inhibition indicates a major role for that pathway.
Diagram Title: Pharmacological Inhibition Workflow for Endocytosis
Protocol 3.2: Quantifying Endosomal Escape Using a Split GFP/CFP Assay
Objective: To measure the efficiency of endosomal escape by detecting cargo (e.g., Cas9 protein) in the cytosol. Key Materials: Cells stably expressing GFP1-10 fragment in the cytosol, purified CRISPR-Cas9 protein conjugated to GFP11 tag (Cas9-GFP11), nanocarrier formulation reagents, live-cell imaging microscope or flow cytometer. Workflow:
- Prepare cargo: Conjugate GFP11 peptide (a short β-strand) to purified Cas9 protein via a non-cleavable linker.
- Formulate: Load Cas9-GFP11 into your nanocarrier (e.g., via electrostatic complexation for polyplexes).
- Transduce: Treat GFP1-10 expressing cells with the formulated Cas9-GFP11 nanocarrier. Include controls: free Cas9-GFP11 (no escape expected), Cas9-GFP11 with a known escape agent (e.g., PEI, positive control).
- Incubate: Incubate for 4-48h (time-course recommended).
- Image/Analyze: Use live-cell fluorescence microscopy or flow cytometry to detect reconstituted GFP signal in the cytosol. The GFP signal intensity correlates directly with the amount of Cas9 that has escaped into the cytosol.
- Normalize: Normalize GFP fluorescence to a transfection control (e.g., co-delivered mCherry plasmid) or total protein.
Diagram Title: Split GFP Assay for Endosomal Escape Detection
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Studying Cellular Entry Mechanisms
| Item | Function/Description | Example Supplier/Product |
|---|---|---|
| Dynasore | Cell-permeable inhibitor of dynamin, blocking clathrin- and caveolae-mediated endocytosis. | Sigma-Aldrich, D7693 |
| Chlorpromazine HCl | Disrupts clathrin-coated pit formation by translocating clathrin and AP2 from the cell surface. | Tocris, 0824 |
| EIPA (Amiloride) | Selective inhibitor of the Na+/H+ exchanger, inhibits macropinocytosis. | Sigma-Aldrich, A3085 |
| Filipin III | Binds to membrane cholesterol, disrupting lipid rafts and caveolae-mediated endocytosis. | Cayman Chemical, 70440 |
| LysoTracker Dyes | Fluorescent probes that accumulate in acidic organelles (endosomes/lysosomes) for live-cell imaging. | Thermo Fisher Scientific, L7526 |
| pHrodo Dextran | pH-sensitive dye; fluorescence increases in acidic endosomes. Used to track phagocytosis and endosomal maturation. | Thermo Fisher Scientific, P10361 |
| CellLight Late Endosomes-GFP (Rab7) | Baculovirus system for labeling late endosomes with GFP for live-cell imaging and co-localization studies. | Thermo Fisher Scientific, C10588 |
| Polyethylenimine (PEI), 25 kDa | Standard cationic polymer for nucleic acid delivery; acts as a positive control via the "proton sponge" effect. | Polysciences, 23966 |
| 1,2-Dioleoyl-sn-glycero-3-phosphoethanolamine (DOPE) | A fusogenic, helper lipid commonly used in LNPs to promote endosomal escape via non-bilayer phase formation. | Avanti Polar Lipids, 850725 |
| Endo-Porter | A peptide that delivers cargo to the cytosol by specifically enhancing endosomal release; used as a positive control. | Gene Tools, LLC |
Signaling Pathways in Endocytic Uptake
Diagram Title: Signaling Pathways in Major Endocytic Uptake Routes
Current Landscape and Market-Leading Nanodelivery Technologies (2024)
This document provides a technical overview and comparative analysis of dominant nanodelivery platforms as of 2024, specifically contextualized for advancing CRISPR-Cas machinery delivery in therapeutic and research applications. Efficient delivery remains the primary bottleneck for CRISPR-based therapies, necessitating robust, scalable, and cell-specific nanocarriers. These application notes detail the operating principles, key performance metrics, and selection criteria for lipid-based, polymeric, and inorganic nanoparticle systems.
Table 1: Comparative Performance Metrics of Leading Nanodelivery Platforms for CRISPR (2024)
| Platform Category | Key Commercial/Research Examples (2024) | Avg. Encapsulation Efficiency (%) | In Vivo Delivery Efficiency (Model) | Primary CRISPR Payload | Key Advantage | Major Limitation |
|---|---|---|---|---|---|---|
| Lipid Nanoparticles (LNPs) | Acuitas ALC-0315 analogue; Intellia's proprietary LNP; Arcturus LUNAR | 85-95% | ~45% (mouse hepatocytes) | Cas9 mRNA + sgRNA (RNP possible) | Clinical validation (Onpattro, COVID-19 vaccines); scalable production. | Primarily hepatic tropism; transient expression. |
| Polymeric Nanoparticles | Poly(beta-amino esters) (PBAEs); JetPEI-CRISPR; PEG-PLGA copolymers | 70-90% | ~15-30% (local tumor model) | Plasmid DNA, RNP | Tunable degradation; potential for controlled release; lower cost. | Variable cytotoxicity; lower efficiency vs. LNPs in systemic delivery. |
| Virus-Like Particles (VLPs) | VLP-CRISPR (e.g., Capsid-Encapsulated Cas9-sgRNA); engineered eVLPs | 60-80% (capsid loading) | ~25% (T cells ex vivo) | Pre-assembled RNP | CRISPR component pre-complexed; reduced off-target editing risk. | Complex manufacturing; lower yield; immunogenicity concerns. |
| Gold Nanoparticles (AuNPs) | CRISPR-Gold; spherical nucleic acids (SNAs) | >95% (surface conjugation) | ~10-20% (local muscle/brain) | Cas9 RNP | High stability; precise RNP delivery; minimal toxicity. | Limited to local/regional administration; no systemic applicability. |
| Hybrid & Advanced Systems | Lipid-Polymer Hybrids; GalNAc-conjugated LNPs; Targeted extracellular vesicles | 80-95% | Varies by targeting (e.g., >50% hepatocytes with GalNAc) | Varied (mRNA, RNP) | Emerging; combines advantages; enables active targeting. | Early-stage; complex regulatory path; characterization challenges. |
Detailed Experimental Protocols
Protocol 1: Formulation and In Vitro Testing of CRISPR-LNPs (mRNA payload)
Objective: To prepare LNPs encapsulating Cas9 mRNA and sgRNA targeting a specific genomic locus and evaluate their transfection efficiency and editing efficacy in vitro.
Research Reagent Solutions:
- Lipid Mixture: ALC-0315 (ionizable lipid), DSPC (structural lipid), Cholesterol, DMG-PEG 2000 (PEG-lipid) in ethanol.
- Aqueous Phase: Cas9 mRNA and sgRNA in citrate buffer (pH 4.0).
- Cell Line: HEK293T cells stably expressing a GFP reporter (editing turns GFP off).
- Microfluidic Device: NanoAssemblr Ignite or comparable.
- Analytical Instruments: DLS/Zetasizer for size/zeta potential, RiboGreen assay for encapsulation efficiency, flow cytometer for GFP analysis.
Procedure:
- Lipid Stock Preparation: Dissolve ionizable lipid, DSPC, cholesterol, and PEG-lipid in ethanol at a molar ratio of 50:10:38.5:1.5. Final total lipid concentration: 10 mM.
- Aqueous Phase Preparation: Combine Cas9 mRNA and sgRNA at a 1:1.2 molar ratio in 25 mM citrate buffer, pH 4.0.
- LNP Formation: Using a microfluidic device, mix the ethanol lipid phase and the aqueous mRNA phase at a 1:3 volumetric flow rate ratio (total flow rate 12 mL/min). Collect the effluent in a vessel containing PBS (pH 7.4) for buffer exchange.
- Purification & Characterization: Dialyze or use tangential flow filtration against PBS to remove ethanol. Measure particle size (target: 70-100 nm), PDI (<0.2), and zeta potential (near neutral). Use a RiboGreen assay to determine encapsulation efficiency (>85% target).
- In Vitro Transfection: Seed HEK293T-GFP cells in a 24-well plate. At 70% confluency, treat with CRISPR-LNPs at an mRNA dose of 50-200 ng/well. Include untreated and lipid-only controls.
- Efficacy Analysis: Harvest cells 72 hours post-transfection. Analyze GFP fluorescence loss via flow cytometry. Extract genomic DNA for T7E1 or NGS assay to quantify indel frequency at the target locus.
Protocol 2: Formulation of Polymeric Nanoparticles for CRISPR RNP Delivery (PBAE-based)
Objective: To synthesize Poly(beta-amino ester) nanoparticles for the delivery of pre-assembled Cas9 RNP.
Research Reagent Solutions:
- Polymer: End-modified PBAE (e.g., acrylate-terminated, synthesized from 1,4-butanediol diacrylate and 5-amino-1-pentanol).
- Payload: Recombinant Cas9 protein complexed with sgRNA (RNP).
- Buffer: Sodium acetate buffer (25 mM, pH 5.2) and HBSS.
- Purification: 100 kDa molecular weight cut-off (MWCO) centrifugal filters.
Procedure:
- Polymer Preparation: Dissolve PBAE polymer in anhydrous DMSO at 100 mg/mL. Dilute to 10 mg/mL in sodium acetate buffer (pH 5.2) prior to use.
- RNP Complexation: Pre-complex Cas9 protein and sgRNA at a 1:1.2 molar ratio in nuclease-free duplex buffer. Incubate at 25°C for 10 minutes to form RNP.
- Nanoparticle Assembly: Rapidly mix the PBAE solution (in acetate buffer) with the RNP solution via pipetting or vortexing. Use a polymer:RNP weight ratio optimized between 30:1 and 60:1. Incubate at room temperature for 30 minutes to allow self-assembly.
- Purification: Transfer the mixture to a 100 kDa MWCO centrifugal filter. Centrifuge at 3000 x g to remove unencapsulated RNP and free polymer. Resuspend the nanoparticles in HBSS or cell culture medium.
- Characterization: Measure hydrodynamic diameter and PDI via DLS. Confirm RNP complexation via gel retardation assay. Test in vitro delivery in relevant cell lines, assessing cytotoxicity (MTT assay) and editing efficiency (ICE analysis).
Signaling Pathways and Workflow Visualizations
Title: LNP Hepatic Delivery & Endosomal Escape Pathway
Title: CRISPR-LNP Preparation & Testing Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for CRISPR Nanodelivery Research
| Item | Function & Role in Protocol | Example Vendor/Product (2024) |
|---|---|---|
| Ionizable Cationic Lipid | Critical for LNP self-assembly, endosomal escape via protonation. | ALC-0315 (MedKoo), SM-102 (Avanti), proprietary lipids (Precision NanoSystems). |
| Recombinant Cas9 Protein | Pre-assembled with sgRNA to form RNP for direct delivery, reducing persistence. | TrueCut Cas9 Protein (Thermo Fisher), Alt-R S.p. Cas9 Nuclease (IDT). |
| CleanCap Cas9 mRNA | Chemically modified mRNA for high-yield, low-immunogenicity LNP encapsulation. | Trilink BioTechnologies. |
| Chemically Modified sgRNA | Enhanced nuclease stability and reduced immunogenicity. | Alt-R CRISPR-Cas9 sgRNA (IDT). |
| Microfluidic Mixer | Enables reproducible, scalable synthesis of monodisperse nanoparticles. | NanoAssemblr Ignite (Precision NanoSystems), Staggered Herringbone Mixer chips. |
| Dynamic Light Scattering (DLS) Instrument | Measures nanoparticle hydrodynamic diameter, PDI, and zeta potential. | Zetasizer Pro (Malvern Panalytical). |
| RiboGreen / Quant-iT Assay | Fluorescent quantification of nucleic acid encapsulation efficiency in LNPs. | Invitrogen RiboGreen RNA Quantitation Kit. |
| Infernal Editor (ICE) Analysis Tool | Web-based or stand-alone software for quantifying CRISPR editing efficiency from Sanger data. | Synthego ICE Analysis (synthego.com). |
| GalNAc Conjugation Ligand | Enables active targeting of nanoparticles to hepatocytes via asialoglycoprotein receptor. | GalNAc-PEG-DSPE (BroadPharm). |
| End-modified PBAE Polymer | Biodegradable, cationic polymer for RNP complexation and pH-responsive release. | Custom synthesis per published methods; PolySciTech. |
From Bench to Bedside: Designing and Applying CRISPR Nanodelivery Systems
Within the thesis research on CRISPR nanotechnology delivery systems, the choice of active CRISPR cargo—sgRNA, Cas9 mRNA, or pre-formed Ribonucleoprotein (RNP)—is fundamental. Each presents distinct formulation challenges and opportunities for encapsulation into lipid nanoparticles (LNPs), polymeric nanoparticles, and other nanocarriers. This Application Notes and Protocols document details current strategies, quantitative benchmarks, and standardized protocols for these critical formulation steps, enabling efficient delivery for therapeutic gene editing.
Cargo Characteristics & Formulation Rationale
The physicochemical properties of the CRISPR cargo dictate the encapsulation strategy.
Table 1: Key Properties of CRISPR Cargos Influencing Encapsulation
| Cargo Type | Size (kDa/nm) | Charge (at pH 7) | Stability | Primary Formulation Goal |
|---|---|---|---|---|
| sgRNA | ~13-15 kDa / ~2-4 nm | Negative (polyanion) | Low (RNase sensitive) | Condensation/protection; often co-encapsulated with cationic carrier. |
| Cas9 mRNA | ~300-4500 kDa / Long linear coil | Negative (polyanion) | Low (RNase sensitive) | Efficient encapsulation in ionizable lipid LNPs; circRNA for enhanced stability. |
| RNP | ~160 kDa (Cas9) + sgRNA / ~5-10 nm | Negative (net -10 to -20) | Moderate (protein degradation) | Maintain functional quaternary structure; achieve high loading efficiency. |
Key Research Reagent Solutions
Table 2: Essential Toolkit for CRISPR Cargo Formulation
| Reagent/Material | Supplier Examples | Function in Formulation |
|---|---|---|
| Ionizable Cationic Lipid (e.g., DLin-MC3-DMA, SM-102) | MedChemExpress, Avanti | Core component of LNPs; positively charged at low pH to complex nucleic acids, neutral at physiological pH to reduce toxicity. |
| Helper Lipids (DSPC, Cholesterol) | Avanti Polar Lipids | Enhance nanoparticle stability, fluidity, and fusion with endosomal membrane. |
| PEGylated Lipid (e.g., DMG-PEG2000) | Avanti Polar Lipids | Controls nanoparticle size, improves colloidal stability, and reduces opsonization. |
| Cationic Polymer (e.g., PEI, PBAE) | Polysciences, Sigma-Aldrich | Condenses nucleic acid cargoes via electrostatic interactions for polymeric nanoparticle formation. |
| Microfluidic Device (e.g., NanoAssemblr) | Precision NanoSystems | Enables reproducible, rapid mixing for consistent LNP production via rapid mixing. |
| RNase Inhibitor | New England Biolabs, Takara | Critical for maintaining integrity of RNA-based cargoes (sgRNA, mRNA) during formulation. |
| Size Exclusion Chromatography Columns | Cytiva (Sephadex) | Purifies formulated nanoparticles from unencapsulated cargo and free reagents. |
| Fluorescent Dye-Labeled Cargo | Trilink Biotechnologies | Allows tracking of encapsulation efficiency and cellular uptake (e.g., Cy5-sgRNA, FAM-mRNA). |
Detailed Experimental Protocols
Protocol 3.1: LNP Encapsulation of Cas9 mRNA/sgRNA via Microfluidic Mixing
This protocol is adapted from current good manufacturing practices for nucleic acid LNPs.
A. Materials Preparation
- Aqueous Phase: Cas9 mRNA and/or sgRNA in citrate buffer (pH 4.0). Include RNase inhibitor.
- Lipid Phase: Ionizable lipid, DSPC, Cholesterol, and DMG-PEG2000 dissolved in ethanol at molar ratios specific to cargo (e.g., 50:10:38.5:1.5 mol%).
- Equipment: Microfluidic mixer (e.g., NanoAssemblr Ignite), syringe pumps, collection vial.
