Nanospike-Induced Bacterial Cell Rupture: Mechanisms, Applications, and Future Directions in Antimicrobial Technology
This article provides a comprehensive review of the cutting-edge field of bacterial cell rupture using nanostructured surfaces.
Nanospike-Induced Bacterial Cell Rupture: Mechanisms, Applications, and Future Directions in Antimicrobial Technology
Abstract
This article provides a comprehensive review of the cutting-edge field of bacterial cell rupture using nanostructured surfaces. It explores the fundamental biophysical mechanisms underpinning nanospike-bacteria interactions, detailing how precise geometry and surface properties induce mechanical stress and lysis. We examine state-of-the-art fabrication methodologies for silicon, titanium, and polymer-based nanospikes, alongside their applications in medical implants, wound dressings, and antimicrobial surfaces. The content addresses critical challenges in fabrication consistency, biological variability, and environmental factors, offering troubleshooting and optimization strategies. Finally, we present rigorous validation techniques, including electron microscopy and live-cell assays, and perform comparative analyses against traditional antibiotics and other physical antimicrobial methods. This synthesis is designed for researchers, scientists, and drug development professionals seeking to leverage physical mechanisms to combat antimicrobial resistance.
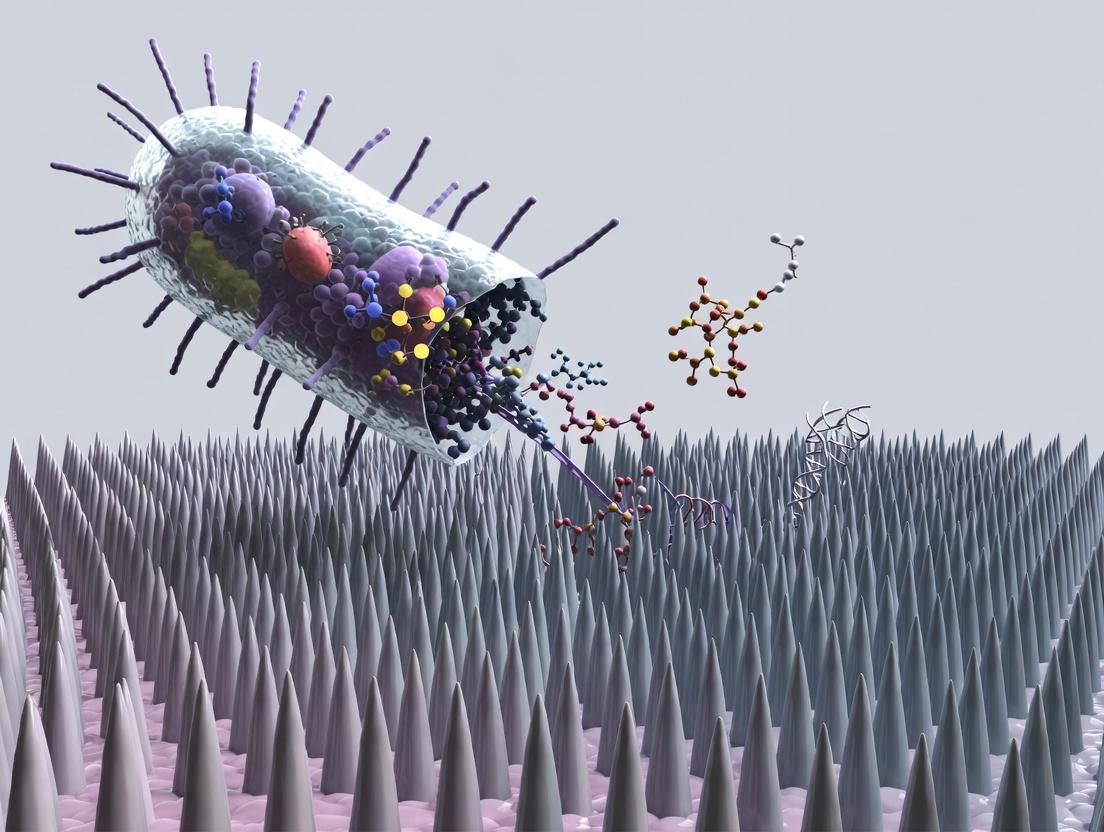
The Physics of Piercing: How Nanospikes Mechanically Rupture Bacterial Cells
Within the research paradigm of developing non-antibiotic antimicrobial surfaces, nanospikes have emerged as a potent physical bactericidal platform. The core thesis of this field posits that surface topography, specifically high-aspect-ratio nanoscale protrusions, can mechanically compromise bacterial cell integrity, leading to cell rupture and death, independent of biochemical agents. The efficacy of this mechanism is not defined by a single parameter but by the intricate interplay of three fundamental characteristics: Geometry, Density, and Material Composition. This whitepaper provides a technical deconstruction of these pillars, serving as a foundational guide for research and development in this area.
Deconstructing the Core Characteristics
Geometry
Geometry refers to the three-dimensional shape and dimensions of individual nanospikes. It is the primary determinant of the stress concentration applied to the bacterial cell membrane.
- Height: Must exceed the local deformation limit of the bacterial cell envelope to ensure penetration. Typically ranges from 200 nm to >1000 nm.
- Tip Diameter/Sharpness: The most critical geometric factor. A sharper tip (sub-50 nm, ideally <20 nm) reduces the contact area, dramatically increasing the localized pressure (Stress = Force/Area).
- Aspect Ratio: The ratio of height to base width. High aspect ratios (>10:1) promote bending and flexibility, which can influence the rupture mechanism.
- Tip Angle: A smaller apical angle creates a more piercing profile.
- Sidewall Profile: Tapered, cylindrical, or needle-like shapes influence bending stiffness and interaction with the cell wall.
Density
Density, or pitch (inter-spike distance), dictates whether a bacterial cell experiences a "bed of nails" effect or a "stretching and piercing" mechanism.
- High Density (Pitch < 100 nm): Prevents membrane sagging between spikes, ensuring the cell body is simultaneously impaled by multiple spikes, leading to massive, simultaneous poration.
- Intermediate Density (Pitch ~100-500 nm): Allows the membrane to stretch and drape between adhesion points, potentially leading to tensile stress-induced rupture before or during puncture.
- Low Density (Pitch > 500 nm): Risks the cell settling between spikes without contact, rendering the surface ineffective.
Material Composition
The material dictates the mechanical (stiffness, modulus), chemical (surface energy, reactivity), and biological (cytotoxicity) properties of the nanospike array.
- Intrinsic Mechanical Properties: A high Young's modulus (e.g., silicon, titanium, diamond-like carbon) prevents spike collapse under load.
- Surface Chemistry: Influences initial bacterial adhesion via hydrophobic/hydrophilic interactions. Can be functionalized for synergistic chemical effects.
- Biocompatibility: Critical for applications in medical implants (e.g., titanium, polycarbonate).
- Manufacturability: Materials like silicon, polymers, and metals each offer different fabrication routes (etching, molding, deposition).
Table 1: Quantitative Parameters of Nanospike Characteristics
| Characteristic | Parameter | Typical Effective Range | Impact on Bactericidal Mechanism |
|---|---|---|---|
| Geometry | Height | 200 - 2000 nm | Must surpass cell envelope deformation limit. |
| Tip Diameter | < 50 nm (ideal: <20 nm) | Directly governs localized puncturing pressure. | |
| Aspect Ratio | 5:1 to 50:1 | Affects buckling stability and penetration mode. | |
| Density | Inter-Spike Pitch (Center-to-Center) | 50 - 400 nm | Determines membrane deformation mode (stretch vs. direct puncture). |
| Areal Density | 10^8 - 10^{10} spikes/cm² | Correlates with probability of lethal contact. | |
| Material | Young's Modulus | > 50 GPa (for rigid spikes) | Ensures spike does not deform or buckle upon contact. |
| Surface Energy | Variable (20-70 mN/m) | Mediates initial adhesion and wetting behavior. |
Experimental Protocols for Characterization & Efficacy Testing
Protocol 1: Fabrication & Physical Characterization of Nanospikes
- Objective: To fabricate a nanospike array and quantify its geometric and material properties.
- Method A: Deep Reactive Ion Etching (DRIE) for Silicon Nanospikes
- Mask Patterning: Deposit and pattern a metal (e.g., Au) or hard mask (SiO₂) on a silicon wafer using photolithography or nanoimprint lithography.
- Bosch Process: Use a cyclical DRIE process (SF₆ etch, C₄F₈ passivation) to etch high-aspect-ratio silicon pillars.
- Sharpening: Perform a brief, isotropic wet etch (e.g., with HF:HNO₃:CH₃COOH) to sharpen pillar tips into nanospikes.
- Method B: Hydrothermal Synthesis for TiO₂ Nanospikes
- Substrate Preparation: Clean a titanium substrate.
- Reaction: Immerse the substrate in a strong alkaline solution (e.g., 5-10 M NaOH) at 150-250°C for 1-24 hours in an autoclave.
- Annealing: Heat-treat the formed sodium titanate nanostructures at 400-600°C to convert them to crystalline TiO₂ (anatase/rutile) nanospikes.
- Characterization:
- SEM: Measure height, tip diameter, pitch, and density.