B. Procedure
- Load the aqueous phase and lipid phase into separate syringes.
- Set total flow rate (TRR) to 12-15 mL/min and flow rate ratio (FRR, aqueous:lipid) to 3:1.
- Initiate simultaneous mixing through the microfluidic cartridge. Collect effluent in a vial.
- Immediately dialyze or use size exclusion chromatography against PBS (pH 7.4) for 2 hours to remove ethanol and exchange buffer.
- Filter sterilize (0.22 µm) and store at 4°C.
C. Analysis
- Encapsulation Efficiency (EE%): Use RiboGreen assay. Measure fluorescence of sample ± Triton X-100 detergent. EE% = (1 – [Free RNA]/[Total RNA]) x 100.
- Size and PDI: Determine by dynamic light scattering (DLS).
- Zeta Potential: Measure in PBS via electrophoretic light scattering.
Protocol 3.2: Co-encapsulation of Cas9 RNP via Charge-Mediated Complexation
This protocol leverages the negative charge of RNP for complexation with cationic materials prior to nanoparticle assembly.
A. Materials Preparation
- RNP Complex: Pre-complex purified Cas9 protein with sgRNA (molar ratio ~1:1.2) in nuclease-free buffer for 10 min at 25°C.
- Cationic Carrier: e.g., a biodegradable cationic polymer or lipid.
B. Procedure
- Dilute the pre-formed RNP complex in HEPES buffer (pH 7.4).
- Slowly add the cationic carrier solution to the RNP solution under vigorous vortexing. Incubate 15-20 min to form cationic RNP complexes.
- For LNP encapsulation: Use these cationic RNP complexes as the aqueous phase in Protocol 3.1.
- For polymeric encapsulation: Add the cationic RNP complexes to a solution of amphiphilic block copolymer (e.g., PLGA-PEG) under sonication or nanoprecipitation.
- Purify via size exclusion chromatography.
D. Analysis
- EE%: Use fluorescently labeled sgRNA or Cas9 protein and compare signals pre- and post-purification.
- Gel Shift Assay: Run purified nanoparticles on agarose gel; retained RNP in the well indicates successful complexation/encapsulation.
- In Vitro Activity: Perform T7E1 or next-generation sequencing (NGS) assay on edited genomic loci from treated cells.
Quantitative Performance Benchmarks
Table 3: Typical Performance Metrics for Different Formulation Strategies
| Cargo | Nanocarrier | Avg. Size (nm) | Avg. PDI | Avg. Encapsulation Efficiency | Key Functional Readout (In Vitro) |
|---|---|---|---|---|---|
| Cas9 mRNA + sgRNA | Ionizable LNP | 80-120 | <0.2 | >90% | >70% protein expression (HeLa) |
| RNP | Cationic Polymer-LNP Hybrid | 100-150 | <0.25 | 60-80% | >40% gene editing (HEK293, EMX1 locus) |
| RNP | Gold Nanoparticle (AuNP) Conjugate | 15-30 (core) | <0.15 | N/A (surface bound) | >30% gene editing (primary T-cells) |
| sgRNA/Cas9 mRNA | Polymeric Micelle | 50-80 | <0.3 | 70-85% | >50% protein expression (HepG2) |
Visualized Workflows and Mechanisms
Title: CRISPR Cargo Formulation Pathways
Title: LNP Endosomal Escape Mechanism
Within the broader thesis on CRISPR-Cas9 nanotechnology delivery systems for therapeutic gene editing, the choice of targeting methodology is paramount. Efficient delivery to target cells and tissues, while minimizing off-target effects, is a critical bottleneck. This application note details and contrasts the two principal targeting strategies: the passive Enhanced Permeability and Retention (EPR) effect and active targeting using ligands and antibodies. The focus is on their application in lipid nanoparticle (LNP) and polymeric nanocarrier systems for CRISPR ribonucleoprotein (RNP) or plasmid DNA delivery.
Core Principles & Quantitative Comparison
Table 1: Comparative Analysis of Passive vs. Active Targeting Methodologies
| Parameter | Passive Targeting (EPR Effect) | Active Targeting (Ligand/Antibody) |
|---|---|---|
| Primary Mechanism | Exploits pathological leaky vasculature and poor lymphatic drainage in tumors/inflamed tissues. | Utilizes molecular recognition between surface-conjugated ligand/antibody and specific cell-surface receptor (e.g., folate receptor, transferrin receptor, EGFR). |
| Targeting Specificity | Low to moderate. Accumulates in any tissue with enhanced vascular permeability (e.g., tumors, sites of inflammation). | High. Binds specifically to cell populations overexpressing the target receptor. |
| Cellular Uptake Pathway | Primarily non-specific endocytosis/phagocytosis following extravasation. | Receptor-mediated endocytosis, often leading to more efficient internalization. |
| Key Dependent Factors | Tumor type, vascularization, interstitial pressure, particle size (10-200 nm optimal), surface charge (neutral/ slightly negative). | Receptor density, ligand affinity, ligand density on nanoparticle, binding site accessibility. |
| Typical In Vivo Tumor Accumulation | 0.5-5% of injected dose per gram of tumor (%ID/g). Highly variable. | Can increase accumulation by 1.5-3x compared to non-targeted EPR alone. |
| Major Limitation | Heterogeneity across patients and tumor types; high interstitial pressure limits penetration. | Potential immunogenicity; "binding site barrier" effect can limit deep tumor penetration. |
| Best Suited For | First-generation nanotherapies; delivery to tissues with inherent EPR (e.g., liver, spleen, tumors with strong EPR). | Precision delivery to defined cell subsets; enhancing cellular internalization post-accumulation. |
Detailed Protocols
Protocol 3.1: Evaluating Passive EPR Effect of CRISPR-LNPs in a Murine Tumor Model
Objective: To quantify the passive accumulation of fluorescently labeled, CRISPR-loaded LNPs in a subcutaneous xenograft tumor via the EPR effect.
Materials:
- CRISPR-LNP Formulation: LNPs encapsulating Cy5.5-labeled sgRNA/Cas9 RNP, 80-100 nm diameter, -5 to +5 mV zeta potential.
- Animal Model: Immunodeficient mice with subcutaneously implanted human tumor cells (e.g., HepG2 liver tumor, ~500 mm³ volume).
- Imaging System: In vivo fluorescence imaging system (IVIS).
Procedure:
- Administration: Inject CRISPR-Cy5.5 LNPs intravenously via the tail vein (dose: 3 mg lipid/kg mouse weight).
- In Vivo Imaging: Anesthetize mice at predetermined time points (1, 4, 12, 24, 48 h). Acquire fluorescence images (Ex/Em: 675/720 nm) using standardized settings.
- Ex Vivo Quantification: Euthanize mice at 48 h. Harvest tumor, liver, spleen, kidney, lung, and heart. Weigh organs and image ex vivo.
- Data Analysis: Use region-of-interest (ROI) analysis to quantify fluorescence intensity. Calculate tumor accumulation as %ID/g using a standard curve of known LNP concentrations.
Protocol 3.2: Conjugation of Targeting Ligands to CRISPR Nanocarriers
Objective: To functionalize the surface of polymeric CRISPR nanoparticles (e.g., PLGA-PEG) with a folate ligand for active targeting to folate receptor-positive cells.
Materials:
- Nanoparticles (NPs): PLGA-PEG-COOH nanoparticles loaded with CRISPR plasmid DNA.
- Targeting Ligand: Folate-PEG-NH₂.
- Coupling Reagents: 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide (EDC) and N-hydroxysuccinimide (NHS).
Procedure:
- Activation of NP Carboxyl Groups: Resuspend 10 mg of CRISPR-PLGA-PEG-COOH NPs in 2 mL of MES buffer (0.1 M, pH 5.5). Add EDC (10 mM final) and NHS (15 mM final). React for 15 min at room temperature with gentle stirring.
- Ligand Conjugation: Purify activated NPs via centrifugal filtration (100kDa MWCO) to remove excess EDC/NHS. Immediately resuspend in PBS (pH 7.4). Add Folate-PEG-NH₂ (5 mol% relative to surface PEG) and react for 2 h at RT.
- Purification & Characterization: Purify Folate-CRISPR-NPs via dialysis (PBS, 24 h). Characterize final product for size (DLS), zeta potential, ligand density (via UV-Vis for folate), and CRISPR encapsulation efficiency.
Protocol 3.3: In Vitro Validation of Active Targeting Specificity
Objective: To compare cellular uptake and gene editing efficiency of targeted vs. non-targeted CRISPR nanoparticles in receptor-positive vs. receptor-negative cell lines.
Materials:
- Cell Lines: Receptor-positive (KB cells, high FRα) and receptor-negative (A549 cells, low FRα).
- Nanoparticles: Folate-targeted and non-targeted CRISPR-Cas9 RNPLNPs (encapsulating GFP-targeting RNP).
- Assays: Flow cytometry, fluorescence microscopy, T7E1 assay.
Procedure:
- Competitive Binding Assay: Seed cells in 24-well plates. Pre-treat FRα+ cells with 1 mM free folic acid for 30 min. Add Cy5-labeled Folate-CRISPR-NPs. Incubate for 4 h at 37°C.
- Quantitative Uptake: Analyze cells by flow cytometry. Compare mean fluorescence intensity (MFI) of Cy5 signal between: a) FRα+ cells with Folate-NPs, b) FRα+ cells with Folate-NPs + free folate blocker, c) FRα+ cells with non-targeted NPs, d) FRα- cells with Folate-NPs.
- Functional Gene Editing: Transfert cells with NPs containing GFP-targeting RNP. After 72 h, analyze GFP knockout efficiency via flow cytometry (loss of GFP signal) and genomic cleavage via T7E1 assay on extracted DNA.
Visualizations
Diagram 1: Passive Targeting Overview
Diagram 2: Active Targeting Mechanism
Diagram 3: Active Targeting Validation Workflow
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for Targeting Methodologies in CRISPR Delivery
| Reagent / Material | Function / Purpose | Example Vendor/Product |
|---|---|---|
| PLGA-PEG-COOH Copolymer | Forms biodegradable nanoparticle core; PEG provides stealth, COOH allows ligand conjugation. | Sigma-Aldrich, PolySciTech (AP-100PEG-COOH) |
| Ionizable Cationic Lipid (DLin-MC3-DMA) | Key component of LNPs for CRISPR encapsulation; promotes endosomal escape. | MedKoo Biosciences (Cat# 510001) |
| Folate-PEG-NH₂ | Targeting ligand for conjugation to nanoparticles; targets folate receptor-α overexpressing cells. | Nanocs (PG2-FANL-5k) |
| Anti-EGFR scFv or Cetuximab | Antibody/ fragment for targeting EGFR-overexpressing cancers. Can be conjugated to NPs. | Creative Biolabs (Multiple scFv offerings) |
| EDC & NHS Crosslinkers | Activate carboxyl groups on nanoparticles for covalent conjugation of amine-containing ligands. | Thermo Fisher Scientific (Cat# PG82079) |
| Fluorescent Dye (Cy5.5, DiR) | Label nanoparticles for quantitative in vivo biodistribution and tumor accumulation studies. | Lumiprobe (Cy5.5 NHS ester) |
| T7 Endonuclease I (T7E1) | Detects CRISPR-Cas9 induced indel mutations in genomic DNA to confirm functional delivery. | NEB (Cat# M0302L) |
| Dynamic Light Scattering (DLS) System | Measures nanoparticle size (hydrodynamic diameter), polydispersity index (PDI), and zeta potential. | Malvern Panalytical (Zetasizer) |
| In Vivo Imaging System (IVIS) | Enables non-invasive, longitudinal tracking of fluorescently labeled nanoparticles in live animals. | PerkinElmer (IVIS Spectrum) |
Within the broader research thesis on CRISPR nanotechnology delivery systems, selecting the optimal in vivo administration route is paramount. The choice between systemic, local, and organ-specific delivery directly impacts biodistribution, targeting efficacy, off-target effects, and therapeutic outcome. This document provides application notes and detailed protocols for these routes, framed for advanced researchers developing non-viral, nanoparticle-based CRISPR-Cas9 delivery platforms.
Systemic Administration (Intravenous Injection)
Systemic delivery, primarily via intravenous (IV) injection, is the standard for achieving whole-body distribution, essential for targeting disseminated sites or hematopoietic systems.
Application Notes:
- Primary Use: Targets reticuloendothelial system (RES) organs (liver, spleen), tumor vasculature (via EPR effect), and circulating cells.
- Key Challenge: Rapid clearance by the RES, serum protein opsonization, and potential off-target accumulation.
- Nanoparticle Design Criterion: Requires stealth properties (e.g., PEGylation) and often active targeting ligands (e.g., galactose for hepatocyte ASGPR) to overcome biological barriers.
Quantitative Data Summary: Table 1: Comparison of Systemic Delivery Outcomes for Lipid Nanoparticle (LNP)-CRISPR Formulations
| Target Organ | Nanoparticle Surface Mod | Average % Editing Efficiency (in vivo) | Peak Expression Time | Major Clearance Organs |
|---|---|---|---|---|
| Liver | GalNAc ligand | 60-80% in hepatocytes | 48-72 hours | Liver, Spleen |
| Liver | PEG only (stealth) | 20-40% in hepatocytes | 24-48 hours | Liver, Spleen |
| Spleen | PEG only | 10-25% in APCs | 24 hours | Liver, Spleen |
| Lung | Cationic charge | 5-15% in endothelial cells | 8-24 hours | Liver, Lungs |
| Tumors (xenograft) | EPR effect (no ligand) | 1-10% (highly variable) | 24-96 hours | Liver, Tumors |
Protocol: Tail-Vein IV Injection for Murine Liver Editing
Objective: Deliver CRISPR-LNPs systemically to achieve hepatocyte-specific gene editing.
Materials:
- CRISPR-LNP Formulation: Cas9 mRNA/sgRNA or RNP encapsulated in GalNAc-targeted LNPs.
- Animals: C57BL/6 mice (8-10 weeks).
- Equipment: Animal warmer, 29G insulin syringes, restraint device, alcohol wipes.
Procedure:
- Preparation: Warm mouse tail under infrared lamp (37°C for 1-2 min) to vasodilate veins.
- Restraint: Secure mouse in a tail-vein injection restrainer.
- Injection: Wipe tail with alcohol. Using a 29G syringe, inject formulation (dose: 0.5-1.0 mg mRNA/kg or 2-5 mg lipid/kg) into a lateral tail vein. Total volume ≤ 200 µL for a 25g mouse.
- Post-injection: Apply gentle pressure to the site, return animal to cage.
- Analysis: Euthanize animals at 72-hour post-injection. Harvest liver, process for genomic DNA extraction, and assess editing via NGS or T7E1 assay.
The Scientist's Toolkit: Key Reagents for Systemic CRISPR-LNP Delivery
| Reagent/Material | Function/Explanation |
|---|---|
| PEG-DMG or PEG-DSPE | Lipid conjugate providing "stealth" properties, reduces opsonization and extends circulation half-life. |
| Ionizable Cationic Lipid (e.g., DLin-MC3-DMA) | Enables RNA encapsulation during nano-precipitation and endosomal escape post-cellular uptake. |
| GalNAc-PEG-DSPE Ligand | Targets asialoglycoprotein receptor (ASGPR) on hepatocytes for active, liver-specific targeting. |
| Cholesterol | Stabilizes lipid bilayer structure of the nanoparticle. |
| DSPC or DOPE | Helper phospholipids that contribute to membrane integrity and fusogenicity. |
| Cas9 mRNA (or purified protein) | The effector molecule for gene editing. mRNA allows in situ translation; RNP offers faster action. |
| sgRNA (chemically modified) | Guides the Cas9 nuclease to the specific genomic locus. Chemical modifications enhance stability. |
Diagram Title: Systemic IV Delivery Pathway & Barriers
Local Administration
Local delivery involves direct injection into a specific tissue or cavity, minimizing systemic exposure and enhancing local concentration.
Application Notes:
- Primary Use: Ideal for accessible sites (eye, skin, muscle, brain, solid tumors) and for creating local therapeutic niches.
- Key Challenge: Limited diffusion from injection site, potential tissue damage, and applicability only to localized diseases.
- Nanoparticle Design Criterion: Often requires formulations with tissue-retentive properties (e.g., hydrogels) and may prioritize intracellular delivery over stealth.
Protocol: Intratumoral Injection for Solid Tumor Editing
Objective: Directly deliver CRISPR nanoparticles to a subcutaneous tumor to disrupt an oncogene.
Materials:
- Formulation: CRISPR-Cas9 RNP complexed with cationic polymer (e.g., PEI) or lipidoids.
- Tumor Model: Mice bearing subcutaneous syngeneic or xenograft tumors (~100-150 mm³).
- Equipment: 30G syringe, calipers.
Procedure:
- Tumor Measurement: Anesthetize mouse. Measure tumor dimensions with calipers.
- Injection: Insert 30G needle at a shallow angle into the tumor mass. Inject formulation in multiple small boluses (total volume 20-50 µL) while slowly retracting needle to distribute material.
- Post-injection: Monitor for leakage. Allow animal to recover.
- Analysis: Harvest tumor 3-7 days post-injection. Process for: (a) Western blot for target protein knockdown, (b) TIDE analysis of editing, (c) IHC for tumor proliferation markers.
Organ-Specific Administration (Non-IV)
These routes exploit unique anatomical access points to deliver nanoparticles directly to an organ, bypassing systemic filtration.
Quantitative Data Summary: Table 2: Efficacy Metrics for Organ-Specific Administration Routes
| Administration Route | Target Organ | Typical Formulation | Estimated Delivery Efficiency to Target Cells | Key Advantage |
|---|---|---|---|---|
| Intranasal (IN) | Lungs | Chitosan or PLGA nanoparticles | 15-40% of lung epithelial cells | Non-invasive, targets alveolar and airway cells |
| Intracerebroventricular (ICV) or Intrathecal (IT) | Central Nervous System | Cationic LNPs or AAVs | High local concentration in CSF/spinal cord | Bypasses BBB, direct CNS access |
| Subretinal (SR) or Intravitreal (IVT) | Eye (Retina) | PEGylated nanoparticles | High in retinal pigmented epithelium (SR) | Compartmentalized, minimal systemic spillover |
Protocol: Intranasal Instillation for Pulmonary Delivery
Objective: Deliver CRISPR nanoparticles to the lung epithelium for correcting genetic defects (e.g., in cystic fibrosis models).
Materials:
- Formulation: Cas9/sgRNA RNP complexed with chitosan nanoparticles or LNPs.
- Animals: Anesthetized mouse or rat.
- Equipment: Micropipette with fine tip, Isoflurane anesthesia setup.
Procedure:
- Anesthesia: Deeply anesthetize mouse using isoflurane (3-4% induction, 1-2% maintenance).
- Positioning: Place mouse on its back in a supine position on a slanted platform (~30°).
- Instillation: Using a pipette tip, slowly administer 20-40 µL of formulation dropwise onto the nares. Allow animal to inhale each droplet naturally.
- Recovery: Keep animal supine until fully recovered from anesthesia to ensure deposition in lungs.
- Analysis: Harvest lung tissue 5-7 days later. Perform bronchoalveolar lavage (BAL) for immune profiling and homogenize lung for genomic editing analysis.
Diagram Title: Organ-Specific Delivery Routes & Advantages
Protocol: Intracerebroventricular (ICV) Injection in Neonates
Objective: Deliver CRISPR nanoparticles to the central nervous system of neonatal mice for brain-wide gene editing.
Materials:
- Formulation: CRISPR-Cas9 RNP encapsulated in cationic, brain-penetrant LNPs.
- Animals: Postnatal day 0-2 (P0-P2) mouse pups.
- Equipment: Fine glass capillary needle (calibrated), microinjector, stereotaxic apparatus for neonates, ice bucket for cryoanesthesia.
Procedure:
- Anesthesia: Induce cryoanesthesia by placing pup on ice for 2-3 minutes until immobile and unresponsive to toe pinch.
- Positioning: Secure pup in a custom neonatal stereotaxic device. Identify bregma.
- Injection: Insert glass capillary needle at coordinates: 2 mm rostral to lambda, 1 mm lateral to midline, 2 mm depth. Inject 2-3 µL of formulation at a slow, steady rate (1 µL/min).
- Post-injection: Leave needle in place for 1 min before withdrawal. Warm pup on a heating pad until fully active, then return to dam.
- Analysis: Analyze brain tissue at weaning age (P21) via sequencing or immunohistochemistry to assess widespread editing.
The integration of advanced nanotechnology with a strategic choice of administration route is critical for unlocking the full in vivo potential of CRISPR-Cas9 therapeutics. Systemic IV delivery, when combined with sophisticated targeting ligands, enables precise organ editing. Local and organ-specific routes offer powerful alternatives for compartmentalized diseases, often with superior safety profiles. The protocols herein provide a foundational framework for empirical evaluation within a comprehensive thesis on optimizing delivery systems for genome editing.
This article details Application Notes and Protocols for CRISPR-based therapeutics, framed within the ongoing research thesis on advanced nanotechnology delivery systems designed to overcome in vivo delivery barriers.
Table 1: CRISPR Therapeutic Applications & Key Metrics
| Disease Area | Target Gene / Pathway | Delivery System (Nanotech Focus) | Key Efficacy Metric (Recent Preclinical/Clinical) | Primary Challenge |
|---|---|---|---|---|
| Genetic (e.g., Transthyretin Amyloidosis) | TTR (Mutant allele) | Lipid Nanoparticles (LNPs) | >90% serum TTR reduction in patients (NTLA-2001 trial) | Durability of effect; organ-specific targeting |
| Oncology (Solid Tumors) | PD-1 (in T cells) | Polymer-based or Virus-like Particle (VLP) | ~20% editing in tumor-infiltrating lymphocytes in vivo | Tumor microenvironment penetration; immune cell specificity |
| Oncology (CAR-T Engineering) | TRAC, B2M | Electroporation (ex vivo) | >95% knockout efficiency in primary T cells | Translating ex vivo efficiency to in vivo editing |
| Infectious (HIV-1) | HIV proviral DNA (LTR, gag) | GalNAc-conjugated LNP or AAV | 2-3 log reduction in viral reservoirs in humanized mice | Reaching latent reservoir cells; off-target risks |
| Infectious (SARS-CoV-2) | Viral RNA (via Cas13) | LNP or extracellular vesicle | >99% reduction in viral load in lung tissue (murine model) | Prophylactic vs. therapeutic timing; immune response to system |
Table 2: Nanocarrier Performance Comparison for Liver Delivery
| Nanocarrier Type | Average Size (nm) | Surface Charge (Zeta, mV) | Primary Targeting Ligand | Reported Editing Efficiency In Vivo (Liver) |
|---|---|---|---|---|
| Ionizable LNPs | 70-100 | -2 to +5 | None (apolipoprotein adsorption) | 40-60% (hepatocytes) |
| Polymeric NPs (e.g., PBAE) | 80-150 | +20 to +40 | GalNAc | 30-50% (hepatocytes) |
| Gold Nanoparticles (AuNPs) | 15-25 | -30 to -40 | PEG only (passive) | 10-20% (Kupffer cells) |
| DNA Nanoclews | ~50 | -15 to -25 | Transferrin | 15-25% (hepatocytes) |
Experimental Protocols
Protocol 1: In Vivo CRISPR-Cas9 Delivery for Liver Genetic Disease (LNP-Mediated)
Objective: To achieve targeted gene knockout in hepatocytes for treating hereditary transthyretin amyloidosis (hATTR).
Materials: See Scientist's Toolkit (Table 3).
Methodology:
- Formulation: Prepare ionizable lipid (e.g., DLin-MC3-DMA), DSPC, cholesterol, and PEG-lipid at a molar ratio of 50:10:38.5:1.5. Dissolve in ethanol.
- Aqueous Phase: Prepare sodium acetate buffer (pH 4.0) containing CRISPR components (sgRNA and Cas9 mRNA).
- Microfluidic Mixing: Use a precision microfluidic mixer. Combine the ethanol lipid phase and aqueous phase at a 1:3 volumetric flow rate ratio (total flow rate 12 mL/min) to form LNPs via rapid precipitation.
- Buffer Exchange & Characterization: Dialyze against 1X PBS (pH 7.4) for 4 hours. Characterize particle size (DLS), PDI, and zeta potential. Measure encapsulation efficiency using RiboGreen assay.
- Animal Dosing: Administer formulated LNPs via tail-vein injection to a murine model of hATTR at a dose of 1.0 mg Cas9 mRNA/kg body weight.
- Analysis (14 days post-injection):
- Efficacy: Collect serum, quantify TTR protein reduction via ELISA.
- Editing: Isolate genomic DNA from liver. Assess indel frequency at the TTR locus via next-generation sequencing (NGS) of PCR-amplified target region.
- Safety: Monitor serum alanine aminotransferase (ALT) levels. Perform off-target analysis via GUIDE-seq or related unbiased methods.
Protocol 2: Ex Vivo Engineering of PD-1 Knockout T Cells for Solid Tumors
Objective: Generate tumor-infiltrating lymphocytes (TILs) with disrupted PD-1 checkpoint for enhanced anti-tumor activity.
Materials: See Scientist's Toolkit (Table 3).
Methodology:
- TIL Isolation & Activation: Isolate TILs from human tumor digest or peripheral blood mononuclear cells (PBMCs). Activate cells using anti-CD3/CD28 antibodies in TexMACS medium with IL-2 (3000 IU/mL) for 48 hours.
- RNP Complex Formation: Chemically synthesize sgRNA targeting the PDCD1 (PD-1) gene exon 1. Complex high-purity S. pyogenes Cas9 protein with sgRNA at a molar ratio of 1:2.5 in PBS. Incubate at room temperature for 15 min to form ribonucleoprotein (RNP).
- Electroporation: Wash activated T cells, resuspend in electroporation buffer. Add RNP complex (final concentration 5 µM Cas9) to cell suspension. Electroporate using a 4D-Nucleofector (program EO-115). Immediately transfer cells to pre-warmed culture medium.
- Expansion & Validation: Expand edited T cells for 7-10 days with IL-2.
- Flow Cytometry: Assess PD-1 surface protein knockout efficiency.
- Functional Assay: Co-culture edited T cells with target tumor cells; measure IFN-γ secretion (ELISA) and tumor cell killing (incucyte-based assay).
- In Vivo Efficacy: Use immunodeficient NSG mice with established human tumor xenografts. Adoptively transfer 5-10 x 10^6 edited PD-1 knockout T cells. Monitor tumor volume weekly compared to control groups.
Diagrams
Title: LNP-Mediated CRISPR Delivery to Hepatocytes
Title: PD-1 KO T Cell Bypasses Tumor Checkpoint
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Featured Protocols
| Item | Function / Role | Example Product / Type |
|---|---|---|
| Ionizable Cationic Lipid | Core component of LNP; enables nucleic acid encapsulation and endosomal escape. | DLin-MC3-DMA, SM-102 |
| sgRNA (synthetic) | Guides Cas nuclease to specific genomic DNA sequence. | Chemically modified, HPLC-purified sgRNA |
| Cas9 mRNA | Template for in vivo translation of nuclease; reduced immunogenicity vs. protein. | Pseudouridine-modified, capped & tailed mRNA |
| GalNAc Ligand | Targets nanocarriers to hepatocyte-specific asialoglycoprotein receptor (ASGPR). | N-Acetylgalactosamine (GalNAc), conjugated to lipid/polymer |
| Nucleofector System | Enables high-efficiency ex vivo delivery of RNP complexes into primary immune cells. | 4D-Nucleofector X Unit (Lonza) |
| Anti-CD3/CD28 Beads | Polyclonal activation of primary T cells prior to editing, enhancing viability. | Dynabeads Human T-Activator CD3/CD28 |
| RiboGreen Assay Kit | Quantifies encapsulated nucleic acid in LNPs post-formulation. | Quant-iT RiboGreen RNA Assay Kit (Thermo Fisher) |
| INDEL Detection Kit | Facilitates analysis of CRISPR editing efficiency by NGS. | Illumina CRISPResso2 pipeline or IDT xGen NGS solutions |
Application Note 1: Targeting OncogenicKrasG12D in Pancreatic Cancer
Study Overview: A 2024 study demonstrated a lipid nanoparticle (LNP) co-delivering Cas9 mRNA and a single guide RNA (sgRNA) targeting the oncogenic KrasG12D mutation in a genetically engineered pancreatic ductal adenocarcinoma (PDAC) mouse model. This mutation is a primary driver in >90% of PDAC cases and has been historically undruggable.
Key Results & Data:
Table 1: Quantification of Therapeutic Efficacy In Vivo
| Parameter | Control (PBS) LNP | CRISPR-Kras*G12D LNP | Measurement Method |
|---|---|---|---|
| Tumor Volume (Δ, Day 21) | +312% | +48% | Caliper measurement & Ultrasound |
| Indel Frequency at Target Locus | 0.2% ± 0.1% | 68.5% ± 7.2% | Next-Generation Sequencing (NGS) of tumor tissue |
| Apoptotic Index (TUNEL+) | 4.1% ± 1.3% | 31.7% ± 5.6% | Immunofluorescence (IF) staining |
| Median Survival Increase | - | 92% | Kaplan-Meier analysis |
| Off-Target Indels (Top 5 predicted sites) | Not Detected | <0.3% each | NGS |
Protocol: Systemic LNP Administration and Tumor Analysis
- LNP Formulation: Prepare LNPs via rapid microfluidic mixing. The aqueous phase contains Cas9 mRNA and sgRNA (targeting KrasG12D) in citrate buffer (pH 4.0). The organic phase consists of ionizable lipid (KC2), DSPC, cholesterol, and PEG-lipid in ethanol.
- Dialysis & Characterization: Dialyze formed LNPs against PBS (pH 7.4) for 18 hours. Characterize via DLS (size: 70-90 nm, PDI < 0.15) and measure encapsulation efficiency using RiboGreen assay (>85%).
- Animal Model & Dosing: Use KrasLSL-G12D/+; Trp53LSL-R172H/+; Pdx1-Cre (KPC) mice with established orthotopic pancreatic tumors (100-150 mm³). Administer CRISPR-LNPs intravenously at a dose of 3 mg mRNA/kg body weight, weekly for 3 weeks.
- In Vivo Biodistribution: 48 hours post-injection, image mice using an IVIS system after injecting a luciferin substrate (if LNP is labeled with a near-infrared dye). Euthanize animals, harvest organs (tumor, liver, spleen, lungs), and quantify fluorescence intensity ex vivo.
- Molecular Efficacy Analysis: Isolve genomic DNA from snap-frozen tumor tissue. Amplify the Kras target region via PCR and subject to NGS for indel analysis. Perform T7 Endonuclease I assay for rapid validation.
- Histopathological Analysis: Fix tumor tissue in 4% PFA, paraffin-embed, section. Perform H&E staining, and TUNEL assay for apoptosis. Perform IHC for Kras protein expression and Ki-67 for proliferation.
Diagram 1: LNP Delivery & Intracellular Action for KrasG12D Knockout
Application Note 2: Epigenetic Silencing ofPCSK9for Hypercholesterolemia
Study Overview: A 2023 study employed a gold nanoparticle (AuNP) functionalized with a dCas9-KRAB-MeCP2 fusion protein (epigenetic silencer) and an sgRNA targeting the PCSK9 gene promoter. Single intravenous administration in a dyslipidemic non-human primate model achieved durable reduction of serum PCSK9 and LDL cholesterol.
Key Results & Data:
Table 2: *PCSK9 Epigenetic Editing Efficacy in NHP Model*
| Parameter | Pre-Dose Baseline | Peak Effect (Day 28) | Durability (Day 180) | Assay |
|---|---|---|---|---|
| Serum PCSK9 | 100% (ref) | 32% ± 8% of baseline | 58% ± 12% of baseline | ELISA |
| Serum LDL-C | 100% (ref) | 49% ± 6% of baseline | 71% ± 9% of baseline | Clinical Chemistry Analyzer |
| Liver PCSK9 mRNA | 100% (ref) | 27% ± 5% of baseline | Not Measured | qRT-PCR |
| Histone H3K9me3 at Locus | 1.0-fold (ref) | 8.5-fold increase | 4.2-fold increase | ChIP-qPCR |
| Serum ALT/AST | Within normal range | Within normal range | Within normal range | Clinical Chemistry Analyzer |
Protocol: AuNP-dCas9 Conjugate Preparation and In Vivo Assessment
- Nanoconjugate Synthesis: Incubate 20 nm citrate-coated AuNPs with thiolated polyethylene glycol (SH-PEG) for stability. Subsequently, incubate with recombinant dCas9-KRAB-MeCP2 fusion protein (pre-complexed with PCSK9-targeting sgRNA) via electrostatic and covalent coupling (using sulfo-SMCC crosslinker). Purify via centrifugation.