- AFM: Quantify nanoscale topography and mechanical properties (modulus via force spectroscopy).
- XPS/EDS: Determine surface elemental composition and chemistry.
Protocol 2: Standardized Bactericidal Efficacy Assay (ISO 22196 / JIS Z 2801 Modified)
- Objective: To quantitatively assess the rate of bacterial killing on a nanospike substrate.
- Materials: Test strain (e.g., E. coli ATCC 25922, S. aureus ATCC 6538), nutrient broth, neutralizer solution (e.g., Letheen Broth), PBS.
- Procedure:
- Inoculation: Apply 100 µL of bacterial suspension (≈ 3 x 10^5 CFU/mL in PBS) onto the nanospike surface and a smooth control surface. Cover with a sterile, inert film to spread evenly.
- Incubation: Incubate samples at 35°C and >90% relative humidity for 24 hours.
- Neutralization & Recovery: Transfer each sample to a vessel containing 10 mL of neutralizer solution. Vortex vigorously for 1-2 minutes to dislodge cells.
- Enumeration: Perform serial dilutions and plate on nutrient agar. Count Colony Forming Units (CFU) after 24-48 hours incubation.
- Calculation: Calculate antibacterial activity (R) = log₁₀(Ctrl CFU) - log₁₀(Test CFU). An R > 2 (99% kill) is considered strongly bactericidal.
Protocol 3: Visualization of Cell Rupture via Electron Microscopy
- Objective: To visually confirm the physical rupture of bacterial cells.
- Procedure:
- Sample Incubation: Incubate nanospike and control surfaces with bacterial suspension for 1-4 hours.
- Fixation: Fix samples in 2.5% glutaraldehyde in 0.1M sodium cacodylate buffer (pH 7.4) for 2 hours at 4°C.
- Dehydration: Rinse with buffer and dehydrate through a graded ethanol series (30%, 50%, 70%, 90%, 100%) for 10 minutes each.
- Drying: Use critical point drying (CPD) or hexamethyldisilazane (HMDS) to preserve nanostructures.
- Coating: Sputter-coat samples with a thin (5-10 nm) layer of gold/palladium.
- Imaging: Analyze using SEM at 5-15 kV to observe cell morphology, adhesion, and puncture sites.
Diagram 1: Bacterial Cell Rupture Pathways on Nanospikes
Diagram 2: Core Experimental Workflow for Nanospike Research
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 2: Key Research Reagents and Materials for Nanospike Bactericidal Studies
| Item | Function/Description | Example(s) |
|---|---|---|
| Standard Test Strains | Gram-negative and Gram-positive bacteria for standardized efficacy testing. | Escherichia coli ATCC 25922, Staphylococcus aureus ATCC 6538, Pseudomonas aeruginosa ATCC 9027. |
| Neutralizer Solution | Halts antimicrobial action during recovery to ensure accurate viable counts. | Letheen Broth, D/E Neutralizing Broth, or PBS with Tween 80 & Lecithin. |
| Fixative for EM | Preserves bacterial cell morphology and spatial relationship to nanospikes for SEM imaging. | 2.5-4% Glutaraldehyde in 0.1M Sodium Cacodylate or Phosphate Buffer. |
| Critical Point Dryer (CPD) | Removes liquid from SEM samples without surface tension-induced collapse of cells or nanostructures. | Using CO₂ as transition fluid. |
| Sputter Coater | Applies a thin, conductive metal layer to non-conductive samples for SEM imaging. | Gold/Palladium (Au/Pd) target, 5-10 nm thickness. |
| Reactive Ion Etch Gases | For precise, anisotropic etching of silicon to create high-aspect-ratio spikes. | Sulfur Hexafluoride (SF₆, etch) & Octafluorocyclobutane (C₄F₈, passivation) for the Bosch process. |
| Hydrothermal Reaction Agents | For growing metal oxide nanospikes on substrates. | Sodium Hydroxide (NaOH) pellets for TiO₂ nanospike synthesis on Ti. |
| Atomic Layer Deposition (ALD) Precursors | For conformal coating of nanospikes with different materials to study composition effects. | Trimethylaluminum (TMA) for Al₂O₃, Tetrakis(dimethylamido)titanium (TDMAT) for TiN. |
This whitepaper, framed within a broader thesis on bacterial cell rupture via nanospikes, provides an in-depth technical examination of the bacterial cell envelope as a primary target for mechanical stress. For researchers in antimicrobial surfaces and drug development, understanding the precise failure mechanics of the cell wall and membrane under physical deformation is critical for designing next-generation bactericidal platforms.
Structural Targets: Peptidoglycan and Membranes
The Peptidoglycan Sacculus
The peptidoglycan (PG) layer, a mesh-like polymer of glycan strands cross-linked by peptides, is the primary stress-bearing component. Its mechanical properties dictate cell shape and resistance to turgor pressure.
Table 1: Mechanical Properties of Gram-Positive and Gram-Negative Envelopes
| Property | Gram-Positive | Gram-Negative | Measurement Technique |
|---|---|---|---|
| PG Thickness | 20-80 nm | 2-7 nm | TEM, Atomic Force Microscopy (AFM) |
| Young's Modulus (PG) | 10-50 MPa | 10-50 MPa | AFM Nanoindentation |
| Turgor Pressure | 1.5-3.0 MPa | 0.3-0.5 MPa | Plasmolysis, AFM |
| Critical Stress for Rupture | ~100-300 mN/m (of sacculus) | ~10-50 mN/m (of sacculus) | Optical Trap, Micropipette Aspiration |
The Cytoplasmic and Outer Membranes
The cytoplasmic membrane (CM) is a phospholipid bilayer with embedded proteins. In Gram-negatives, the outer membrane (OM) is an asymmetric bilayer with lipopolysaccharide (LPS) in the outer leaflet, providing additional structural integrity and barrier function. Mechanical puncture leads to rapid depolarization, osmotic imbalance, and content leakage.
Experimental Protocols for Envelope Stress Analysis
Protocol: Atomic Force Microscopy (AFM) Nanoindentation for Stiffness Mapping
Objective: To measure local Young's modulus and rupture force of bacterial envelopes. Methodology:
- Sample Preparation: Grow bacterial culture to mid-log phase. Immobilize cells on poly-L-lysine coated glass slides or porous membranes. Rinse gently with appropriate buffer (e.g., PBS or MOPS).
- AFM Setup: Use a sharp, silicon nitride tip (nominal radius < 20 nm). Calibrate the cantilever spring constant using thermal tuning.
- Measurement: In fluid cell, position tip over a cell pole or mid-section. Approach at 0.5-1 µm/s. Record force-distance curves. Apply Hertz or Sneddon contact models for spherical or conical tips to extract Young's modulus from the indentation curve's slope.
- Rupture Detection: A sudden discontinuity ("jump") in the retraction or approach curve indicates envelope puncture. The corresponding force is the rupture force.
Protocol: Membrane Integrity Assays Post-Mechanical Stress
Objective: To quantify loss of envelope integrity following nanospike contact or other mechanical insult. Methodology:
- Stress Application: Expose bacterial suspension to nanostructured surfaces for a defined time or use sonication for controlled shear stress.
- Viability Staining: Use a LIVE/DEAD BacLight bacterial viability kit. Mix SYTO 9 (green, penetrates all cells) and propidium iodide (PI, red, enters only compromised membranes) as per protocol. Incubate with stressed cells for 15 min in dark.
- Quantification: Analyze via fluorescence microscopy or flow cytometry. Calculate percentage of PI-positive (ruptured) cells.
- Cytoplasmic Leakage: Measure release of cytoplasmic components like ATP (using luciferase assays) or UV-absorbing materials (A260) from supernatants of centrifuged samples.
Mechanosensing and Response Pathways
Bacteria possess envelope stress response systems that detect and counteract damage. Mechanical perturbation can activate these pathways.
Title: Envelope Stress Response to Mechanical Damage
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for Bacterial Envelope Mechanics Research
| Item | Function/Application | Example Product/Strain |
|---|---|---|
| AFM Cantilevers | High-resolution force measurement and nanoindentation. | Bruker MSNL (sharp) or BioScope Catalyst tips. |
| Fluorescent Membrane Dyes | Visualizing membrane integrity and potential. | DiBAC₄(3) (depolarization), FM 4-64 (membrane insertion). |
| LIVE/DEAD BacLight | Differentiating intact vs. compromised membranes via flow cytometry or microscopy. | Thermo Fisher Scientific L7012. |
| Lysozyme | Enzymatic digestion of peptidoglycan for controlled weakening of the cell wall. | Sigma-Aldrich L6876. |
| Osmotic Stabilizers | Maintaining cell integrity in hypotonic buffers during mechanical manipulation. | Sucrose (0.25-0.5 M), Sorbitol. |
| Penicillin Binding Protein (PBP) Labels | Visualizing PG synthesis activity under stress. | Bocillin FL (fluorescent ampicillin derivative). |
| E. coli MG1655 ΔtolC | Strain with increased OM permeability, useful for compound penetration studies. | Keio collection strain JW5501. |
| B. subtilis 168 | Model Gram-positive organism with well-characterized cell envelope stress responses. | ATCC 23857. |
| Nanostructured Surfaces | Black Silicon, Diamond Nanospikes, or TiO₂ nanotubes for applied mechanical stress studies. | Commercial or custom-fabricated substrates. |
Experimental Workflow for Nanospike Rupture Studies
Title: Nanospike Bactericidal Study Workflow
Quantitative Rupture Thresholds
Table 3: Reported Mechanical Rupture Thresholds from Literature
| Stress Type | Organism | Critical Force/Stress | Method | Proposed Failure Point |
|---|---|---|---|---|
| Nanospike Indentation | S. aureus (Gram+) | 5-20 nN per spike | AFM on Si nanospikes | Local PG puncture, followed by membrane rupture. |
| Shear Stress (Flow) | E. coli (Gram-) | 10-50 Pa | Microfluidic shear device | OM detachment, then PG and CM failure. |
| Uniaxial Stretching | B. subtilis (Gram+) | 5-10% areal strain | Microfabricated stretchers | PG layer fracture, turgor-driven CM extrusion. |
| Osmotic Downshock | E. coli (Gram-) | ~0.5 MPa (ΔP) | Rapid dilution | CM lysis due to excessive turgor on weakened OM/PG. |
Targeting the bacterial cell envelope with precisely engineered mechanical stress, such as that from nanospikes, presents a potent physical antibacterial strategy. Its efficacy is governed by quantifiable material properties and failure thresholds of the PG and membranes. Combining advanced force measurement, genetic tools, and standardized integrity assays allows researchers to deconstruct the sequence of envelope failure, informing the rational design of antimicrobial surfaces and adjunct therapies that potentiate traditional antibiotics.
This whitepaper details the primary mechanical rupture mechanisms of bacterial cells upon interaction with nanostructured surfaces, specifically nanospikes. The analysis is framed within a broader thesis investigating the physicochemical determinants of bactericidal activity on biomimetic nanotextured materials. Understanding the dichotomy between direct penetration and stress-induced stretching is critical for the rational design of antimicrobial surfaces in medical devices, drug delivery systems, and industrial coatings.
Core Rupture Mechanisms: A Comparative Analysis
Membrane Penetration (Nano-Spearing)
This mechanism involves the direct physical piercing of the cell envelope by nanospikes. The sharp topography overcomes the mechanical strength of the lipid bilayer and cell wall, creating irreversible pores.
Key Physical Determinants:
- Spike Geometry: Tip radius, aspect ratio, and sharpness.
- Cell Envelope Mechanics: Local stiffness and puncture resistance.
- Interaction Forces: Adhesion, gravitational, and capillary forces driving penetration.
Stretching-Induced Lysis (Membrane Expansion)
This mechanism occurs when the bacterial cell is suspended between multiple spikes, leading to large-area deformation. Lysis results from the over-stretching of the membrane beyond its critical areal strain, causing bilayer failure without direct puncture.
Key Physical Determinants:
- Spike Density and Arrangement: Inter-spike distance relative to cell diameter.
- Membrane Elasticity: Critical areal expansion limit before failure.
- Adhesion Strength: Determines the contact area and transmitted tensile stress.
Table 1: Comparative Analysis of Rupture Mechanisms
| Parameter | Membrane Penetration | Stretching-Induced Lysis |
|---|---|---|
| Primary Driver | Local pressure at spike tip | Global tensile stress on membrane |
| Critical Spike Property | Tip radius < 50 nm | Inter-spike spacing 100-300 nm |
| Typical Failure Strain | Low (< 5%) at puncture site | High (>> 5%) over large area |
| Energy Requirement | High energy density at tip | Lower energy density, distributed |
| Visual Signature (SEM) | Clear punctures, localized damage | Sagging membrane, large tears |
| Dependence on Turgor Pressure | High: Higher pressure facilitates rupture | Moderate: Can occur across pressures |
| Kinetics | Fast (milliseconds) | Slower (seconds to minutes) |
Table 2: Experimental Measurements from Recent Studies (2022-2024)
| Study (Model System) | Spike Dimensions (Height/Tip Radius) | Rupture Force (Calculated) | Dominant Mechanism Observed |
|---|---|---|---|
| Black Silicon vs. E. coli | 500 nm / 10 nm | ~5 nN per spike | Penetration |
| ZnO Nanorods vs. S. aureus | 1 µm / 30 nm | ~1-2 nN per spike | Mixed (Penetration & Stretching) |
| TiO₂ Nanopillars vs. P. aeruginosa | 200 nm / 50 nm | N/A (Stretching-dominated) | Stretching-Induced Lysis |
| Polymer Nanoneedles vs. B. subtilis | 2 µm / <5 nm | ~7 nN per spike | Penetration |
Experimental Protocols for Mechanism Differentiation
Protocol: In-Situ Atomic Force Microscopy (AFM) Nanomechanical Mapping
Objective: To measure real-time force curves during bacterial contact with a single nanospike. Materials: AFM with a sharp, spike-functionalized probe (e.g., Si tip with deposited carbon nanotube), bacterial cell immobilized on poly-L-lysine coated substrate in liquid medium. Procedure:
- Calibrate the AFM cantilever spring constant using thermal tuning.
- Approach the functionalized tip towards the center of a bacterial cell at a controlled rate (e.g., 500 nm/s).
- Record the force-distance curve. A sudden, rapid discontinuity in the approach curve indicates puncture (Penetration Mechanism).
- For stretching, retract the tip after initial contact. A long, nonlinear retraction curve with high adhesion force indicates membrane tether formation and stretching.
- Repeat on multiple cells (n>50) and locations.
Protocol: Static Incubation & Post-Mortem SEM/TEM Analysis
Objective: To correlate nanospike geometry with morphological damage signatures. Materials: Nanostructured substrate, bacterial suspension, fixatives (2.5% glutaraldehyde, 1% osmium tetroxide), ethanol series for dehydration, critical point dryer. Procedure:
- Incubate bacterial suspension on nanostructured surface for a fixed time (e.g., 60 min).
- Gently rinse with PBS to remove non-adhered cells.
- Fix samples in glutaraldehyde (2h), then osmium tetroxide (1h).
- Dehydrate through a graded ethanol series (30%, 50%, 70%, 90%, 100%).
- Perform critical point drying.
- Sputter-coat with a thin (5 nm) iridium layer for SEM, or prepare ultrathin sections for TEM.
- Image and categorize damage: Punctures (clean holes) vs. Stretching (draping, large indentations, tears between adhesion points).
Visualization of Mechanobactericidal Pathways
(Decision Logic for Primary Rupture Mechanism)
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Nanospike-Bacteria Rupture Studies
| Item | Function & Rationale |
|---|---|
| Black Silicon Substrates | Model nanostructured surface with high-aspect-ratio, sharp silicon nanospikes for penetration-dominant studies. |
| Polymer (e.g., PU, PCL) Nanoneedle Arrays | Tunable, flexible nanostructures to study the interplay of stiffness and geometry on stretching. |
| Fluorescent Membrane Probes (e.g., FM dyes, DiI) | To visualize real-time membrane integrity loss and differentiate leaky pores (penetration) from large tears (stretching). |
| Osmoprotectants (e.g., Sucrose, Sorbitol) | To modulate bacterial turgor pressure, a key variable affecting susceptibility to both rupture mechanisms. |
| Atomic Force Microscopy (AFM) with Fluid Cell | For quantitative, single-cell nanomechanical measurements of rupture forces and adhesion energies. |
| Cryo-Fixation Equipment (HPF/Freeze Substitution) | For optimal preservation of native cell-spike interaction morphology for TEM, avoiding drying artifacts. |
| Lipid Bilayer Models (e.g., GUVs - Giant Unilamellar Vesicles) | Simplified model systems to study pure membrane mechanics without the complexity of the cell wall. |
| Finite Element Analysis (FEA) Software (e.g., COMSOL) | To computationally model stress distributions and predict failure modes based on spike geometry and material properties. |
The Role of Surface Wettability and Chemistry in Bacterial Adhesion and Fate.
Research into bactericidal nanostructured surfaces, such as nanospikes, has demonstrated remarkable physical rupture of adhered cells. However, the initial bacterial adhesion—a critical prerequisite for this mechanical killing—is governed not by topography alone, but by the interplay of surface wettability and chemistry. This whitepaper posits that optimizing wettability and surface chemistry is essential to maximize the probability of bacterial adhesion onto lethal nanospikes, thereby enhancing the overall efficacy of the bactericidal surface. Understanding and controlling these interfacial properties allows researchers to design surfaces that selectively promote bacterial attachment for subsequent physical disruption, while potentially minimizing fouling by organic matter or eukaryotic cells.
Fundamental Principles of Bacterial Adhesion
Bacterial adhesion is a two-stage process: initial, reversible attachment mediated by non-specific physicochemical forces, followed by irreversible adhesion reinforced by specific molecular interactions and biofilm matrix production.
2.1. Key Physicochemical Parameters:
- Surface Free Energy (SFE) & Wettability: Commonly characterized by water contact angle (WCA).
- Hydrophobic Surfaces (WCA > 90°): Often favor adhesion of hydrophobic bacteria via hydrophobic interactions.
- Hydrophilic Surfaces (WCA < 90°): May resist adhesion via strong hydration layers, but can attract bacteria via electrostatic or acid-base interactions.