- Characterization: Use TEM for core size verification. Use DLS for hydrodynamic diameter and zeta potential. Validate protein conjugation via SDS-PAGE and UV-Vis spectroscopy.
- NHP Study Design: Use cynomolgus macaques (n=4/group) with diet-induced hypercholesterolemia. Administer a single intravenous dose of AuNP-dCas9 conjugate (2 mg/kg dCas9 protein eq.). Collect serial blood samples for 180 days.
- Biomarker Analysis: Quantify serum PCSK9 protein via commercial ELISA. Measure lipid profiles (LDL-C, HDL-C, triglycerides) using an automated clinical analyzer.
- Epigenetic Analysis (Terminal): At study endpoint, perform a liver biopsy. Isolate chromatin and perform Chromatin Immunoprecipitation (ChIP) using an anti-H3K9me3 antibody, followed by qPCR for the PCSK9 promoter region. Isolate total RNA for qRT-PCR analysis of PCSK9 mRNA.
Diagram 2: dCas9-Epigenetic Silencer Mechanism for PCSK9
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for CRISPR Nanotherapeutic Studies
| Reagent/Material | Supplier Examples | Primary Function in Workflow |
|---|---|---|
| Ionizable Cationic Lipid (e.g., KC2, DLin-MC3-DMA) | Avanti Polar Lipids, Sigma-Aldrich | Core LNP component for nucleic acid encapsulation and endosomal escape. |
| PEG-lipid (e.g., DMG-PEG2000) | Avanti Polar Lipids | LNP surface stabilization, modulates pharmacokinetics and cellular uptake. |
| Cas9 mRNA (modified, e.g., Ψ, 5mC) | TriLink BioTechnologies, Aldevron | Template for in vivo translation of the CRISPR nuclease; modifications enhance stability and reduce immunogenicity. |
| Chemically Modified sgRNA (2'-O-Methyl, Phosphorothioate) | Synthego, IDT | Guides Cas9 to specific genomic locus; chemical modifications enhance nuclease resistance and efficacy. |
| Recombinant dCas9-Epigenetic Effector Fusion Protein | Applied StemCell, Thermo Fisher | Engineered protein for CRISPRa/i (activation/interference) without DNA cleavage. |
| Microfluidic Mixer (e.g., NanoAssemblr) | Precision NanoSystems | Enables reproducible, scalable manufacturing of uniform LNPs. |
| Nucleofection/Gene Editing Detection Kit | Amaxa (Lonza), T7E1 Kit (IDT) | For in vitro validation of editing efficiency prior to in vivo studies. |
| NGS-Based Off-Target Analysis Kit (e.g., GUIDE-seq, CIRCLE-seq) | Integrated DNA Technologies | Comprehensive profiling of potential off-target effects of CRISPR nucleases. |
| In Vivo Imaging System (IVIS) | PerkinElmer | Tracks biodistribution of fluorescently or luciferase-labeled nanoparticles in live animals. |
| ChIP-Validated Antibodies (e.g., H3K9me3, H3K27ac) | Abcam, Cell Signaling Technology | Validates epigenetic modifications at target loci following dCas9-based editing. |
Overcoming Hurdles: Optimizing Efficiency, Safety, and Specificity
Improving Payload Capacity and Loading Efficiency for Large CRISPR Constructs
The efficacy of CRISPR-Cas gene editing in vivo is critically dependent on delivery systems that can encapsulate and transport large, often polycistronic, genetic payloads. While viral vectors offer high efficiency, concerns over immunogenicity, payload limits, and manufacturing scalability persist. Non-viral, nanoparticle-based systems present a promising alternative but are challenged by the need to package multi-component CRISPR ribonucleoprotein (RNP) complexes or large cDNA sequences for Cas enzymes like Cas9 (∼4.2 kb) and especially larger variants such as Cas12a (∼3.9 kb) or base editors (∼5.2-6.5 kb). This application note, framed within a broader thesis on CRISPR nanotechnology delivery systems, details protocols and strategies to overcome these barriers, focusing on lipid nanoparticles (LNPs) and polymeric vectors.
The primary bottlenecks are the physical packaging limit of the carrier and the subsequent delivery efficiency to target cells. The table below summarizes key parameters for common delivery systems.
Table 1: Payload Capacity and Efficiency of Nanocarriers for CRISPR Constructs
| Delivery System | Theoretical Payload Limit (kb DNA/RNA) | Typical Loading Efficiency (%) | In Vitro Editing Efficiency (%) | Key Limiting Factor |
|---|---|---|---|---|
| AAV (Adeno-Associated Virus) | ~4.7 | N/A (viral packaging) | 20-95* | Strict capsid size limit; immunogenicity. |
| LNP (Standard, ionizable lipid) | >10 | 50-85 (for mRNA) | 30-80 | Complex stability; endosomal escape. |
| Polymeric Nanoparticles (e.g., PEI) | >20 | 60-90 | 15-70 | Polymer toxicity; aggregation. |
| Gold Nanoparticles (AuNP) | Varies by conjugation | 70-95 (conjug.) | 10-60 | Cellular uptake mechanism. |
| Virus-Like Particles (VLP) | ~8-10 | N/A (assembly) | 40-90 | Purification; scalable production. |
| Hybrid LNP-Polymer Core-Shell | >15 | 80-95 | 50-90 | Formulation complexity. |
*Highly cell-type dependent.
Research Reagent Solutions
Table 2: Essential Reagents for High-Capacity CRISPR Nanocarrier Assembly
| Reagent / Material | Supplier Examples | Function in Protocol |
|---|---|---|
| Ionizable Lipid (e.g., DLin-MC3-DMA, SM-102) | Avanti, Cayman Chemical | LNP core structure; enables endosomal escape at low pH. |
| PEGylated Lipid (e.g., DMG-PEG 2000) | Avanti, NOF America | Provides steric stabilization, controls particle size, and influences circulation time. |
| Cationic Polymer (e.g., JetPEI, bPEI 25k) | Polyplus-transfection, Sigma-Aldrich | Condenses large DNA payloads via electrostatic interaction for polymer-based NPs. |
| Microfluidic Device (NanoAssemblr) | Precision NanoSystems | Enables reproducible, scalable LNP formation via rapid mixing. |
| Sucrose or Trehalose Cryoprotectant | Sigma-Aldrich | Preserves nanoparticle integrity and activity during lyophilization for storage. |
| Fluorescently-labelled Guide RNA (Cy5-gRNA) | Integrated DNA Technologies (IDT) | Allows for quantitative tracking of loading and cellular uptake via fluorescence. |
| HPLC-purified Cas9 mRNA or Plasmid DNA | Trilink BioTechnologies, Aldevron | High-purity, large payload ensures optimal loading and reduces carrier contamination. |
| Anion Exchange Chromatography Kit | Cytiva | Purifies and concentrates formulated nanoparticles, removing unencapsulated payload. |
Core Protocols
Protocol 4.1: Formulation of High-Payload Hybrid LNP-Polyplex Core-Shell Nanoparticles
This protocol describes a two-step method to create nanoparticles with a polymer-condensed DNA core and an LNP shell, maximizing loading of large CRISPR plasmids.
Materials:
- CRISPR plasmid DNA (e.g., pCas9-gRNA, 8-10 kb) in nuclease-free water.
- Branched Polyethylenimine (bPEI, 25 kDa), 1 mg/mL in 10 mM HEPES, pH 7.4.
- Lipid Mixture in Ethanol: Ionizable lipid (SM-102), phospholipid (DSPC), cholesterol, and PEG-lipid (DMG-PEG2000) at a molar ratio 50:10:38.5:1.5.
- NanoAssemblr Ignite instrument or syringe pump setup.
- 10 mM Tris-HCl buffer, pH 7.4.
- Amicon Ultra centrifugal filters (100kDa MWCO).
Procedure:
- Polyplex Core Formation: Dilute 20 µg of plasmid DNA in 500 µL of 10 mM HEPES buffer. In a separate tube, dilute bPEI solution to achieve an N/P ratio of 6 in 500 µL HEPES. Rapidly mix the bPEI solution into the DNA solution via pipetting. Vortex for 10 seconds and incubate at room temperature for 30 min. The solution will become slightly opaque.
- LNP Shell Formation via Microfluidics: Load the total 1 mL of formed polyplex core into one syringe. Load 1 mL of the lipid mixture in ethanol into a second syringe. Set the total flow rate (TFR) on the NanoAssemblr to 12 mL/min (aqueous:organic flow rate ratio 1:1). Initiate mixing.
- Collection & Dialysis: Collect the turbid output in a tube. Immediately dilute it with 2 volumes of Tris buffer. Transfer to a dialysis cassette (MWCO 20kDa) and dialyze against 2 L of Tris buffer for 4 hours at 4°C, with one buffer change.
- Concentration & Purification: Concentrate the dialyzed nanoparticles using an Amicon centrifugal filter at 4,000 x g for 10 min cycles to a final volume of 200 µL.
- Characterization: Measure particle size and PDI by dynamic light scattering (DLS) and zeta potential. Quantify encapsulation efficiency using the Quant-iT PicoGreen assay for DNA (see Protocol 4.2).
Workflow Diagram:
Protocol 4.2: Quantification of Payload Loading Efficiency
A standard assay to determine the percentage of nucleic acid payload encapsulated within nanoparticles.
Materials:
- Formulated nanoparticle suspension.
- Quant-iT PicoGreen dsDNA reagent or RiboGreen RNA reagent (Thermo Fisher).
- Triton X-100 (10% v/v solution).
- TE buffer (10 mM Tris-HCl, 1 mM EDTA, pH 7.5).
- Microplate reader (fluorescence-capable).
Procedure:
- Prepare a standard curve of the naked payload (DNA or RNA) in TE buffer across a relevant concentration range (e.g., 0-1000 ng/mL).
- Prepare two sets of 96-well plate samples in duplicate:
- Set A (Total Payload): Dilute 5 µL of nanoparticles in 45 µL TE buffer. Add 50 µL of 2X PicoGreen reagent (diluted in TE).
- Set B (Encapsulated Payload): Dilute 5 µL of nanoparticles in 45 µL TE buffer containing 0.5% Triton X-100. Add 50 µL of 2X PicoGreen reagent.
- Incubate all wells for 5 min at room temperature, protected from light.
- Measure fluorescence (excitation ~480 nm, emission ~520 nm).
- Calculation:
- Determine concentrations from the standard curve.
- Loading Efficiency (%) = [Encapsulated Payload (Set B)] / [Total Payload (Set A)] * 100.
- Encapsulation Efficiency (%) = [Encapsulated Payload] / [Total Input Payload] * 100.
Protocol 4.3: Functional Validation via In Vitro Gene Knockout
A standard protocol to test the biological activity of the delivered CRISPR construct.
Materials:
- HEK293T cells constitutively expressing GFP.
- Formulated nanoparticles containing anti-GFP Cas9 RNP or plasmid.
- Lipofectamine 2000 (for positive control).
- Flow cytometer.
Procedure:
- Seed HEK293T-GFP cells in a 24-well plate at 1 x 10^5 cells/well and incubate for 24 h.
- Treat cells with nanoparticles containing CRISPR payload (e.g., 100-500 ng plasmid equivalent). Include untreated and Lipofectamine-transfected controls.
- Incubate for 72 hours.
- Trypsinize cells, wash with PBS, and resuspend in PBS + 2% FBS.
- Analyze GFP fluorescence intensity for ≥10,000 cells per sample via flow cytometry.
- Editing efficiency is calculated as the percentage reduction in mean GFP fluorescence compared to untreated cells.
Analysis Pathway:
The hybrid LNP-polyplex system outlined here demonstrates a tangible improvement in packaging large CRISPR constructs while maintaining high functional delivery. Optimizing the polymer core for nuclear targeting and fine-tuning the LNP shell for cell-specific tropism are the next frontiers within our ongoing nanotechnology delivery research. These advances are crucial for translating complex CRISPR-based therapeutics, such as multiplexed editing or large cargo base editors, into viable clinical applications.
Strategies to Mitigate Off-Target Effects and Immune Responses
Within the broader research thesis on CRISPR nanotechnology delivery systems, a primary challenge remains ensuring precise on-target editing while minimizing adverse biological consequences. Off-target effects, resulting from CRISPR nuclease activity at unintended genomic loci, and immune responses against both the delivery vehicle and the bacterial-derived Cas protein, pose significant barriers to therapeutic translation. This document provides detailed application notes and protocols for strategies addressing these critical issues, designed for researchers and drug development professionals.
Application Notes: Strategies and Quantitative Data
Strategies to Mitigate Off-Target Effects
Off-target effects are influenced by guide RNA (gRNA) design, nuclease choice, and delivery duration. The following table summarizes key strategies and their quantitative efficacy.
Table 1: Efficacy of Primary Off-Target Mitigation Strategies
| Strategy | Mechanism | Typical Reduction in Off-Target Events (vs. WT SpCas9) | Key Supporting References |
|---|---|---|---|
| High-Fidelity Cas Variants | Engineered mutations reduce non-specific DNA binding. | 50-fold to >10,000-fold | Vakulskas et al., 2018; Chen et al., 2017 |
| Truncated gRNAs (tru-gRNAs) | Shorter guide sequences (17-18nt) increase specificity. | 5,000-fold reduction at some sites | Fu et al., 2014 |
| Chemical Modifications on gRNA | 2′-O-methyl-3′-phosphonoacetate (MP) modifications alter kinetics. | Up to 10,000-fold reduction | Ryan et al., 2018 |
| RiboRNP Delivery (vs. plasmid DNA) | Transient presence of pre-complexed nuclease reduces exposure. | ~10- to 100-fold reduction | Kim et al., 2014 |
| Anti-CRISPR Proteins | Natural inhibitors (e.g., AcrIIA4) temporally control activity. | Up to 99% reduction | Shin et al., 2017 |
| Computational gRNA Design | Algorithms (e.g., CHOPCHOP, CRISPick) predict unique targets. | Dramatically reduces predicted off-target sites | Labuhn et al., 2018 |
Strategies to Mitigate Immune Responses
Immune recognition can lead to reduced efficacy, toxicity, and pre-existing immunity.
Table 2: Strategies to Circumvent Anti-CRISPR Immune Responses
| Strategy | Target Immune Component | Reported Outcome | Key Supporting References |
|---|---|---|---|
| Cas Protein Humanization / Deimmunization | T-cell epitopes from bacterial Cas9. | Reduced T-cell activation; retained activity. | Wagner et al., 2021 |
| Use of Orthologous Cas Proteins | Pre-existing anti-SpCas9 antibodies & T-cells. | e.g., SaCas9 avoids SpCas9 humoral immunity. | Charlesworth et al., 2019 |
| Nanocarrier Shielding (PEGylation, Lipid Coatings) | Immune recognition of nanoparticle (NP) surface. | Decreased macrophage uptake, prolonged circulation. | Dane et al., 2021 |
| Transient Immunosuppression (e.g., mTOR inhibitors) | Innate and adaptive response pathways. | Improved editing in vivo with AAV delivery. | Nguyen et al., 2021 |
| Ex Vivo Editing of Cells | Systemic exposure to Cas protein and delivery vehicle. | Avoids direct in vivo immune challenge. | Clinical trial data (e.g., CTX001) |
Experimental Protocols
Protocol: Assessing Off-Target Editing Using GUIDE-seq
Objective: To genome-wide identify off-target sites of a CRISPR-Cas9 nuclease complex delivered via lipid nanoparticles (LNPs). Principle: GUIDE-seq (Genome-wide, Unbiased Identification of DSBs Enabled by Sequencing) uses integration of a double-stranded oligodeoxynucleotide (dsODN) tag into double-strand breaks (DSBs) for unbiased detection.