- Surface Chemistry: Determines functional groups present (e.g., -CH3, -OH, -NH2, -COOH), which influence SFE, charge, and hydrogen bonding capacity.
- Surface Charge (Zeta Potential): Bacteria typically carry a negative charge; surfaces with positive charge can promote adhesion via electrostatic attraction.
2.2. Thermodynamic Approach: The change in free energy (ΔG) of adhesion between a bacterium (B), surface (S), and liquid (L) dictates the tendency for attachment. Surfaces with low ΔGadh favor adhesion.
Quantitative Data on Wettability/Chemistry vs. Adhesion Outcomes
Table 1: Impact of Surface Chemistry & Wettability on Bacterial Adhesion Density and Viability.
| Surface Chemistry | Water Contact Angle (°) | Bacterial Strain | Adhesion Density (cells/cm²) | Key Finding | Ref. Year |
|---|---|---|---|---|---|
| Hydrocarbon (-CH₃) | 110±3 (Hydrophobic) | S. aureus | ~1.2 x 10⁶ | High adhesion, but low subsequent killing on flat surface. | 2023 |
| Amino (-NH₂) | 45±2 (Hydrophilic) | E. coli | ~8.5 x 10⁵ | Moderate adhesion with significant membrane stress. | 2022 |
| Carboxyl (-COOH) | 25±3 (Super-Hydrophilic) | P. aeruginosa | ~2.0 x 10⁵ | Low initial adhesion, but strong binding if attached. | 2024 |
| PEG-like (Anti-fouling) | 35±4 (Hydrophilic) | S. epidermidis | ~5.0 x 10⁴ | Very low adhesion, preventing any fate mechanism. | 2023 |
Table 2: Synergy of Nanospikes with Underlying Surface Chemistry.
| Nanospike Substrate Chemistry | Adhesion on Nanospikes vs. Flat | Cell Rupture Efficiency | Proposed Mechanism |
|---|---|---|---|
| Hydrophobic (SiO₂ silanized) | 1.8x higher | 94±3% | Hydrophobic forces enhance contact, maximizing membrane stretch over spikes. |
| Hydrophilic (TiO₂) | 0.7x lower | 88±5% | Lower adhesion, but adhered cells are highly susceptible due to synergistic oxidative stress. |
| Mixed Charge (Zwitterionic) | 0.3x lower | 32±7% | Adhesion is strongly inhibited, preventing spike-cell interaction. |
Experimental Protocols for Characterization and Adhesion Assays
4.1. Protocol: Surface Functionalization via Vapor-Phase Silanization.
- Objective: To create self-assembled monolayers (SAMs) with defined terminal chemistry on oxide-coated surfaces (e.g., Si wafers with nanospikes).
- Materials: Piranha-cleaned substrates, (3-Aminopropyl)triethoxysilane (APTES), trichloro(1H,1H,2H,2H-perfluorooctyl)silane (FOTS), anhydrous toluene, nitrogen glovebox.
- Steps:
- Place dried substrates in a vacuum desiccator with a glass vial containing 50 µL of silane.
- Evacuate the desiccator for 5 min, then seal and incubate at room temperature for 3 hours.
- Vent the desiccator and bake substrates at 110°C for 30 min to cure the SAM.
- Rinse thoroughly with toluene, ethanol, and DI water, then dry under N₂ stream.
4.2. Protocol: Static Bacterial Adhesion Assay with Viability Staining.
- Objective: To quantify adhered bacteria and determine live/dead ratio on functionalized surfaces.
- Materials: Bacterial culture (OD₆₀₀=0.1 in PBS), functionalized substrates, Live/Dead BacLight stain (SYTO9 & PI), epifluorescence/confocal microscope, image analysis software (e.g., ImageJ).
- Steps:
- Place substrates in 12-well plate, add 2 mL bacterial suspension per well.
- Incubate statically at 37°C for 2 hours.
- Gently rinse with PBS 3x to remove non-adhered cells.
- Add 300 µL of Live/Dead stain mixture, incubate in dark for 15 min.
- Image immediately using appropriate filter sets. Count at least 10 fields per sample.
4.3. Protocol: Water Contact Angle (WCA) Measurement.
- Objective: To quantify surface wettability.
- Materials: Contact angle goniometer, automated dispenser, DI water.
- Steps:
- Place dry, functionalized substrate on the instrument stage.
- Dispense a 3 µL sessile water droplet onto the surface.
- Capture image within 5 seconds of droplet contact.
- Use software to fit the droplet profile and calculate the angle. Average over 5 locations.
Visualization of Key Concepts and Workflows
Title: Determinants of Bacterial Adhesion and Fate on Functionalized Nanospikes.
Title: Experimental Workflow for Studying Adhesion on Functionalized Surfaces.
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 3: Key Reagent Solutions for Surface Wettability/Chemistry and Adhesion Studies.
| Reagent/Material | Function/Description | Key Application |
|---|---|---|
| Organosilanes (APTES, FOTS, PEG-silane) | Forms self-assembled monolayers (SAMs) to present specific terminal functional groups (-NH₂, -CF₃, -Oligoethylene glycol) on oxide surfaces. | Precise control of surface chemistry and wettability. |
| Live/Dead BacLight Bacterial Viability Kit | Contains SYTO9 (green, stains all cells) and Propidium Iodide (red, stains dead cells with compromised membranes). | Differentiating live vs. dead/ruptured bacteria post-adhesion. |
| Phosphate Buffered Saline (PBS), 1X | Isotonic, pH-stabilized salt solution. | Used for bacterial suspension and rinsing to maintain physiological conditions without growth. |
| Polydimethylsiloxane (PDMS) Sylgard 184 | Two-part silicone elastomer. | Creating microfluidic channels for controlled shear adhesion assays or replicating topographies. |
| Atomic Force Microscopy (AFM) Cantilevers (with functionalized tips) | Microfabricated silicon nitride tips, can be coated with specific chemicals or bacteria. | Measuring single-cell adhesion forces (e.g., force spectroscopy) on engineered surfaces. |
| X-ray Photoelectron Spectroscopy (XPS) Standard Samples | Certified reference materials (e.g., Au, Cu, SiO₂). | Calibrating and verifying elemental composition and bonding states of functionalized surfaces. |
This technical guide details the core biophysical parameters governing the mechanobactericidal efficacy of nanostructured surfaces. Framed within a thesis on bacterial cell rupture on nanospikes, this whitepaper provides a quantitative and mechanistic analysis for researchers in microbiology, materials science, and antimicrobial drug development.
The physical rupture of bacterial cells via surface nanospikes presents a promising non-chemical antibiotic strategy. The efficacy is governed by four interdependent biophysical parameters: Spike Height (H), Tip Radius (R), Aspect Ratio (AR = H/Base Width), and Spacing/Pitch (S). This guide synthesizes current research to establish design principles for optimizing these surfaces.
Parameter Definition & Mechanistic Role
Spike Height (H)
- Role: Determines the effective engagement depth with the bacterial cell envelope. Must be sufficient to bridge the pericellular space and apply stress to the cell membrane.
- Critical Threshold: Height must exceed the distance between the base surface and the bacterium's bottom membrane, typically >500 nm for Gram-negative bacteria considering surface adhesins and glycocalyx.
Tip Radius (R)
- Role: Governs the stress concentration applied to the cell membrane. Smaller radii produce higher local pressure (P ~ 1/R for elastic deformation).
- Lethal Range: Optimal tip radii are typically sub-50 nm, approaching the thickness of the lipid bilayer (~5-10 nm) to induce puncture.
Aspect Ratio (AR)
- Role: The ratio of height to base width influences mechanical stability and bending modulus. High AR spikes are more susceptible to buckling under cell-adhesion loads.
- Design Trade-off: A high AR is needed for cell penetration, but too high leads to structural failure, reducing durability.
Spacing/Pitch (S)
- Role: Dictates the distribution of load and the ability of the cell membrane to stretch and deform between spike tips. Determines whether the mechanism is primarily "stretching-induced rupture" or "direct penetration."
- Critical Regime: Spacing must be less than the characteristic deformation length of the bacterial cell wall. Too wide allows membrane sagging; too dense can strengthen the surface via neighbor effects.
Table 1: Efficacy Ranges for Key Biophysical Parameters Against Gram-Negative Bacteria (e.g., P. aeruginosa, E. coli)
| Parameter | Symbol | Sub-optimal Range | Optimal Bactericidal Range | Supra-optimal/Destructive Range | Primary Mechanism Impacted |
|---|---|---|---|---|---|
| Spike Height | H | < 200 nm | 500 nm - 2000 nm | > 3000 nm | Engagement Depth |
| Tip Radius | R | > 100 nm | 10 nm - 50 nm | < 5 nm (fragile) | Stress Concentration |
| Aspect Ratio | AR | < 3 | 5 - 20 | > 25 | Mechanical Stability |
| Center-to-Center Spacing | S | > 250 nm | 100 nm - 200 nm | < 50 nm | Membrane Stress Distribution |
Table 2: Calculated Membrane Pressure vs. Tip Radius (Theoretical Model)
| Tip Radius (R) | Estimated Local Pressure (MPa)* | Observed Bactericidal Efficacy (%) |
|---|---|---|
| 5 nm | ~ 180 MPa | High (>99%) but tip fragility |
| 25 nm | ~ 36 MPa | Optimal (99.9%) |
| 50 nm | ~ 18 MPa | Moderate (~80%) |
| 100 nm | ~ 9 MPa | Low (<20%) |
Assuming a constant applied force. *Representative data for E. coli on silicon nanospikes.
Detailed Experimental Protocols
Protocol: Fabrication of Silicon Nanospikes via Deep Reactive Ion Etching (DRIE)
Objective: To create surfaces with precisely tunable H, R, AR, and S. Materials: P-type silicon wafer, photoresist, chrome mask, ICP-RIE system (e.g., Bosch process). Steps:
- Cleaning: Clean wafer in piranha solution (3:1 H₂SO₄:H₂O₂) for 15 min, rinse in DI water, dry.
- Photolithography: Spin-coat photoresist, soft bake, expose through chrome mask defining etch pits, develop.
- Metal Mask Deposition: E-beam evaporate a 50 nm Cr layer, lift-off in acetone to create a hard mask.
- Bosch Process Etching: In ICP-RIE, cycle between:
- Etch Step: SF₆ plasma (isotropic silicon etching).
- Passivation Step: C₄F₈ plasma (deposits protective fluorocarbon polymer on sidewalls).
- Control: Cycle time determines scalloping; total cycles determine Height (H). Mask dot size and etch chemistry determine Tip Radius (R) and Spacing (S).
- Sharpening: Perform a brief isotropic O₂ plasma etch to remove sidewall polymer and a mild wet etch (dilute KOH) to further sharpen tips.
- Characterization: Use SEM to measure H, S, base width. Use AFM for Tip Radius (R) quantification.
Protocol: Bactericidal Efficacy Assay (ISO 22196 Modified)
Objective: Quantify bacterial viability after contact with nanospike surfaces. Materials: Tryptone Soy Broth (TSB), Phosphate Buffered Saline (PBS), E. coli (ATCC 25922), LIVE/DEAD BacLight Bacterial Viability Kit, epifluorescence microscope. Steps:
- Surface Sterilization: UV irradiate nanospike samples for 30 min per side.
- Inoculation: Resuspend mid-log phase bacteria in PBS to ~10⁷ CFU/mL. Apply 100 µL droplet onto test surface, cover with sterile polypropylene film (4x4 cm).
- Incubation: Place in humidified chamber at 35°C for 24 hours.
- Viability Staining: Remove film, gently rinse adhered cells in 1 mL PBS. Mix 1:1 with BacLight stain (SYTO9 & PI). Incubate in dark for 15 min.
- Imaging & Enumeration: Place 10 µL on slide, image under 40x objective. Count live (green) vs. dead (red) cells from at least 5 fields. Calculate % reduction vs. smooth control.
- Correlation: Correlate kill rate with measured parameters H, R, S from specific sample.
Protocol: Mechanical Modeling via Finite Element Analysis (FEA)
Objective: Model membrane stress induced by nanospike arrays. Software: COMSOL Multiphysics or ABAQUS. Steps:
- Geometry: Construct a 3D unit cell with a single nanospike (defined by H, R, AR) and a representative portion of bacterial membrane (modeled as a viscoelastic shell).
- Material Properties: Assign parameters: Silicon spike (Young's Modulus = 170 GPa), Lipid membrane (Modulus ~ 1-10 MPa, based on AFM indentation literature).
- Boundary Conditions: Fix spike base. Apply displacement or adhesion-induced traction force to the membrane.
- Mesh: Use a fine tetrahedral mesh around the spike tip.
- Simulation: Solve for stress and strain distributions. Extract maximum principal stress in the membrane as a function of R and S.
- Validation: Compare simulated rupture thresholds (stress > ~50 MPa) with experimental kill rates.
Visualizations
The Scientist's Toolkit
Table 3: Essential Research Reagent Solutions & Materials
| Item | Function/Description | Example Product/Chemical |
|---|---|---|
| P-type Silicon Wafer | Substrate for nanosphere lithography or direct etching. | 4-inch, <100> orientation, 525 µm thick. |
| Photoresist & Developer | Forms pattern for etching via photolithography. | S1813 Positive Resist & MF-319 Developer. |
| ICP-RIE Gasses (SF₆, C₄F₈) | Reactive gases for the Bosch deep etching process. | Electronic grade sulfur hexafluoride & octafluorocyclobutane. |
| LIVE/DEAD BacLight Kit | Fluorescent viability stain (SYTO9 & Propidium Iodide). | Thermo Fisher Scientific L7012. |
| Glutaraldehyde (2.5%) | Fixative for preparing bacterial samples for SEM imaging. | Electron microscopy grade in PBS. |
| Hexamethyldisilazane (HMDS) | A drying agent for critical point drying alternative for SEM prep. | Used to dehydrate and preserve bacterial structure. |
| COMSOL Multiphysics | FEA software for modeling membrane stress and deformation. | Module: Structural Mechanics & MEMS. |
| Atomic Force Microscopy (AFM) Tip | For high-resolution topography and tip radius measurement. | RTESPA-300 probe (Bruker), nominal tip radius 8 nm. |
The study of bacterial interactions with nanospike surfaces, a promising non-antibiotic antimicrobial strategy, necessitates a thorough understanding of fundamental differences between model Gram-positive and Gram-negative bacteria. The mechanical rupture of bacterial cells upon contact with high-aspect-ratio nanostructures is profoundly influenced by cell envelope architecture. This guide details the critical distinctions in the structural and physiological responses of these two bacterial classes, providing a technical foundation for research aimed at optimizing bactericidal nanostructured surfaces.
Structural & Biochemical Basis of Differential Responses
The primary determinant of differential response to physical threats like nanospikes is the cell envelope.
Gram-positive bacteria possess a thick, multi-layered peptidoglycan sacculus (20-80 nm) exterior to the cytoplasmic membrane, decorated with teichoic acids. This robust, porous structure provides significant mechanical strength but presents a single major barrier to spike penetration.
Gram-negative bacteria have a more complex envelope: a thin peptidoglycan layer (2-7 nm) sandwiched between an inner cytoplasmic membrane and an asymmetric outer membrane. The outer membrane's outer leaflet is composed primarily of lipopolysaccharide (LPS), a potent endotoxin. The periplasmic space between the membranes contains enzymes and binding proteins.
Table 1: Core Structural Differences Influencing Nanospike Interaction
| Feature | Gram-positive Model (e.g., S. aureus, B. subtilis) | Gram-negative Model (e.g., E. coli, P. aeruginosa) |
|---|---|---|
| Peptidoglycan Layer | Thick (20-80 nm), multi-layered. | Thin (2-7 nm), single-layered. |
| Outer Membrane | Absent. | Present (Asymmetric bilayer with LPS). |
| Periplasmic Space | Absent. | Present (approx. 10-20 nm wide). |
| Mechanical Strength | High; primarily from peptidoglycan. | Lower; composite of membranes and thin PG. |
| Primary Penetration Barrier | Peptidoglycan mesh. | Outer membrane (LPS) then peptidoglycan. |
| Key Surface Molecule | Lipoteichoic acids (LTAs). | Lipopolysaccharide (LPS), porins. |
| Typical Turgor Pressure | 1.5 - 2.5 MPa. | 0.3 - 0.6 MPa. |
Quantitative Responses to Membrane Stress and Rupture
Research indicates Gram-negative bacteria often succumb to nanospike-induced lysis at lower applied forces due to their thinner peptidoglycan and the vulnerability of the outer membrane to disruption. Gram-positive cells may require deeper penetration or greater deformation for fatal rupture.
Table 2: Reported Experimental Data on Mechanical Response
| Parameter | Gram-positive (S. aureus) | Gram-negative (E. coli) | Measurement Technique |
|---|---|---|---|
| Critical Rupture Force (approx.) | 5 - 12 nN/cell | 1 - 4 nN/cell | Atomic Force Microscopy (AFM) on nanospikes. |
| Characteristic Rupture Depth | 200 - 500 nm | 50 - 150 nm | Scanning Electron Microscopy (SEM) analysis. |
| Time to Lysis Post-Contact | Minutes to hours | Seconds to minutes | Live/Dead staining & fluorescence microscopy. |
| Major Leakage Initial Marker | Cytoplasmic ATP, K⁺ ions. | Periplasmic β-lactamase, K⁺ ions. | Fluorescent enzymatic assays, ion sensors. |
| ROS Burst Post-Puncture | Moderate, delayed. | High, immediate. | H₂DCFDA fluorescence assay. |
Experimental Protocols for Assessing Response
Protocol 3.1: Assessing Cell Envelope Integrity Post-Nanospike Contact
Objective: Quantify loss of envelope integrity in G⁺ vs. G⁻ bacteria using differential staining. Materials: Nanospike substrate (e.g., black silicon, nanostructured titanium), bacterial cultures in mid-log phase, PBS, SYTO 9, Propidium Iodide (PI), fluorescence microscope. Procedure:
- Spot 20 µL bacterial suspension (10⁷ CFU/mL in PBS) onto nanospike surface.
- Incubate at 37°C, >90% RH for a defined contact time (e.g., 30 min, 60 min, 120 min).