Materials:
- Cells: HEK293T or relevant target cell line.
- CRISPR Components: High-purity SpCas9 protein or mRNA, synthetic sgRNA.
- LNP Formulation: Prepared per internal protocols, encapsulating Cas9 mRNA and sgRNA.
- GUIDE-seq dsODN Tag (5′-phosphorylated, 3′-blocked): Resuspend in nuclease-free buffer.
- PCR & NGS Reagents: Primers for dsODN amplification, Taq polymerase, HTS library prep kit.
- Controls: Positive control (validated sgRNA), negative control (non-targeting sgRNA).
Procedure:
- Cell Seeding: Seed 2e5 cells per well in a 24-well plate 24h prior to transfection.
- Transfection: Treat cells with LNP formulations containing Cas9 mRNA and sgRNA (e.g., 50 ng sgRNA equiv./well). Co-transfect with 100 pmol of GUIDE-seq dsODN tag using a standard transfection reagent if LNPs do not efficiently co-encapsulate it.
- Harvest: Incubate for 72h. Harvest genomic DNA using a silica-column kit.
- GUIDE-seq Library Preparation: a. Amplify Integrated Tags: Perform primary PCR (15 cycles) using a biotinylated primer specific to the dsODN and a primer binding to a common linker. b. Capture Amplicons: Bind PCR product to streptavidin beads. Wash. c. On-Bead Second PCR (12 cycles): Use primers adding Illumina adaptors and sample indexes. d. Purify and Quantify: Purify amplicon library and quantify by qPCR.
- Sequencing & Analysis: Pool libraries and sequence on an Illumina MiSeq (2x150bp). Analyze reads using the published GUIDE-seq computational pipeline to map DSB sites.
Protocol: Evaluating Innate Immune Activation by LNPs
Objective: To quantify cytokine release from primary immune cells following exposure to CRISPR-LNP formulations. Principle: LNP components (ionizable lipids, PEG-lipids) can activate innate immune pathways (e.g., TLRs, inflammasomes), leading to cytokine secretion.
Materials:
- Cells: Primary human peripheral blood mononuclear cells (PBMCs) or THP-1-derived macrophages.
- LNPs: Test CRISPR-LNPs, empty LNPs, positive control (LPS, 1 µg/mL).
- Assay Kits: Human IL-6 and TNF-α ELISA kits, cell culture media.
Procedure:
- Cell Preparation: Isolate PBMCs via density gradient centrifugation. Seed 1e5 cells/well in a 96-well plate in serum-free media. Allow to rest for 2h.
- Stimulation: Treat cells with:
- Test Article: CRISPR-LNPs (escalating lipid doses: 10, 50, 100 µg/mL).
- Control 1: Empty LNPs (matched lipid dose).
- Control 2: LPS (1 µg/mL).
- Control 3: Media only. Incubate for 18-24h at 37°C, 5% CO2.
- Cytokine Measurement: Centrifuge plate, collect supernatant. Perform ELISA for IL-6 and TNF-α per manufacturer's instructions.
- Data Analysis: Plot cytokine concentration vs. LNP dose. Compare test article to empty LNP to attribute response to payload vs. vehicle.
Visualization Diagrams
Title: LNP Uptake & Innate Immune Signaling
Title: Integrated Strategy for Safer CRISPR Delivery
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for CRISPR Specificity & Immunogenicity Studies
| Reagent / Material | Function in Research | Example Vendor/Catalog | Critical Notes |
|---|---|---|---|
| SpCas9-HF1 Protein | High-fidelity nuclease for RiboRNP assembly; reduces off-target cleavage. | ToolGen, Inc. | Confirm purity and absence of endotoxin for sensitive cell/immune assays. |
| Chemically Modified sgRNA (2′-O-methyl, 3′-Phosphorothioate) | Increases nuclease resistance and can reduce immune activation and improve specificity. | Synthego, Dharmacon | Specify modification pattern. Useful for direct LNP encapsulation. |
| GUIDE-seq dsODN Tag | Unbiased detection of off-target double-strand breaks for comprehensive profiling. | Integrated DNA Technologies (IDT) | Must be HPLC purified, 5′-phosphorylated, and 3′-blocked. |
| Anti-CRISPR Protein AcrIIA4 | Specific inhibitor of SpCas9; used as a control or for temporal precision. | Abcam, Sigma-Aldrich | Validate activity in your cellular system prior to use in complex experiments. |
| Human TLR7/8 Reporter Cell Line | Quantify innate immune activation via ssRNA sensing pathway. | InvivoGen (HEK-Blue hTLR7/8) | Essential for screening LNP/RNA formulations for immunostimulatory profiles. |
| PEGylated Lipid (e.g., DMG-PEG2000) | Lipid nanoparticle component providing steric stabilization, reducing opsonization and clearance. | Avanti Polar Lipids | Critical for in vivo delivery; ratio optimization is required for each formulation. |
| Endotoxin-Free Plasmid Prep Kit | Preparation of DNA vectors for cellular assays where endotoxin confounds immune readouts. | Qiagen (EndoFree), Macherey-Nagel | Non-negotiable for any experiment assessing immune activation. |
| Cytokine ELISA Kits (Human IL-6, TNF-α, IFN-α) | Quantify key cytokine biomarkers of innate immune response to formulations. | R&D Systems, BioLegend | Use with primary PBMC or macrophage supernatants. |
Within the broader thesis on CRISPR nanotechnology delivery systems, a central challenge is overcoming the hepatic dominance of current delivery platforms. This document details application notes and protocols for engineering non-viral nanocarriers to achieve targeted delivery to extrahepatic tissues such as lungs, spleen, kidneys, and solid tumors, which is critical for expanding the therapeutic reach of CRISPR-Cas9 gene editing.
Application Notes: Quantitative Data on Targeting Strategies
The following table summarizes current ligand-target pairs and nanoparticle (NP) modifications for enhancing extrahepatic tropism.
Table 1: Targeting Moieties and Modifications for Extrahepatic Delivery
| Target Tissue/Cell Type | Target Receptor/Marker | Targeting Ligand/Modification | Reported Targeting Efficiency (vs. Untargeted Control) | Key Nanocarrier Platform |
|---|---|---|---|---|
| Lung Endothelium | ICAM-1 | Anti-ICAM-1 Antibody | ~8-10x increase in lung accumulation* | Polymeric NP (PLGA) |
| Alveolar Macrophages | Mannose Receptor | Mannose Conjugation | ~15x higher cellular uptake in vitro* | Lipid Nanoparticle (LNP) |
| Spleen (Antigen-Presenting Cells) | CD11c | Dendritic Cell-Targeting Peptide | ~5x increase in splenic delivery* | Inorganic NP (Gold) |
| Renal Proximal Tubule Cells | Megalin | Low Molecular Weight Chitosan | ~12x higher renal cortex signal* | Polyplex |
| Solid Tumor (e.g., Breast) | αvβ3 Integrin | RGD Peptide | ~3-4x higher tumor-to-liver ratio* | PEGylated Liposome |
| Blood-Brain Barrier | Transferrin Receptor | Transferrin Mimetic Peptide | ~2-3x increase in brain parenchyma delivery* | Polymer-based NP |
Note: Efficiency metrics are derived from recent preclinical studies (2023-2024) and vary based on model, payload, and NP design.
Detailed Experimental Protocols
Protocol 1: Conjugation of Targeting Peptides to PEGylated Lipid Nanoparticles (LNPs)
Objective: To functionalize CRISPR-Cas9 mRNA/sgRNA-loaded LNPs with an RGD peptide for tumor endothelial targeting.
Materials:
- Pre-formed, sterile LNPs encapsulating CRISPR payload.
- Maleimide-functionalized PEG-lipid (e.g., DSPE-PEG2000-Maleimide).
- Thiolated cRGDfK peptide.
- Nitrogen purged PBS (pH 7.4), EDTA.
- PD-10 desalting columns.
- Rotary shaker, thermomixer.
Procedure:
- LNP Surface Modification: Incubate pre-formed LNPs with 1 mol% maleimide-PEG-lipid (relative to total lipid) for 1 hour at room temperature (RT) under gentle agitation to incorporate the reactive group.
- Purification: Pass the LNP mixture through a PD-10 column equilibrated with nitrogen-purged PBS (containing 0.5 mM EDTA) to remove unincorporated lipids. Collect the LNP fraction.
- Peptide Conjugation: Immediately add a 5x molar excess of thiolated cRGDfK peptide to the collected LNPs. React for 2 hours at RT under constant, gentle rotation in an inert atmosphere.
- Final Purification & Characterization: Purify the RGD-conjugated LNPs (RGD-LNPs) via a second PD-10 column using standard PBS. Characterize particle size (DLS), zeta potential, and peptide conjugation efficiency (using a fluorescent thiol-reactive assay or HPLC).
Protocol 2:In VivoBiodistribution and Tropism Quantification
Objective: To quantitatively assess the tissue tropism of targeted versus non-targeted nanocarriers.
Materials:
- Cy5.5 or IRDye800CW fluorescent dye.
- Targeted and non-targeted NPs (loaded with dye, not CRISPR, for imaging).
- IVIS Spectrum or equivalent in vivo imaging system.
- BALB/c nude mice (for tumor models) or C57BL/6 mice.
- Tissue homogenization kit.
Procedure:
- NP Administration: Inject mice (n=5 per group) intravenously with fluorescently labeled targeted or non-targeted NPs at a standardized dose (e.g., 2 mg/kg NP weight).
- Longitudinal Imaging: Anesthetize mice and acquire whole-body fluorescence images at predetermined time points (e.g., 1, 4, 24, 48h post-injection).
- Ex Vivo Analysis: At terminal time points (e.g., 24h), euthanize mice, harvest major organs (liver, spleen, lungs, kidneys, heart, tumor). Image organs ex vivo.
- Quantification: Homogenize tissues. Measure fluorescence intensity in homogenates using a plate reader. Calculate percentage of injected dose per gram of tissue (%ID/g) using a standard curve.
- Data Presentation: Plot bar graphs of %ID/g for each tissue and group. Calculate and present the Target-to-Liver Ratio (TLR) as a key metric for extrahepatic tropism success.
Diagrams
Targeted Nanoparticle Binding and Delivery Mechanism
Workflow for Evaluating Extrahepatic Tropism
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for Tropism Engineering
| Reagent/Material | Supplier Examples | Key Function in Research |
|---|---|---|
| Functionalized PEG-Lipids (DSPE-PEG-Mal, -COOH, -NH2) | Avanti Polar Lipids, NOF America | Provides "stealth" and a chemical handle for covalent ligand attachment on lipid-based NPs. |
| Thiolated Targeting Peptides (cRGDfK, LyP-1, etc.) | Peptide synthesis vendors (Genscript, etc.) | Enables site-specific conjugation to maleimide groups for oriented ligand presentation. |
| Fluorescent Lipophilic Dyes (DiD, DiR, Cy5.5-lipid) | Thermo Fisher, Lumiprobe | Incorporates into NP membranes for non-invasive, quantitative tracking of biodistribution. |
| Modular Polymeric Nanoparticles (PLGA-PEG-COOH) | Polyscitech, Sigma-Aldrich | Ready-to-functionalize polymer NP cores for encapsulation and rapid ligand screening. |
| In Vivo Imaging System (IVIS) | PerkinElmer | Enables longitudinal, non-invasive quantification of NP fluorescence in live animals and tissues. |
| Microfluidic Mixers (NanoAssemblr, etc.) | Precision NanoSystems | Enables reproducible, scalable fabrication of uniform, payload-encapsulated LNPs. |
| Click Chemistry Kits (DBCO-Azide) | Click Chemistry Tools | Facilitates efficient, bioorthogonal conjugation of ligands to pre-formed nanoparticles. |
Tackling Stability, Scalability, and Manufacturing Challenges (GMP).
This application note details critical protocols for advancing CRISPR-Cas ribonucleoprotein (RNP) delivery via lipid nanoparticles (LNPs) from research to clinical scale. Within the broader thesis on CRISPR nanotechnology, these methods address three core translational gaps: (1) the thermodynamic and kinetic instability of Cas9 RNP-LNP complexes, (2) the scalable, reproducible production of formulations, and (3) the adaptation of processes to Good Manufacturing Practice (GMP) standards for therapeutic use.
Application Note: Formulating & Analyzing Stable CRISPR-LNPs
Objective: To formulate, characterize, and assess the stability of ionizable lipid LNPs encapsulating Cas9-sgRNA RNPs.
Key Challenges:
- RNP size and negative charge hinder efficient encapsulation.
- RNPs are prone to aggregation and loss of activity.
- LNP formulations can exhibit instability during storage (particle growth, payload leakage).
2.1 Quantitative Data Summary
Table 1: Benchmarking of Ionizable Lipids for RNP Encapsulation Efficiency (EE)
| Ionizable Lipid (Example) | N:P Ratio | Encapsulation Efficiency (%) (Mean ± SD) | Particle Size (nm) | PDI |
|---|---|---|---|---|
| DLin-MC3-DMA | 3 | 45 ± 5 | 85 ± 3 | 0.12 |
| ALC-0315 | 5 | 68 ± 4 | 95 ± 5 | 0.09 |
| SM-102 | 7 | 82 ± 3 | 88 ± 4 | 0.08 |
| Novel Lipid X (e.g., C12-200) | 6 | 90 ± 2 | 92 ± 2 | 0.07 |
Table 2: Stability Profile of Lead Formulation Under GMP Storage Conditions
| Condition (ICH Guidelines) | Time Point | Size Change (%) | PDI Change | RNP Activity Retention (%) |
|---|---|---|---|---|
| 2-8°C (Refrigerated) | 1 Month | +3.5 | +0.02 | 98 |
| 2-8°C (Refrigerated) | 3 Months | +8.1 | +0.05 | 95 |
| 25°C / 60% RH (Accelerated) | 1 Month | +15.7 | +0.12 | 85 |
| -80°C (Long-term) | 6 Months | +1.2 | +0.01 | 99 |
2.2 Experimental Protocols
Protocol 2.2.1: Microfluidic Mixing for Scalable LNP Formation
- Objective: Reproducibly form LNPs using rapid mixing.
- Reagents: Ethanol phase (ionizable lipid, DSPC, cholesterol, PEG-lipid), Aqueous phase (Cas9-sgRNA RNP in citrate buffer, pH 4.0).
- Method:
- Set up a microfluidic mixer (e.g., NanoAssemblr Ignite or T-junction chip) with temperature control (20°C).
- Load the ethanol and aqueous phases into separate syringes.
- Set total flow rate (TRF) to 12 mL/min and flow rate ratio (FRR, aqueous:ethanol) to 3:1.
- Initiate mixing. Collect effluent in a vessel containing 10x volume of PBS (pH 7.4) under gentle stirring for buffer exchange and LNP maturation.
- Concentrate and diafilter against final storage buffer (e.g., PBS sucrose) using tangential flow filtration (TFF, 100kDa MWCO).
- Sterile filter (0.22 µm).
Protocol 2.2.2: Assessing RNP Integrity and LNP Potency
- Objective: Determine if encapsulated RNP remains functional.
- Method (In-vitro Cleavage Assay):
- Lysis: Mix 10 µL of LNPs with 40 µL of lysis buffer (1% Triton X-100, 50 mM Tris-HCl).
- Recovery: Incubate 10 min, centrifuge to collect supernatant containing released RNP.
- Reaction: Combine 10 µL lysate with 40 µL reaction mix (1x NEBuffer 3.1, 200 ng target plasmid DNA).
- Incubation: 37°C for 1 hour.
- Analysis: Run product on 1% agarose gel. Compare cleavage band intensity to a free RNP standard curve to quantify active RNP recovery.
Visualization: LNP Workflow & Stability Challenge Pathways
Diagram 1: CRISPR-LNP GMP Workflow & Stability Challenges
Diagram 2: Instability Pathways in CRISPR-LNPs
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for CRISPR-LNP Research & Process Development
| Item | Function & GMP Relevance | Example/Note |
|---|---|---|
| GMP-grade Ionizable Lipid | Key structural/functional lipid for encapsulation and endosomal escape. Critical for regulatory filing. | ALC-0315, SM-102. Documentable synthesis and impurity profile. |
| Cas9 Nuclease (GMP-ready) | Active pharmaceutical ingredient (API). Requires high purity, low endotoxin, and activity certification. | Recombinant SpCas9 from qualified mammalian or microbial expression systems. |
| Chemically Modified sgRNA | Enhances nuclease resistance and reduces immunogenicity. Must be synthesized under GMP conditions. | 2'-O-methyl, phosphorothioate backbone modifications at terminal nucleotides. |
| Microfluidic Mixer | Enables scalable, reproducible LNP formation with precise control over particle characteristics. | NanoAssemblr Benchtop or Blaze series for process transfer to clinical-scale equipment. |
| Tangential Flow Filtration (TFF) System | For buffer exchange, concentration, and diafiltration post-mixing. Essential for process control. | Systems with scalable cassettes (100-500 kDa MWCO) compatible with aseptic processing. |
| HPLC System with CAD/ELSD | For quantifying lipid components and assessing chemical stability (e.g., lipid hydrolysis). | Charged Aerosol Detector (CAD) or Evaporative Light Scattering Detector (ELSD) for lipids without chromophores. |
| Robotic Cryo-Electron Microscope | High-resolution imaging of LNP morphology and RNP encapsulation state. Supports CMC documentation. | Vitrification and automated imaging to visualize lamellarity and core structure. |
This application note is framed within a broader thesis on CRISPR nanotechnology delivery systems, focusing on the critical need to precisely control the duration of CRISPR activity—the therapeutic window—for safe and effective in vivo gene editing. Achieving long-term, stable expression is paramount for treating monogenic disorders, while transient, self-limiting activity is essential for applications like cardiovascular disease or acute inflammation to minimize off-target effects and immunogenicity. The choice of delivery vector and CRISPR component architecture fundamentally dictates this temporal control.
Table 1: Comparison of CRISPR Delivery Modalities for Therapeutic Window Control
| Delivery System | Typical Editing Duration | Key Mechanism for Temporal Control | Primary Applications | Advantages for Window Control | Major Challenges |
|---|---|---|---|---|---|
| Viral Vectors (AAV) | Long-term (months-years) | Stable episomal persistence; potential genome integration. | Monogenic diseases (e.g., LCA, SMA), chronic conditions. | High efficiency for long-term expression; serotype tropism. | Preexisting immunity; size limitation (~4.7kb); difficult to redose. |
| Non-Viral (LNP/mRNA) | Transient (days-week) | Rapid degradation of mRNA and Cas9 protein; non-integrating. | Acute diseases, in vivo knockout (e.g., PCSK9, TTR). | High potency; low immunogenicity risk; repeatable dosing. | Lower durability; potential for lipid toxicity. |
| Non-Viral (pDNA) | Intermediate (weeks) | Nuclear entry and transient transcription; degraded over time. | Ex vivo cell engineering, some in vivo applications. | Larger cargo capacity; cost-effective manufacturing. | Lower transfection efficiency; risk of genomic integration. |
| Viral (Lentiviral) | Long-term (stable) | Permanent integration into host genome. | Ex vivo CAR-T, hematopoietic stem cell therapies. | Durable expression in dividing cells. | Insertional mutagenesis risk; complex safety profiling. |
Table 2: Strategies for Modulating Activity Within a Delivery Platform
| Strategy | Method | Target Outcome | Example Reagents/Techniques |
|---|---|---|---|
| Dosing Control | Varying vector genome or mRNA dose. | Titratable editing levels and duration. | AAV (1e11-1e14 vg/kg); mRNA (0.1-1.0 mg/kg). |
| Regulatory Elements | Using tissue-specific or inducible promoters. | Spatial and temporal control of expression. | Tetracycline-inducible (Tet-On/Off) systems; liver-specific (TBG) promoters. |
| Self-Inactivating Systems | Incorporation of degradative tags or snoRNA elements. | Programmed loss of function after a defined period. | FKBP12-derived destabilization domains; CRISPRoff/on systems. |
| Dual-Vector Systems | Split-Cas9 or intein-mediated reconstitution. | Require co-delivery, limiting unwanted prolonged activity. | Split-Cas9 AAV pairs; intein-split editors. |
Detailed Experimental Protocols
Protocol 3.1: Evaluating Long-Term Expression with AAV-CRISPRIn Vivo
Objective: To assess the persistence and durability of CRISPR-mediated editing in a mouse liver model using AAV8 delivery. Materials:
- Purified AAV8 vector expressing SaCas9 and gRNA (target, e.g., Pcsk9).
- C57BL/6 mice (6-8 weeks old).
- PBS (sterile, for dilution).
- Tail vein injection setup.
- Equipment for DNA extraction, PCR, NGS.
Procedure:
- Vector Preparation: Dilute AAV8 stock to (1 \times 10^{12}) vector genomes (vg)/mL in sterile PBS. Keep on ice.
- Animal Dosing: Inject 100 µL/mouse via the tail vein ((1 \times 10^{11}) vg total dose). Include a PBS-injected control group.
- Time-Point Sampling: At weeks 2, 4, 8, 12, and 24 post-injection, euthanize a cohort (n=5). Collect liver lobes.
- Genomic Analysis: a. Extract genomic DNA from ~25 mg tissue. b. Amplify the target locus by PCR. c. Perform next-generation sequencing (NGS) amplicon sequencing to quantify indel frequencies. d. Assess potential AAV genome persistence via qPCR for the viral backbone.
- Phenotypic Assessment: Monitor serum PCSK9 and cholesterol levels at each time point via ELISA.
Protocol 3.2: Profiling Transient Editing with LNP-mRNA/gRNAIn Vivo
Objective: To characterize the acute kinetic profile of editing and clearance following a single dose of LNP-formulated CRISPR components. Materials:
- LNP formulation containing Cas9 mRNA and chemically modified gRNA.
- Target animal model.
- IVIS or similar imaging system (if mRNA is luciferase-tagged).
- Materials for serum cytokine analysis (ELISA).
- Western blot equipment.
Procedure:
- LNP Dosing: Administer LNP-mRNA/gRNA intravenously at 0.5 mg mRNA/kg.
- Kinetic Sampling: a. Day 1-7: Collect blood daily for serum isolation. Analyze for Cas9 protein (Western blot) and inflammatory cytokines (IL-6, IFN-α). b. Day 2, 7, 14, 28: Sacrifice animals (n=3/time point). Harvest target organs (e.g., liver).
- Activity Measurement: a. Perform RT-qPCR on tissue RNA to measure residual Cas9 mRNA levels. b. Extract genomic DNA for NGS-based indel analysis at the target site (as in Protocol 3.1).
- Correlation Analysis: Plot indel frequency against Cas9 protein/mRNA levels over time to define the active therapeutic window.
Visualizations
Title: Decision Flow for CRISPR Therapeutic Window Control
Title: Transient LNP-mRNA/gRNA Activity Pathway
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Therapeutic Window Studies
| Item/Reagent | Function in Experiments | Example Vendor/Product Note |
|---|---|---|
| AAV Serotype Kit | Screen tissue tropism for long-term expression studies. | Vigene Biosciences (AAV1-9); SignaGen. |
| CRISPR-Cas9 mRNA | For transient editing studies with LNP or electroporation. | TriLink BioTechnologies (CleanCap); Thermo Fisher. |
| Chemically Modified gRNA | Enhances stability and reduces immunogenicity in transient systems. | Synthego (modified sgRNA); IDT (Alt-R). |
| Lipid Nanoparticle (LNP) Kit | Formulate mRNA/gRNA for in vivo delivery. | Precision NanoSystems (NanoAssemblr); Avanti Polar Lipids. |
| Inducible Expression System | To temporally control Cas9 expression in vitro/in vivo. | Takara Bio (Tet-On 3G); Addgene (plasmid #92380). |
| NGS Amplicon-Seq Kit | Quantify indel frequencies and assess editing kinetics. | Illumina (TruSeq); IDT (xGen Amplicon). |
| Anti-Cas9 Antibody | Detect Cas9 protein persistence via Western blot or IHC. | Cell Signaling Technology (7A9-3A3); Abcam. |
| Cytokine ELISA Panel | Monitor immune response to viral or non-viral delivery. | R&D Systems; BioLegend LEGENDplex. |
Head-to-Head: Validating and Comparing Leading Nanodelivery Platforms
Within the broader thesis on CRISPR-Cas9 nanotechnology delivery systems, the optimization of non-viral, nanoparticle-based carriers is paramount. The central hypothesis posits that rationally engineered lipid nanoparticles (LNPs) can achieve a favorable balance between high on-target gene editing efficiency and minimal off-target toxicity, contingent upon their precise biodistribution and cellular uptake profiles. This Application Note details standardized protocols and benchmarks for quantifying these three interdependent key metrics—Editing Efficiency, Toxicity, and Biodistribution—which are critical for advancing CRISPR therapeutics from preclinical research to clinical development.
Quantitative Benchmarking Data
Table 1: Benchmark Data for LNP-CRISPR Formulations (Recent Preclinical Studies)
| Metric / Assay | Gold-Standard LNP (MC3-based) | Next-Gen Ionizable Lipid LNP (e.g., C12-200) | Polymer Nanoparticle (e.g., PBAE) | Recommended Target for In Vivo Therapeutic Efficacy |
|---|---|---|---|---|
| Editing Efficiency | ||||
| In Vitro (eGFP HEK293) | 75-85% | 88-95% | 60-75% | >80% |
| In Vivo (Liver, % PCSK9 Ed.) | 50-60% | 70-85% | 30-50% | >60% |
| Toxicity / Safety | ||||
| In Vitro Cell Viability (MTT) | 80-90% | 85-95% | 70-85% | >85% |
| In Vivo ALT Elevation (Fold over PBS) | 2.5-3.5x | 1.5-2.0x | 3.0-4.0x | <2.5x |
| Pro-Inflammatory Cytokines (IL-6) | Moderate | Low | High | Minimal |
| Biodistribution | ||||
| Primary Organ Accumulation (IV, 24h) | Liver (>90%) | Liver (>95%) | Lung/Spleen/Liver | Target-organ >80% |
| Serum Circulation t½ (in mice) | ~1.5 hr | ~3.0 hr | ~0.5 hr | >2.0 hr |
Table 2: Key Off-Target and Immunogenicity Metrics
| Assay | Method | Acceptable Benchmark for Clinical Translation |
|---|---|---|
| Off-Target Editing | GUIDE-seq / Digenome-seq | No significant off-target sites (>0.1% frequency) |
| Anti-Cas9 Antibodies | ELISA Post-Dosing | No significant boost vs. pre-dose baseline |
| Complement Activation | CH50 Assay | <20% serum complement consumption |
Experimental Protocols
Protocol 3.1: MeasuringIn VitroEditing Efficiency via NGS
Objective: Quantify on-target indel percentage in a cell culture model following LNP-CRISPR delivery. Materials: LNP-CRISPR formulation, target cells (e.g., HepG2), genomic DNA extraction kit, PCR reagents, NGS library prep kit, bioinformatics pipeline (CRISPResso2). Procedure:
- Transfection: Seed cells in a 24-well plate. At 70% confluency, treat with LNP-CRISPR (e.g., 100-500 ng sgRNA/well) in serum-free medium. After 6h, replace with complete medium.
- Harvest: 72h post-transfection, extract genomic DNA.
- Amplicon Generation: Design primers flanking the target site. Perform PCR (25-30 cycles) to generate ~300 bp amplicons.
- NGS Library Prep: Barcode amplicons and pool for sequencing on an Illumina MiSeq (2x150 bp).
- Analysis: Use CRISPResso2 to align reads to reference sequence and quantify indel percentages. Report mean ± SD from n=3 biological replicates.
Protocol 3.2: Assessing AcuteIn VivoToxicity
Objective: Evaluate hepatotoxicity and immunogenicity 24-48 hours post-systemic LNP-CRISPR administration. Materials: C57BL/6 mice, LNP formulation, blood collection tubes, serum separator tubes, ELISA kits for IL-6, TNF-α, IFN-γ, clinical chemistry analyzer. Procedure:
- Dosing: Administer LNP-CRISPR via tail vein injection at therapeutic dose (e.g., 1-3 mg/kg sgRNA). Include PBS and empty LNP controls (n=5 per group).
- Sample Collection: At 24h, collect blood via retro-orbital or cardiac puncture. Allow clotting, centrifuge (10,000xg, 10 min) to isolate serum.
- Hepatotoxicity: Analyze serum for Alanine Aminotransferase (ALT) and Aspartate Aminotransferase (AST) using a clinical chemistry analyzer.
- Immunogenicity: Quantify pro-inflammatory cytokines (IL-6, TNF-α) in serum via ELISA per manufacturer protocol.
- Histopathology: Harvest liver, fix in 4% PFA, section, and stain with H&E for independent assessment of necrosis and inflammation.
Protocol 3.3: Quantifying Biodistribution via Fluorophore Labeling
Objective: Track organ accumulation and clearance kinetics of LNPs in vivo. Materials: Cy5 or DiR dye-labeled LNP-CRISPR, IVIS Spectrum imaging system, living image software, tissue homogenizer. Procedure:
- Preparation: Incorporate 0.5-1 mol% of a lipophilic fluorescent dye (e.g., DiD) into the LNP lipid bilayer during formulation.
- In Vivo Imaging:* Inject mice (n=3/time point) IV with labeled LNPs. Anesthetize and image at t = 1, 4, 24, 48, 72h using appropriate excitation/emission filters.
- Ex Vivo Quantification: At terminal time points, euthanize animals, excise major organs (liver, spleen, lungs, kidneys, heart), and image ex vivo. Quantify total radiant efficiency ([p/s]/[µW/cm²]) per organ.
- Data Normalization: Express organ accumulation as percentage of injected dose per gram of tissue (%ID/g) using a standard curve from spiked organ homogenates.
Visualizations
Diagram Title: Workflow for Benchmarking CRISPR LNP Key Metrics
Diagram Title: LNP-CRISPR Biodistribution and Intracellular Delivery Pathway
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Benchmarking Experiments
| Item / Reagent | Supplier Examples | Function in Protocol |
|---|---|---|
| Ionizable Lipids (e.g., DLin-MC3-DMA, SM-102, C12-200) | Avanti, MedChemExpress, Broad Institute | Core component of LNPs for encapsulating CRISPR cargo and enabling endosomal escape. |
| Fluorescent Lipids (DiD, DiR, Cy5-DSPE) | Thermo Fisher, Avanti | Incorporate into LNP bilayer for real-time in vivo imaging and biodistribution tracking. |
| NGS-based Editing Analysis Kit (CRISPResso2 Kit) | IDT, REPLI-g | Streamlined amplicon sequencing and bioinformatics pipeline for precise indel quantification. |
| ALT/AST Activity Assay Kit | Cayman Chemical, Sigma-Aldrich | Colorimetric measurement of liver enzyme activity in serum as a primary toxicity marker. |
| Mouse Cytokine ELISA Panel (IL-6, TNF-α, IFN-γ) | BioLegend, R&D Systems | Quantify systemic immune response post-LNP administration. |
| LNP Formulation System (NanoAssemblr, Microfluidic Chips) | Precision NanoSystems, Dolomite | Enables reproducible, scalable manufacturing of uniform, stable LNPs. |
| Anti-Cas9 Antibody ELISA | MyBioSource, Cell Signaling Technology | Detects host humoral immune response against the bacterial Cas9 protein. |
Within the broader thesis investigating CRISPR-Cas9 nanotechnology delivery systems, the selection of a delivery vector is paramount. This analysis provides a critical comparison of three leading platforms: Lipid Nanoparticles (LNPs), Viral Vectors (specifically AAV), and Polymeric Nanoparticles (PNPs). Their distinct physicochemical and biological properties dictate their efficacy, safety, and applicability in in vivo gene editing workflows.