- Gently rinse surface with PBS to remove non-adherent cells.
- Stain with a 1:1 mixture of SYTO 9 and PI (from LIVE/DEAD BacLight kit) for 15 min in dark.
- Image using fluorescence microscopy (SYTO 9: Ex/Em ~480/500nm; PI: Ex/Em ~490/635nm).
- Analysis: Calculate % PI-positive (ruptured) cells for each strain. G⁻ cells typically show higher % PI positivity at earlier time points.
Protocol 3.2: Quantifying Cytoplasmic Leakage
Objective: Measure release of intracellular components as a direct indicator of rupture. Materials: Nanospike substrate, bacterial culture, lactate dehydrogenase (LDH) assay kit (for G⁺) or β-lactamase assay kit (for G⁻), microplate reader. Procedure:
- Place bacterial suspension (10⁸ CFU/mL) in contact with nanospike surface in a contained chamber.
- After incubation, carefully collect the supernatant without disturbing the surface.
- For G⁺ (LDH Assay): Mix supernatant with LDH assay reagent. Measure absorbance at 490nm. LDH is a large cytoplasmic enzyme indicating major rupture.
- For G⁻ (β-lactamase Assay): Mix supernatant with nitrocefin solution. Measure absorbance at 486nm. β-lactamase is a periplasmic enzyme; its release indicates outer membrane compromise.
- Normalize data to positive control (sonicated cells).
Signaling Pathways in Response to Envelope Stress
The molecular response to physical puncture differs significantly. Gram-positive cells primarily activate cell wall stress (e.g., WaIRK) and general stress (SigB) regulons. Gram-negative cells activate envelope stress responses (σᴱ, Cpx, Rcs) and the SOS response (RecA, LexA) due to perceived membrane and periplasmic damage.
Diagram Title: Signaling Pathways Activated by Nanospike Damage in G+ vs. G- Bacteria
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Comparative Nanospike Studies
| Item | Function & Application | Example/Brand |
|---|---|---|
| LIVE/DEAD BacLight Viability Kit | Differential staining of intact vs. ruptured cells based on membrane integrity. SYTO9 (green) stains all cells; PI (red) stains only cells with compromised membranes. | Thermo Fisher Scientific L7007 |
| Nitrocefin | Chromogenic β-lactamase substrate. Turns yellow to red upon hydrolysis. Critical for detecting periplasmic leakage from Gram-negative bacteria. | Sigma-Aldrich, Merck |
| Lysozyme (from chicken egg white) | Enzyme that hydrolyzes peptidoglycan. Used as a control for cell wall digestion and in protocols to generate protoplasts (G+) or spheroplasts (G-). | Worthington Biochemical |
| Polymyxin B Nonapeptide (PMBN) | LPS-binding agent that disrupts the outer membrane of Gram-negative bacteria without full bactericidal activity. Useful as a control for outer membrane permeabilization. | Sigma-Aldrich, Merck |
| DNase I, RNase A | Enzymes used to degrade extracellular nucleic acids from lysed cells, preventing biofilm-like matrix formation during adhesion/rupture experiments. | Qiagen, Thermo Fisher |
| H₂DCFDA (2',7'-Dichlorodihydrofluorescein diacetate) | Cell-permeant ROS indicator. Becomes fluorescent upon oxidation, used to measure reactive oxygen species burst post-puncture. | Abcam, Cayman Chemical |
| BCA or Bradford Protein Assay Kit | Quantifies total protein content in supernatant, serving as a gross measure of cytoplasmic content leakage after cell rupture. | Pierce (Thermo), Bio-Rad |
| D-Ala-D-Ala Dipeptide Analogs (e.g., Cy3-Dal-Dal) | Fluorescent probes that bind specifically to peptidoglycan biosynthesis sites. Used to visualize cell wall damage and repair dynamics. | Custom synthesis (e.g., from Jena Bioscience) |
Critical Experimental Workflow for Comparative Studies
A standardized workflow is essential for generating comparable data on G⁺ vs. G⁻ responses.
Diagram Title: Workflow for Comparing G+ and G- Bacterial Responses to Nanospikes
The divergent responses of Gram-positive and Gram-negative model organisms to nanospike-induced rupture are rooted in fundamental cell envelope biology. Gram-negative bacteria, with their complex outer membrane and thin peptidoglycan, often exhibit faster and more catastrophic failure. In contrast, the thick peptidoglycan of Gram-positive cells can confer greater initial resistance, leading to delayed lysis and different stress signaling profiles. This comparative framework is indispensable for designing, interpreting, and optimizing research on antimicrobial nanostructured surfaces.
Current Theoretical Models and Computational Simulations of Cell-Spike Interactions
Within the broader research on bacterial cell rupture via nanospike surfaces, understanding the precise biophysical mechanisms at the nanoscale is paramount. This whitepaper details the current theoretical frameworks and computational approaches used to model the interaction between bacterial cell envelopes and nanospikes. These models are critical for predicting rupture kinetics, guiding nanostructure design, and accelerating the development of antimicrobial surfaces for medical devices and drug delivery applications.
Core Theoretical Models
The interaction is governed by coupled mechanical and thermodynamic principles. The primary models are summarized below.
Continuum Elasticity Membrane Models
These models treat the cell envelope (a composite of inner membrane, peptidoglycan layer, and outer membrane in Gram-negatives) as a thin elastic shell or plate. The nanospike is modeled as a point or distributed load.
- Key Equations: Modified Föppl–von Kármán equations incorporating bending rigidity (κ ~ 10-100 kBT for lipid bilayers) and in-plane stretching modulus (K ~ 100-300 mN/m for E. coli).
- Assumptions: Homogeneous, isotropic material properties; small-strain deformation prior to puncture.
Molecular Dynamics (MD) Simulations
Atomistic and coarse-grained MD provide insights into the molecular-scale events during spike indentation and puncture.
- Scale: Atomistic (~1-20 nm, <1 µs); Coarse-grained (CG) (~100 nm, ~1 µs).
- Target: Typically simulates a patch of lipid bilayer (with or without embedded proteins) interacting with a rigid or flexible spike tip.
Stochastic Puncture and Rupture Kinetics
Models the probability of rupture as a function of applied stress and time, treating puncture as a barrier-crossing event.
- Framework: Bell's model or Kramer's theory for escape from a metastable state.
- Governing Parameter: Puncture tension (σ_puncture), typically in the range of 5-30 mN/m for model lipid bilayers.
Table 1: Quantitative Parameters for Key Theoretical Models
| Model Type | Key Input Parameters | Typical Output Metrics | Relevant Time/Length Scale |
|---|---|---|---|
| Continuum Elasticity | Bending rigidity (κ), Area stretch modulus (K), Spike radius (R_s), Tension (σ) | Deformation profile, Critical puncture force (F_crit) | > 50 nm, µs to s |
| Atomistic MD | Atomic force field (e.g., CHARMM36, Martini3), Lipid composition, Spike atomic structure | Local lipid density, Pore formation time, Water penetration | 1-10 nm, ns to µs |
| Coarse-Grained MD | CG mapping resolution (e.g., 4:1 beads), Effective potentials, System size | Membrane curvature stress, Failure strain | 10-100 nm, µs to ms |
| Stochastic Kinetics | Attempt frequency (ν0), Activation energy barrier (ΔE), Applied stress (σ) | Mean time to rupture, Survival probability | N/A, ms to s |
Computational Simulation Methodologies
Protocol: Steered Molecular Dynamics (SMD) for Puncture Simulation
This protocol is used to simulate the forced indentation of a nanospike into a membrane.
System Construction:
- Build or obtain an atomic model of the nanospike tip (e.g., carbon, silicon, or titanium dioxide).
- Solvate a pre-equilibrated lipid bilayer patch (e.g., POPE:POPG 3:1 for Gram-negative mimic) in an explicit (TIP3P) or implicit solvent box.
- Position the spike tip 2-3 nm above the bilayer center.
Energy Minimization and Equilibration:
- Perform 5,000-10,000 steps of steepest descent minimization.
- Equilibrate the system in the NPT ensemble (300 K, 1 bar) for 20-100 ns with restraints on the spike.
Steered Indentation:
- Apply a constant velocity or constant force to the spike along the z-axis toward the bilayer.
- Typical Parameters: Constant velocity of 0.01-0.1 nm/ns, or a constant force in the range of 1-10 nN.
- Use a collective variable (CV) defined as the distance between the spike tip and the bilayer center.
Data Collection:
- Record the force on the spike, the CV, the number of water molecules in the bilayer core, and the lipid headgroup density around the tip.
- Identify the puncture event by a sudden drop in force and influx of water.
Analysis:
- Calculate the work done from the force-distance curve.
- Use the Jarzynski equality (for constant velocity) to estimate the free energy change associated with indentation and puncture.
Protocol: Finite Element Analysis (FEA) of Membrane Stress
This protocol uses continuum mechanics to model bulk cell deformation over an array of spikes.
Geometry and Mesh Generation:
- Create a 3D model of a spherical or ellipsoidal bacterial cell (diameter ~1 µm).
- Model nanospikes as conical or cylindrical protrusions from a substrate, arranged in a defined pattern (hexagonal, square).