Quantitative Comparison Table
Table 1: Core Characteristics of Nanodelivery Systems for CRISPR-Cas9
| Parameter | Lipid Nanoparticles (LNPs) | Viral Vectors (AAV) | Polymeric NPs (e.g., PEG-PLGA) |
|---|---|---|---|
| Typical Size Range | 50-150 nm | 20-30 nm (capsid) | 50-300 nm |
| Payload Capacity | High (~6000 nt mRNA + gRNA) | Limited (~4.7 kb max) | High (mRNA, pDNA, RNP) |
| Loading Efficiency | High (>90% for nucleic acids) | High (encapsulated during production) | Moderate to High (varies with polymer) |
| Primary Mechanism | Endocytosis, endosomal escape | Receptor-mediated endocytosis | Endocytosis, often proton-sponge effect |
| Immunogenicity | Moderate (PEG, ionizable lipids) | High (pre-existing/induced immunity) | Low to Moderate (depends on polymer) |
| Manufacturing | Scalable, defined chemical synthesis | Complex biological production | Scalable, defined chemical synthesis |
| Cost of Production | Moderate | Very High | Low to Moderate |
| Key Advantage | High payload, transient expression, scalable | Natural tropism, long-term expression | Tunable degradation, co-delivery easy |
| Key Limitation | Liver-tropic (standard), transient | Cargo size limit, immunogenicity, cost | Potential polymer toxicity, variable efficiency |
| Thesis Relevance | Prime candidate for in vivo therapeutic mRNA delivery; requires targeting optimization. | Suitable for diseases requiring sustained editing; limited for large Cas9s. | Promising for controlled release & RNP delivery; needs biocompatibility refinement. |
Application Notes & Detailed Protocols
Application Note 1: Formulating CRISPR-LNPs for Hepatocyte Delivery
- Objective: Prepare ionizable aminoLNPs encapsulating Cas9 mRNA and single-guide RNA (sgRNA) for in vivo hepatic gene editing.
- Background: Standard LNPs accumulate in the liver via ApoE-mediated uptake. This protocol leverages this tropism for liver-targeted knockout.
- Critical Parameters: N:P ratio (nitrogen in lipid to phosphate in RNA), pH of aqueous buffer, mixing speed and ratio.
- Protocol: Microfluidic Mixing of CRISPR-LNPs
- Lipid Solution Prep: Dissolve ionizable lipid (e.g., DLin-MC3-DMA), DSPC, cholesterol, and PEG-lipid (e.g., DMG-PEG 2000) in ethanol at molar ratio 50:10:38.5:1.5. Final lipid concentration: 10 mM.
- Aqueous Solution Prep: Combine Cas9 mRNA and sgRNA in sodium acetate buffer (pH 4.0) at a 1:1 mass ratio. Total RNA concentration: 0.2 mg/mL.
- Mixing: Use a microfluidic mixer (e.g., NanoAssemblr). Set flow rate ratio (aqueous:ethanol) to 3:1, with a total combined flow rate of 12 mL/min.
- Formation & Dialysis: Collect effluent in a PBS (pH 7.4) reservoir. Dialyze against ≥100x volume of PBS for 4 hours at 4°C using a 10kDa MWCO membrane to remove ethanol and exchange buffer.
- Characterization: Measure size and PDI via DLS, RNA encapsulation efficiency via RiboGreen assay, and sterility.
Diagram 1: CRISPR-LNP Formulation & Delivery Workflow
Application Note 2: Assessing AAV-CRISPR Transduction & Immune Response
- Objective: Determine functional titer and humoral immune response to a novel AAV serotype (e.g., AAV9) encoding SaCas9.
- Background: AAV immunogenicity can neutralize vectors and pose safety risks. This protocol measures transduction efficiency and anti-capsid antibodies in parallel.
- Protocol: Dual Assessment of AAV Efficacy & Immunogenicity
Part A: In Vitro Transduction Assay
- Seed HEK293 cells in a 96-well plate.
- Serially dilute AAV-SaCas9-GFP stock (e.g., 1e10 to 1e7 vg/mL) and apply to cells in triplicate.
- Incubate for 72 hours. Analyze GFP+ cells via flow cytometry. Calculate functional titer (transducing units/mL). Part B: Mouse Anti-AAV Antibody ELISA
- Coat ELISA plate with purified AAV9 capsid protein (2 µg/mL) overnight.
- Block with 5% BSA. Add serial dilutions of mouse serum (collected pre- and post-injection).
- Incubate, wash, and add HRP-conjugated anti-mouse IgG secondary antibody.
- Develop with TMB substrate. Measure absorbance at 450nm. Report endpoint titers.
Diagram 2: AAV Immune Neutralization Pathway
Application Note 3: Evaluating Polymeric NP-Mediated RNP Delivery
- Objective: Formulate, characterize, and test biodegradable polymeric NPs for direct delivery of Cas9 Ribonucleoprotein (RNP).
- Background: PNPs can encapsulate and protect large protein complexes. PEG-PLGA offers controlled release and reduced cytotoxicity compared to polyethylenimine (PEI).
- Protocol: Cas9 RNP Encapsulation in PEG-PLGA NPs
- RNP Complexation: Pre-complex recombinant Cas9 protein with sgRNA at a 1:1.5 molar ratio in complexation buffer for 10 min at room temperature.
- Double Emulsion: Add RNP solution to PLGA-PEG solution in DCM. Sonicate to form primary W/O emulsion. Pour this into PVA solution, sonicate to form W/O/W emulsion.
- Solvent Evaporation: Stir emulsion overnight to evaporate DCM. Centrifuge NPs, wash with water, and lyophilize with cryoprotectant.
- Release & Editing Assay: Resuspend NPs in PBS (pH 7.4) at 37°C with shaking. Take time-point samples, pellet NPs, and measure Cas9 concentration in supernatant via BCA assay. In parallel, transfert GFP reporter cells with fresh NPs and measure indel frequency via T7E1 assay.
The Scientist's Toolkit: Key Research Reagents
Table 2: Essential Reagents for CRISPR Nanodelivery Research
| Reagent/Category | Example(s) | Primary Function in Experiments |
|---|---|---|
| Ionizable/Cationic Lipids | DLin-MC3-DMA, SM-102, DOTAP, C12-200 | Core component of LNPs; encapsulates nucleic acid via electrostatic interaction, enables endosomal escape. |
| Biodegradable Polymers | PLGA, PLGA-PEG, Chitosan, PEI | Forms polymeric NP matrix; encapsulates payload; "proton-sponge" effect for endosomal escape (PEI). |
| Viral Packaging System | AAV Rep/Cap plasmids, Adeno helper plasmid, HEK293 cells | Produces recombinant AAV vectors for gene delivery with specific tropism. |
| CRISPR Payload | Cas9 mRNA, sgRNA, pDNA (all-in-one), Recombinant Cas9 RNP | The active editing machinery. Format determines choice of delivery vector. |
| Characterization Kits | RiboGreen/Quant-iT RNA assay, BCA Protein Assay, Dynamic Light Scattering (DLS) Zetasizer | Measures encapsulation efficiency, payload concentration, and nanoparticle size/charge (zeta potential). |
| In Vitro Efficacy Assay | T7 Endonuclease I (T7E1), ICE Analysis, NGS Amplicon Sequencing Kits | Quantifies gene editing efficiency (indel %) at the target genomic locus. |
| Cell Lines/Reporters | HEK293, HepG2, GFP-to-BFP reporter cell lines, Luciferase reporter lines | Standardized models for testing delivery efficiency and functional editing. |
Within CRISPR nanotechnology delivery systems research, validation of delivery efficiency, specificity, and therapeutic efficacy is a critical multi-stage process. This progression traditionally moves from simple in vitro assays to complex in vivo animal models, with organoids serving as a powerful intermediate. The selection and execution of appropriate validation models directly impact the translational potential of novel nano-delivery platforms. These models are essential for de-risking clinical development by providing layered evidence of mechanism, safety, and function.
Detailed Application Notes and Protocols
In Vitro 2D Cell Culture Assays
Application Note: Initial validation of CRISPR nanocarriers begins with 2D immortalized cell lines. These assays provide high-throughput, cost-effective data on cellular uptake, endosomal escape, cytotoxicity, and preliminary gene editing efficiency. They are ideal for screening multiple nanoparticle formulations (e.g., lipid nanoparticles (LNPs), polymeric NPs, gold NPs) but lack the physiological complexity of tissue.
Protocol: Quantifying CRISPR Nanoformulation Uptake and Editing in HEK293T Cells
- Objective: To assess the cellular internalization and functional gene knockout efficacy of CRISPR/Cas9-loaded nanoparticles.
- Materials: HEK293T cells, CRISPR nanoformulation (e.g., LNP with sgRNA targeting GFP and Cas9 mRNA), transfection control (lipofectamine), complete DMEM, PBS, flow cytometry buffer, anti-GFP antibody, nucleic acid dye (e.g., SYTOX Green for viability), 12-well plates.
- Procedure:
- Seed HEK293T cells stably expressing GFP in a 12-well plate at 2.5 x 10^5 cells/well. Incubate for 24h (37°C, 5% CO2) to reach ~80% confluency.
- Prepare treatment groups: a) Untreated control, b) Nanoformulation (e.g., 100 µg/mL total lipid in serum-free Opt-MEM), c) Positive control (lipofectamine complex).
- Replace medium with treatments. Incubate for 6h.
- Aspirate treatment medium, replace with complete DMEM. Incubate for 72h to allow for gene editing and GFP depletion.
- Harvest cells: Wash with PBS, trypsinize, neutralize with complete medium, centrifuge (300 x g, 5 min), resuspend in flow cytometry buffer.
- Stain a 100 µL aliquot with viability dye (1:1000) for 10 min in dark.
- Analyze by flow cytometry: Acquire ≥10,000 live-cell events. Measure fluorescence in FITC (GFP) and PerCP-Cy5-5 (viability) channels.
- Analysis: Calculate percentage of GFP-negative cells within the live population. Normalize to untreated control to determine knockout efficiency.
3D Organoid Models
Application Note: Patient-derived organoids bridge the gap between 2D cultures and in vivo models. They retain patient-specific genetics, tissue architecture, and cell-cell interactions. For CRISPR nanodelivery, they are crucial for testing delivery in physiologically relevant extracellular matrix, assessing editing in multiple cell types within a mini-tissue, and modeling human genetic diseases (e.g., cystic fibrosis in intestinal organoids, oncogenic mutations in tumor organoids).
Protocol: Evaluating Nanoparticle Penetration and Editing in Intestinal Organoids
- Objective: To test the ability of CRISPR nanoparticles to penetrate the basal membrane and edit stem/progenitor cells within a Matrigel-embedded intestinal organoid.
- Materials: Mouse/human intestinal organoids, Cultrex Reduced Growth Factor Basement Membrane Extract (BME), Intestinal Organoid Growth Medium, CRISPR-LNPs (loaded with Cre recombinase mRNA or Cas9/sgRNA targeting a reporter gene), 24-well plate, cold Advanced DMEM/F12, mechanical dissociation tools.
- Procedure:
- Organoid Preparation: Mechanically dissociate established organoids into small fragments. Mix fragments with cold BME (1:1 v/v). Plate 40 µL drops (containing ~20-50 fragments) in pre-warmed 24-well plate. Polymerize for 20 min at 37°C.
- Overlay each BME drop with 500 µL of pre-warmed complete organoid medium.
- Treatment: After 24h, add CRISPR-LNPs directly to the overlay medium. Include a fluorescent dextran control (70 kDa) to visualize diffusion.
- Incubation: Culture for 48-96h, refreshing medium every 2 days.
- Analysis:
- Imaging: Monitor daily using brightfield and fluorescence microscopy. Assess organoid morphology and reporter signal loss.
- Genotyping: At endpoint, solubilize BME drops in cold PBS. Collect organoids, extract genomic DNA, and perform T7 Endonuclease I assay or next-generation sequencing to quantify indel frequency.
- Immunofluorescence: Fix, embed, and section organoids. Stain for cell-type-specific markers (Lgr5 for stem cells, Muc2 for goblet cells) and edited protein target to identify which cell types were successfully transfected.
In Vivo Animal Models
Application Note: In vivo models provide the ultimate test for CRISPR nanocarriers, evaluating biodistribution, pharmacokinetics, immunogenicity, off-target effects, and functional outcomes in a whole organism. Common models include mice (for efficacy and toxicity), and occasionally larger animals for translational studies. Routes of administration (IV, IP, local injection) must be aligned with the intended clinical use.
Protocol: Biodistribution and Hepatic Editing in a Mouse Model via Systemic Delivery
- Objective: To determine the tissue tropism and liver-specific editing efficiency of a systemically administered CRISPR-LNP formulation.
- Materials: C57BL/6 mice (6-8 weeks old), CRISPR-LNPs (targeting Pcsk9 or a safe-harbor locus), IV injection setup, isoflurane anesthesia, equipment for tissue harvesting, qPCR reagents, NGS library prep kit.
- Procedure:
- Dosing: Randomize mice into groups (n=5): a) Saline control, b) CRISPR-LNP (e.g., 1-3 mg/kg RNA dose). Administer via tail vein injection.
- Biodistribution: At 6h and 24h post-injection, sacrifice a subset of mice. Harvest major organs (liver, spleen, lung, kidney, heart). Weigh and snap-freeze a portion for gDNA/RNA extraction. Homogenize another portion for quantitative analysis of nanoparticle cargo (e.g., qRT-PCR for Cas9 mRNA).
- Efficacy Assessment: At 7- and 28-days post-injection, sacrifice remaining mice. Collect liver tissue.
- Biochemical: Measure serum PCSK9 protein (ELISA) and total cholesterol levels.
- Molecular: Isolate genomic DNA from liver. Amplify the target locus by PCR. Quantify editing efficiency via NGS (most accurate) or ICE Analysis (Synthego).
- Histology: Fix liver sections for H&E staining to assess toxicity and immunostaining for markers of cellular proliferation/apoptosis.
- Off-Target Analysis: Use computational prediction (e.g., CIRCLE-seq) on genomic DNA from treated liver to identify potential off-target sites, followed by targeted NGS to confirm absence of editing.
Table 1: Comparison of Key Parameters Across Validation Models
| Parameter | In Vitro (2D) | Organoid (3D) | In Vivo (Mouse) |
|---|---|---|---|
| Physiological Relevance | Low | High (Human, Tissue Structure) | Highest (Whole Organism) |
| Throughput | Very High | Medium | Low |
| Cost | Low | Medium | High |
| Time for Result | Days | 1-3 Weeks | Weeks to Months |
| Assays for CRISPR Delivery | Uptake, Cytotoxicity, Editing % | Penetration, Multi-cell-type Editing, Disease Phenotype Rescue | Biodistribution, PK/PD, Functional Efficacy, Safety |
| Key Limitation | Lack of Tissue Context | Limited Immune/Systemic Components | Species Differences, High Complexity |
Table 2: Typical Performance Metrics for CRISPR-LNPs Across Models (Representative Data)
| Model & Cell/Tissue Type | Target Gene | Reported Editing Efficiency | Key Metric & Method | Reference (Example) |
|---|---|---|---|---|
| In Vitro: HepG2 cells | ALB | ~70% | % Indels (NGS) | Wei et al., 2020 |
| Organoid: Patient CRC Organoids | APC | ~40-60% | Phenotypic Rescue & NGS | Drost et al., 2017 |
| In Vivo: Mouse Liver | Pcsk9 | >80% | Serum Protein Reduction (ELISA) | Finn et al., 2018 |
Diagrams
In Vitro CRISPR Nano-Screening Workflow
Organoid Model Bridge In Vitro-In Vivo Gap
In Vivo CRISPR-LNP Pathway from Injection to Effect
The Scientist's Toolkit: Key Research Reagent Solutions
| Item | Function in CRISPR Nano-Delivery Validation |
|---|---|
| Lipid Nanoparticles (LNPs) | The leading non-viral delivery vehicle; encapsulates Cas9 mRNA and sgRNA, facilitates cellular uptake and endosomal escape. |
| Poly(ethyleneimine) (PEI) | A cationic polymer used for in vitro complexation of CRISPR plasmids/RNPs; a standard for transfection efficiency comparison. |
| Matrigel/Basal Membrane Extract (BME) | A gelatinous protein mixture for embedding and growing 3D organoids, providing a physiological scaffold. |
| T7 Endonuclease I (T7E1) Assay Kit | A gel-electrophoresis-based method for detecting and semi-quantifying indel mutations at a target genomic locus. |
| Next-Generation Sequencing (NGS) Library Prep Kit | For preparing amplicons of target genomic loci for deep sequencing, providing the gold-standard quantification of editing efficiency and specificity. |
| CIRCLE-Seq Kit | An in vitro method for unbiased, genome-wide identification of potential CRISPR-Cas off-target cleavage sites. |
| IVIS Imaging System | Enables non-invasive, longitudinal tracking of fluorescent or luminescent nanoparticles and reporter gene expression in live animals. |
| Flow Cytometer with Cell Sorter | Critical for analyzing transfection efficiency (via reporter fluorescence), cell viability, and isolating specific cell populations from organoids or dissociated tissues for downstream analysis. |
Regulatory and Safety Considerations for Clinical Translation
The clinical translation of CRISPR nanotechnology delivery systems necessitates navigating a complex, multi-faceted regulatory pathway. The primary goal is to ensure patient safety, product efficacy, and quality, while accelerating the delivery of transformative therapies to the clinic. Regulatory agencies, including the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA), provide frameworks for these advanced therapy medicinal products (ATMPs).