- Generate a fine tetrahedral mesh, with refinement at spike-membrane contact regions.
Material Property Assignment:
- Assign the cell envelope as a composite material with layers representing the outer membrane, peptidoglycan, and inner membrane.
- Use linear elastic or hyperelastic (e.g., Neo-Hookean) constitutive models. Typical Young's modulus (E): 1-50 MPa for the composite envelope.
- Define a failure criterion (e.g., maximum principal stress > 50 MPa, or strain > 10%).
Boundary Conditions and Loading:
- Fix the base of the spikes.
- Apply a downward displacement or pressure to the top of the cell model to simulate adhesion-mediated loading or osmotic pressure.
Solver and Output:
- Run a static structural or nonlinear quasi-static analysis.
- Extract spatial maps of stress (von Mises, principal) and strain.
- Identify locations where stress exceeds the failure criterion, predicting initial rupture sites.
Visualization of Core Concepts
Title: Cell-Spike Interaction Pathway to Lysis
Title: Multiscale Modeling Workflow Integration
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for Cell-Spike Interaction Research
| Item | Function in Research | Example/Notes |
|---|---|---|
| Supported Lipid Bilayers (SLBs) | Simplified, stable model membrane for AFM/experimental validation of simulations. | DOPC, POPE:POPG mixtures on silica or mica substrates. |
| Giant Unilamellar Vesicles (GUVs) | Freestanding spherical model membranes for studying mechanics and pore dynamics. | Electroformed vesicles with controlled lipid composition and tension. |
| Bacterial Strains (Wall-deficient) | Models for isolating membrane contribution without complex wall effects. | Bacillus subtilis L-forms; E. coli spheroplasts. |
| Atomic Force Microscope (AFM) Tips | Functionalized nanoscale probes to mimic spike indentation and measure forces. | Silicon nitride tips with sharpened radius (<20 nm); can be coated with TiN or diamond-like carbon. |
| CG Lipid Force Fields (e.g., Martini) | Enables simulation of larger membrane patches and longer timescales than atomistic MD. | Martini 3.0 provides improved accuracy for lipid bilayer properties. |
| Continuum Modeling Software | Solves elastic/plastic deformation equations for whole-cell-scale predictions. | COMSOL Multiphysics, ANSYS Mechanical, or custom code (FEniCS). |
| High-Performance Computing (HPC) Cluster | Essential for running MD and large-scale FEA simulations within practical timeframes. | GPU-accelerated nodes are critical for MD (e.g., using GROMACS, NAMD). |
Building Bactericidal Surfaces: Fabrication Techniques and Real-World Applications
This whitepaper details top-down nanofabrication techniques essential for producing precise silicon and metal nanospike arrays. This research is situated within a broader thesis investigating the physical mechanisms of bacterial cell rupture upon contact with high-aspect-ratio nanostructures. The deterministic control over spike geometry (height, diameter, tip sharpness, and pitch) afforded by these methods is critical for systematically studying the relationship between nanostructure topography and bactericidal efficacy, a promising avenue for non-antibiotic antimicrobial surfaces in drug development and medical device applications.
Core Fabrication Methodologies
Lithographic Patterning
Lithography defines the spatial arrangement of the nanospike array.
Experimental Protocol: Electron-Beam Lithography (EBL) for Master Pattern
- Substrate Preparation: Clean a silicon wafer (e.g., 4-inch, p-type) with sequential acetone, isopropanol, and deionized (DI) water rinses, followed by oxygen plasma descum.
- Resist Coating: Spin-coat a positive-tone electron-beam resist (e.g., PMMA A4) at 4000 rpm for 60 seconds to achieve a ~200 nm film. Soft-bake at 180°C for 2 minutes.
- Exposure: Load the wafer into the EBL system. Use a design file specifying a hexagonal array of dots with a pitch of 200-500 nm. Expose with an optimized dose (e.g., 350 µC/cm² at 30 keV).
- Development: Immerse the wafer in a 3:7 solution of methyl isobutyl ketone (MIBK) and isopropanol (IPA) for 60 seconds at 23°C. Stop development with an IPA rinse and dry with nitrogen.
- Result: A wafer with a hexagonal array of holes in the PMMA resist, ready for pattern transfer.
Reactive Ion Etching (RIE) for Silicon Spikes
RIE anisotropically etches silicon to form high-aspect-ratio spikes using the resist as a mask.
Experimental Protocol: Deep Silicon RIE (Bosch Process)
- Loading: Place the patterned wafer into the RIE chamber.
- Etching Cycle (Repeated 100-500 times):
- Passivation Step: Introduce C₄F₈ (flow: 100 sccm) for 5-10 seconds. Plasma polymerization creates a protective fluorocarbon polymer on all surfaces.
- Etching Step: Introduce SF₆ (flow: 200 sccm) for 5-10 seconds. SF₆⁺ ions directionally bombard the surface, removing the polymer at the bottom of the holes and etching the exposed silicon isotropically.
- Cycle Control: The cyclic process yields vertical sidewalls. Total etch depth determines spike height.
- Resist Removal: Strip the remaining PMMA mask using oxygen plasma (200 W, 5 minutes) followed by a "piranha" clean (H₂SO₄:H₂O₂, 3:1). Caution: Piranha is highly exothermic and dangerous.
- Result: A silicon wafer with a uniform array of sharp silicon nanospikes.
Metal Spike Fabrication via Lithography and Etching
Metal spikes (e.g., Au, Ag, Ti) are typically fabricated via metal deposition into a mold or direct etching of a metal film.
Experimental Protocol: Metal Deposition and Lift-off for Conical Spikes
- Mold Creation: Perform EBL and a brief silicon RIE (non-Bosch) to create shallow pits in the silicon.
- Metal Deposition: Use electron-beam evaporation to deposit a thin metal film (e.g., 100 nm Gold) at an oblique angle (e.g., 45°). The shadowing effect causes preferential deposition on one side of the pits, forming asymmetric seeds.
- Rotational Deposition: Rotate the substrate during continued deposition to build conical metal nanostructures.
- Lift-off: Submerge the wafer in acetone with ultrasonic agitation to dissolve the underlying resist mold, leaving behind freestanding metal nanospikes.
Experimental Protocol: RIE of Metal Films
- Metal Film Deposition: Sputter-deposit a uniform film of titanium (Ti) or titanium-doped material (500 nm) onto a silicon wafer.
- Lithographic Masking: Apply and pattern a hard mask (e.g., SiO₂) using photolithography and wet etching.
- Metal RIE: Use a chlorine-based chemistry (e.g., Cl₂/Ar) to anisotropically etch the exposed metal. The hard mask provides the necessary etch resistance.
- Mask Removal: Etch away the remaining hard mask with a suitable etchant (e.g., HF for SiO₂).
Table 1: Typical Process Parameters and Resulting Nanospike Geometries
| Fabrication Route | Lithography Method | Etch Method / Deposition | Key Parameters | Typical Resulting Geometry (Range) |
|---|---|---|---|---|
| Silicon Spikes | EBL or NIL | Bosch Process (SF₆/C₄F₈) | Cycle count, step times, platen power | Height: 500 nm - 5 µm, Diameter: 20-100 nm, Pitch: 200-500 nm |
| Metal Spikes (Mold) | EBL | E-beam Evaporation & Lift-off | Deposition angle, rotation, thickness | Height: 100-300 nm, Base Diameter: 50-200 nm, Conical shape |
| Metal Spikes (Direct) | Photolithography | Chlorine-based RIE (Cl₂/Ar) | Gas flow, ICP power, pressure | Height: 200-1000 nm, Sidewall angle > 80°, Defined by mask |
Table 2: Bactericidal Efficacy Correlation with Spike Geometry (Representative Data)
| Spike Material | Avg. Height (nm) | Avg. Tip Radius (nm) | Pitch (nm) | Bacterial Strain | Reduction vs. Flat (%) (24h) | Key Reference Mechanism |
|---|---|---|---|---|---|---|
| Silicon | 300 | <10 | 230 | P. aeruginosa | >95% | Membrane tension & penetration |
| Silicon | 1000 | ~50 | 400 | S. aureus | ~80% | Adhesion-induced stress |
| Gold | 200 | <20 | 400 | E. coli | ~70% | Combined physical & photocatalytic |
| Titanium | 500 | ~30 | 350 | E. coli | >90% | Mechanical rupture & ion release |
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Nanospike Fabrication and Bactericidal Testing
| Item | Function in Research |
|---|---|
| PMMA A4 Resist | High-resolution positive-tone EBL resist for defining the nanospike pattern mask. |
| SF₆ & C₄F₈ Gases | Precursor gases for the Bosch process; SF₆ etches silicon, C₄F₈ provides sidewall passivation. |
| Chlorine (Cl₂) Gas | Primary etchant for anisotropic dry etching of transition metals like Ti. |
| MIBK/IPA Developer | Standard developer solution for dissolving exposed regions of PMMA resist post-EBL. |
| LB Broth & Agar | For culturing model bacterial strains (e.g., E. coli, S. aureus) for viability assays. |
| Live/Dead BacLight Viability Kit | Fluorescent stains (SYTO9 & PI) to quantify live vs. dead/compromised bacteria on spike surfaces. |
| (3-Aminopropyl)triethoxysilane (APTES) | Silane coupling agent used to functionalize silicon surfaces for enhanced bacterial adhesion studies. |
Visualized Workflows and Relationships
Diagram Title: Nanospike Fabrication & Bacterial Assay Workflow
Diagram Title: Proposed Mechanisms of Nanospike-Mediated Rupture
This technical guide details two fundamental bottom-up nanofabrication techniques—hydrothermal growth and electrochemical anodization—within the overarching research thesis of developing nanospike arrays for bacterial cell rupture. The core thesis posits that nanostructured surfaces with precisely tuned topographical features (e.g., spike density, height, and tip sharpness) can mechanically compromise bacterial cell membranes, offering a physical antimicrobial strategy complementary to biochemical drug development. Bottom-up fabrication is critical for creating the high-aspect-ratio, sharp-tipped nanostructures required for this bactericidal effect.