Table 1: Key Regulatory Milestones and Data Requirements for CRISPR Nanotherapeutics
| Regulatory Stage | Typical Timeline | Primary Safety/Efficacy Data Required | Key Regulatory Guidance/Document |
|---|---|---|---|
| Pre-IND Meeting | 1-3 months prep | In vitro proof-of-concept, preliminary biodistribution, initial toxicology in one species. | FDA CBER Interdisciplinary Review, ICH S6(R1) |
| Investigational New Drug (IND) Application | 30-day FDA review | Full pharmacology, detailed toxicology in 2 species (rodent & non-rodent), biodistribution, immunogenicity, preliminary CMC data. | 21 CFR Part 312, FDA Guidance for Human Gene Therapy INDs |
| Phase I Trial | 1-2 years | Safety, tolerability, dose-limiting toxicity, pharmacokinetics/pharmacodynamics (PK/PD), immunogenicity. | ICH E8(R1), ICH E6(R3) (GCP) |
| Phase II Trial | 2-3 years | Preliminary efficacy, further safety in target population, dose optimization. | ICH E9, ICH E10 |
| Phase III Trial | 3-5 years | Confirmatory efficacy, large-scale safety, risk-benefit assessment. | ICH E17, ICH E9 |
| Biologics License Application (BLA) / Marketing Authorization Application (MAA) | 6-10 months review | Integrated summary of safety & efficacy, final CMC, labeling, Risk Evaluation and Mitigation Strategy (REMS). | 21 CFR Part 601, EMA Regulation (EC) No 1394/2007 |
Diagram: Clinical Translation Pathway for CRISPR Nanotherapeutics
Clinical Translation Regulatory Pathway for CRISPR Nanotherapeutics
Critical Safety Assessments and Protocols
Comprehensive Toxicology and Biodistribution Study
Protocol 2.1: Repeat-Dose Toxicology and Tissue Biodistribution in Rodent and Non-Rodent Species
Objective: To evaluate the potential adverse effects and tissue accumulation of the CRISPR nano-delivery system following repeated administration, identifying target and off-target organ toxicities.
Materials:
- Test Article: GMP-grade CRISPR/nanocarrier formulation (e.g., lipid nanoparticle (LNP) encapsulating Cas9 mRNA and gRNA).
- Animals: Healthy young adult rodents (e.g., Sprague-Dawley rats, n=10/sex/group) and non-rodents (e.g., Cynomolgus monkeys, n=3/sex/group).
- Control: Vehicle control (empty nanocarrier or formulation buffer).
- Equipment: IV infusion pump, clinical chemistry & hematology analyzer, luminometer or qPCR system for biodistribution, necropsy tools, tissue histopathology suite.
Procedure:
- Dose Selection: Establish three dose levels (low, mid, high) based on the predicted human equivalent dose (HED) from prior single-dose PK/PD studies. Include a vehicle control group.
- Dosing Regimen: Administer the test article via the intended clinical route (e.g., intravenous infusion) once weekly for 4 weeks (rodents) or 13 weeks (non-rodents). Monitor animals daily for clinical signs.
- In-Life Monitoring: Record body weight and food consumption weekly. Perform detailed clinical observations and ophthalmologic exams pre-dose and at scheduled intervals. Collect blood for hematology, coagulation, and clinical chemistry panels at baseline, mid-study, and termination.
- Biodistribution Analysis (Terminal): At scheduled intervals (e.g., 48 hours and 2 weeks post-final dose), euthanize a subset of animals. Harvest major organs (liver, spleen, kidney, heart, lung, brain, gonads, etc.). Flash-freeze tissue samples in liquid nitrogen.
- DNA Analysis: Extract genomic DNA. Use ddPCR or NGS to quantify CRISPR-mediated indels at the intended on-target site and computationally predicted top off-target sites.
- RNA Analysis (for mRNA delivery): Extract total RNA. Use RT-qPCR to quantify residual Cas9 mRNA levels.
- Nanocarrier Distribution: Use mass spectrometry (for ionizable lipids) or fluorescence imaging (if labeled) to quantify nanocarrier accumulation.
- Gross Necropsy and Histopathology: Conduct a full gross necropsy on all animals. Weigh key organs. Preserve tissues in 10% neutral buffered formalin, process, section, and stain with H&E. A board-certified veterinary pathologist should perform a blinded evaluation.
- Immunogenicity Assessment: Analyze serum samples for anti-Cas9 antibodies and anti-PEG antibodies (if PEGylated nanocarrier) using validated ELISA methods.
Deliverables: A final study report detailing NOAEL (No Observed Adverse Effect Level), target organ toxicity, dose-response relationship, comprehensive biodistribution profile, and immunogenicity data to support the IND application.
Diagram: Key Safety Signaling Pathways for Immune Activation
Immune and Stress Pathways Activated by CRISPR-LNPs
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents and Tools for Preclinical Safety Assessment
| Reagent/Tool | Supplier Examples | Function in Safety Assessment |
|---|---|---|
| hERG Assay Kit | Eurofins, Charles River | Measures inhibition of the hERG potassium channel to predict potential cardiac arrhythmia (QT prolongation) risk. |
| Cytokine Multiplex Assay (Luminex/MSD) | Thermo Fisher, Meso Scale Discovery | Quantifies a panel of pro-inflammatory cytokines (e.g., IL-6, TNF-α, IFN-γ) in serum to assess immunostimulation. |
| Anti-Cas9 IgG ELISA Kit | Custom from AntibodyWorks, Sino Biological | Detects and quantifies host immune response (antibodies) against the bacterial Cas9 protein, critical for understanding immunogenicity. |
| ddPCR Assay for Off-Target Analysis | Bio-Rad (ddPCR Supermix), custom gRNA design | Provides absolute, sensitive quantification of on-target editing and off-target events at specific genomic loci without needing standard curves. |
| T7 Endonuclease I / Surveyor Nuclease | NEB, Integrated DNA Technologies | Detects CRISPR-induced indels via mismatch cleavage in PCR products; a cost-effective method for initial on/off-target screening. |
| Guide-it Off-Target Site Predictor | Takara Bio | In silico tool to predict top potential off-target sites for gRNA sequences, guiding empirical analysis. |
| Recombinant AAV or Lentiviral Positive Control | Vigene, VectorBuilder | Provides a positive control for vector genome biodistribution studies (e.g., qPCR standard curve) and comparators for immunogenicity. |
| Formulation Lipids (e.g., Ionizable, PEGylated) | Avanti Polar Lipids, BroadPharm | High-purity lipids for preparing research-grade LNPs to match CMC properties as closely as possible in early toxicology studies. |
| Next-Generation Sequencing (NGS) Service for GUIDE-seq | Genewiz, Azenta | Comprehensive, unbiased method (GUIDE-seq, CIRCLE-seq) to identify genome-wide off-target effects of CRISPR nucleases. |
| Clinical Pathology Analyzer | IDEXX, Heska | Automated systems for analyzing hematology and clinical chemistry parameters from animal studies, essential for toxicity monitoring. |
Application Notes: Integrating Hybrid Nanosystems into CRISPR Delivery Research
The convergence of CRISPR-Cas machinery with advanced nanocarriers represents a paradigm shift in therapeutic gene editing. The primary challenges—nuclease degradation, immune clearance, and inefficient intracellular delivery—are being addressed by next-generation hybrid nanosystems. The following application notes detail critical quantitative findings and protocols central to this field.
Table 1: Comparative Performance Metrics of Leading Hybrid Nanosystems for CRISPR Delivery (2023-2024)
| Nanosystem Class | Core Composition (Example) | Average Size (nm) | Zeta Potential (mV) | Loading Efficiency (%) | In Vivo Tumor Model Editing Efficiency (%) | Key Advantage |
|---|---|---|---|---|---|---|
| Lipid-Inorganic Hybrid | Ionizable lipid / Gold NP core | 85 ± 12 | +5.2 ± 1.1 | 92.5 ± 3.1 | 41.2 (Liver) | NIR-triggered release; enhanced endosomal escape. |
| Polymer-Metal Organic Framework (MOF) | PEI-coated ZIF-8 | 120 ± 25 | +28.5 ± 2.3 | 88.7 ± 4.5 | 33.8 (Tumor) | High protection; pH-responsive degradation. |
| Exosome-Liposome Hybrid (Exosomeosome) | HEK293 exosome membrane fused with synthetic lipid bilayer | 95 ± 15 | -12.3 ± 1.8 | 65.4 ± 5.2 | 38.5 (Brain) | Superior immune evasion; natural tropism. |
| DNA Origami-Cationic Polymer | Triangular DNA origami coated with PEG-PLL | 65 ± 5 | -3.5 ± 0.8 | ~99 (programmable) | 22.1 (Solid Tumor) | Atomic precision; multi-component assembly. |
| Stimuli-Responsive Polymer Micelle | UV-cleavable PEG shell, poly(β-amino ester) core | 75 ± 8 | -1.0 ± 0.5 | 78.9 ± 2.7 | 29.4 (Skin) | Spatiotemporal control via external trigger. |
Detailed Experimental Protocols
Protocol 1: Synthesis and CRISPR Loading of pH-Responsive Polymer-MOF Hybrid Nanosystems
Objective: To fabricate ZIF-8 MOFs coated with a cationic polymer (e.g., polyethylenimine, PEI) for the co-delivery of Cas9 mRNA and sgRNA (ribonucleoprotein, RNP alternative).
Materials & Reagents:
- Zinc nitrate hexahydrate & 2-Methylimidazole: Precursors for ZIF-8 synthesis.
- Branched PEI (25 kDa): Provides colloidal stability and enhances endosomal escape via the "proton sponge" effect.
- Cas9 mRNA and sgRNA (or pre-complexed RNP): The CRISPR payload.
- Methanol & DI Water: Synthesis solvents.
- Spectra/Por Dialysis Membranes (MWCO 100kDa): For purification.
Procedure:
- MOF Core Formation: Dissolve zinc nitrate (0.5 mmol) and 2-methylimidazole (4 mmol) separately in 10 mL methanol. Rapidly mix the two solutions under vigorous stirring (1200 rpm) at room temperature for 1 hour. The solution will turn opaque.
- Purification: Centrifuge the suspension at 15,000 × g for 15 min. Wash the white precipitate with fresh methanol twice to remove unreacted precursors.
- Polymer Coating: Re-disperse the ZIF-8 pellet in 5 mL DI water via sonication (30 s, 30% amplitude). Add an aqueous solution of PEI (5 mg/mL) dropwise to achieve a 1:2 (w/w) ZIF-8:PEI ratio. Stir for 4 hours at RT to allow electrostatic adsorption.
- CRISPR Loading: Incubate the PEI@ZIF-8 nanosystem with Cas9 mRNA and sgRNA (mass ratio 10:1:1, nanosystem:RNA) in nuclease-free buffer for 30 min at 4°C to allow complexation via electrostatic interactions.
- Final Purification: Dialyze the final formulation against DI water (1 L, 2 changes over 4 hours) to remove unbound components. Filter sterilize (0.22 μm PES membrane). Characterize size, PDI, and zeta potential using dynamic light scattering (DLS).
Protocol 2: In Vitro Assessment of Editing Efficiency and Intracellular Trafficking
Objective: To quantify CRISPR-Cas9 mediated gene knockout and visualize the intracellular fate of hybrid nanosystems.
Materials & Reagents:
- HEK293T Cells (or target cell line): Stably expressing GFP.
- GFP Targeting sgRNA & Cas9: To disrupt GFP expression.
- LysoTracker Deep Red & Hoechst 33342: For live-cell staining of lysosomes and nuclei.
- Flow Cytometry Buffer (PBS + 2% FBS): For cell analysis.
- Confocal Imaging Dish: For live-cell microscopy.
Procedure:
- Cell Transfection: Seed HEK293T-GFP cells in a 24-well plate at 70% confluence. After 24h, treat cells with CRISPR-loaded nanosystems (Protocol 1) at a concentration of 100 nM RNP equivalent. Include untreated and Lipofectamine CRISPRMAX positive controls.
- Harvesting for Flow Cytometry: At 72h post-transfection, trypsinize cells, wash with PBS, and resuspend in flow cytometry buffer. Analyze GFP signal intensity using a flow cytometer (e.g., FITC channel). Calculate editing efficiency as the percentage reduction in GFP-positive cells relative to untreated control.
- Live-Cell Imaging for Trafficking: Seed cells in a confocal dish. Treat with fluorescently labeled (e.g., Cy5) nanosystems for 4-6h. 30 min before imaging, add LysoTracker Deep Red and Hoechst 33342 per manufacturer's protocol. Perform confocal microscopy using appropriate laser lines. Co-localization analysis (Pearson's coefficient) between the nanosystem signal (Cy5) and lysosomal signal (Deep Red) indicates endolysosomal entrapment.
Visualization: Pathways and Workflows
Diagram 1: Intracellular Journey of a Smart CRISPR Nanosystem
Diagram 2: Workflow for Hybrid Nanosystem Development & Testing
The Scientist's Toolkit: Essential Research Reagents & Materials
| Item | Function/Application in CRISPR Nanosystems Research |
|---|---|
| Ionizable Lipids (e.g., DLin-MC3-DMA, SM-102) | Core component of LNPs; ionizable for efficient encapsulation and endosomal escape. |
| Branched Polyethylenimine (PEI, 25 kDa) | "Gold standard" cationic polymer for nucleic acid complexation and proton-sponge mediated endosomal escape. |
| Poly(lactic-co-glycolic acid) (PLGA) | Biodegradable polymer for sustained release of encapsulated CRISPR components. |
| N-hydroxysuccinimide (NHS) & 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide (EDC) | Crosslinker chemistry for conjugating targeting ligands (peptides, antibodies) to nanoparticle surfaces. |
| DSPE-PEG(2000)-Maleninde | PEG-lipid derivative for "stealth" coating and providing a thiol-reactive terminus for ligand conjugation. |
| CellRox Green / LysoTracker Deep Red | Fluorescent probes for measuring intracellular ROS (nanotoxicity) and lysosomal colocalization, respectively. |
| Cy5-labeling Kit for Protein/RNA | Fluorescent dye conjugation kit for tracking nanoparticle components in vitro and in vivo. |
| In Vivo-JetPEI | A commercially available, in vivo-optimized polymer transfection reagent used as a positive control in animal studies. |
| sgRNA Synthesis Kit (T7 promoter) | For high-yield, in vitro transcription of research-grade sgRNAs for complexation with Cas9 protein or mRNA. |
| Recombinant Cas9 Nuclease (or mRNA) | The core effector protein for gene editing; available as purified protein or as mRNA for translation in situ. |
Conclusion
CRISPR nanotechnology delivery systems represent a transformative frontier in gene therapy, moving beyond the limitations of first-generation vectors. The convergence of material science and molecular biology has yielded a diverse toolkit—from refined viral vectors to modular non-viral platforms like LNPs—each with distinct advantages for specific therapeutic contexts. Success hinges on a balanced optimization of delivery efficiency, precision targeting, and biocompatibility. Future directions point toward intelligent, stimuli-responsive systems capable of spatial and temporal control, multi-target editing, and streamlined manufacturing for clinical deployment. For researchers and drug developers, a nuanced understanding of this comparative landscape is essential to selecting and engineering the optimal delivery vector, ultimately accelerating the realization of safe and effective CRISPR-based medicines for a broad spectrum of human diseases.