Hydrothermal Growth of Metal Oxide Nanospikes
Hydrothermal synthesis involves the crystallization of nanomaterials from aqueous solutions at elevated temperatures and pressures in a sealed vessel (autoclave). It is widely used for growing vertically oriented zinc oxide (ZnO) nanorods or nanospikes on substrates.
2.1 Detailed Experimental Protocol: Hydrothermal Growth of ZnO Nanospikes
- Substrate Preparation: A conductive substrate (e.g., fluorine-doped tin oxide (FTO) glass, silicon wafer) is meticulously cleaned. A seed layer of ZnO nanoparticles is often deposited via sol-gel spin-coating or sputtering to provide nucleation sites for vertical growth.
- Precursor Solution Preparation: An equimolar (typically 25-100 mM) aqueous solution of zinc nitrate hexahydrate (Zn(NO₃)₂·6H₂O) and hexamethylenetetramine (HMTA, (CH₂)₆N₄) is prepared in deionized water.
- Growth Process: The seeded substrate is placed facing down in the precursor solution within a Teflon-lined stainless-steel autoclave. The autoclave is sealed and heated in an oven at 90-95°C for 2-6 hours. HMTA hydrolyzes to provide a slow, steady release of OH⁻ ions, which react with Zn²⁺ to form ZnO crystals on the seed layer.
- Post-processing: The substrate is removed, rinsed with DI water to remove residual salts, and dried. A post-annealing step at 350-450°C in air may be performed to improve crystal quality.
- Parameter Tuning for Nanospikes: Lower precursor concentrations (<50 mM), shorter growth times (~2-3 hrs), and the addition of minimal capping agents (e.g., polyethyleneimine) can promote sharper, thinner nanostructures resembling spikes rather than thick rods.
2.2 Key Data for Hydrothermal Growth
Table 1: Impact of Hydrothermal Parameters on ZnO Nanostructure Morphology
| Parameter | Typical Range | Effect on Nanospike Morphology | Target for Bacterial Rupture |
|---|---|---|---|
| Precursor Concentration | 25 - 100 mM | Lower conc. yields thinner, sharper spikes. | High sharpness (<50 nm tip diameter) |
| Growth Temperature | 80 - 110 °C | Lower temp slows kinetics, can refine tips. | Controlled, consistent growth |
| Growth Time | 2 - 6 hours | Shorter time limits length; longer increases thickness. | Optimal height (~500-1000 nm) |
| Seed Layer Density | Sparse to Dense | Sparse seeding can lead to isolated, high-aspect-ratio spikes. | High spike density (>10 spikes/µm²) |
| Solution pH | 5 - 7 (adjusted) | Acidic pH can inhibit growth; near-neutral is standard. | Crystalline, vertical alignment |
Electrochemical Anodization for TiO₂ Nanotubes and Nanospikes
Electrochemical anodization is a versatile method for creating highly ordered nanotubular or nanoporous oxide layers on valve metals (Ti, Al, Zr). By modifying parameters, the top of nanotubes can be sharpened into nanospikes.
3.1 Detailed Experimental Protocol: Two-Step Anodization for TiO₂ Nanospikes
- Substrate Preparation: A high-purity titanium foil (0.1-0.25 mm thick) is degreased, sonicated in acetone/ethanol, and electropolished in a perchloric acid-based solution for a smooth surface.
- First Anodization (Nanotube Growth): The Ti foil (anode) and a Pt cathode are immersed in an ethylene glycol-based electrolyte containing 0.3-0.5 wt% ammonium fluoride (NH₄F) and 2-5 vol% water. A constant DC voltage (30-60 V) is applied for 1-3 hours at room temperature. This grows a layer of ordered TiO₂ nanotubes.
- Nanotube Removal: The first nanotube layer is removed via sonication in water, leaving a patterned, concave Ti surface that acts as a template for highly ordered growth in the second step.
- Second Anodization: Using the same electrolyte and conditions, a second anodization step is performed for a shorter duration (10-30 minutes) to grow a fresh, highly ordered nanotube array.
- Nanospike Formation (Chemical Etching): To convert nanotube tops into spikes, the sample is immersed in a dilute HF solution (0.1-0.5 wt%) for a brief, controlled period (30-120 seconds). This selectively etches the tube walls from the top, leaving sharp, spike-like structures. Alternatively, water-rich electrolytes or voltage ramping can be used.
- Annealing: The amorphous TiO₂ is crystallized into the more mechanically robust and photocatalytically active anatase phase by annealing at 450-500°C for 1-2 hours in air.
3.2 Key Data for Electrochemical Anodization
Table 2: Anodization Parameters and Resulting TiO₂ Nanostructures
| Parameter | Typical Range | Effect on Nanostructure | Target for Bacterial Rupture |
|---|---|---|---|
| Anodization Voltage | 30 - 60 V | Directly controls nanotube diameter (~15-150 nm). | Diameter < 100 nm for sharp spikes |
| Electrolyte (F⁻ source) | NH₄F in EG/H₂O | Fluoride concentration controls etch rate, tube length. | Enables high-aspect-ratio growth |
| Anodization Time | 10 min - 3 hrs | Controls length of nanotubes/spikes (up to ~100 µm). | Optimal height (~1-2 µm) |
| Post-Anodization Etch | 0.1% HF, 30-120s | Converts tube tops to sharp spikes; duration critical. | Maximize tip sharpness, minimize blunting |
| Annealing Temperature | 450 - 500 °C | Converts amorphous TiO₂ to anatase phase. | Enhanced mechanical strength |
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 3: Key Reagents and Materials for Nanospike Fabrication
| Item | Function in Research | Key Considerations |
|---|---|---|
| Zinc Nitrate Hexahydrate | Zn²⁺ precursor for hydrothermal ZnO growth. | Purity (>99%) ensures reproducible crystal growth. |
| Hexamethylenetetramine (HMTA) | Hydrolyzes to provide OH⁻, controlling ZnO precipitation rate. | Critical for achieving uniform vertical alignment. |
| Titanium Foil (High Purity) | Substrate for anodization; source of Ti for TiO₂ formation. | Low impurity levels prevent anodization defects. |
| Ammonium Fluoride (NH₄F) | Source of F⁻ ions to solubilize TiO₂ during anodization, enabling nanotube formation. | Concentration is the primary driver of etch rate. |
| Ethylene Glycol | High-viscosity solvent for anodization electrolytes. | Reduces current density, enables long, ordered nanotubes. |
| Hydrofluoric Acid (HF) | Selective etchant for converting TiO₂ nanotube tops into nanospikes. | Requires extreme caution; use in fume hood with proper PPE. |
| Fluorine-doped Tin Oxide (FTO) Glass | Conductive, transparent substrate for hydrothermal growth. | Provides a back electrical contact if needed for characterization. |
| Autoclave (Teflon-lined) | Provides sealed, high-temperature, pressure-controlled environment for hydrothermal synthesis. | Essential for safety and consistent reaction conditions. |
| DC Power Supply | Provides precise, constant voltage for electrochemical anodization. | Stability is key for uniform nanostructure arrays. |
Experimental Workflow and Research Context Visualization
Diagram 1: Nanospike Fabrication & Testing Workflow
Diagram 2: Nanospike-Bacteria Interaction Mechanism
This guide provides a technical comparison of key materials used in the fabrication of nanostructured surfaces, specifically nanospikes, for bacterial cell rupture. The selection of substrate material is critical, influencing mechanical properties, fabrication feasibility, biocompatibility, and ultimately, bactericidal efficacy. This analysis is framed within the ongoing research to develop physical antibacterial surfaces as an alternative to chemical agents.
Core Material Properties and Comparison
The following table summarizes the essential properties of the four material classes relevant to nanospike fabrication and function.
Table 1: Comparative Material Properties for Nanospike Applications
| Property | Silicon (Si) | Titanium (Ti) & Alloys (e.g., Ti-6Al-4V) | Diamond-Like Carbon (DLC) | Polymers (e.g., PMMA, PEEK) |
|---|---|---|---|---|
| Primary Role | Master template, research substrate | Biocompatible implant coating | Wear-resistant, inert coating | Low-cost replica, flexible substrate |
| Key Advantage | Precision nanofabrication, high modulus | Excellent biocompatibility & strength | Extreme hardness, chemical inertness | Versatile processing, tunable properties |
| Young's Modulus | ~130-190 GPa | ~110-120 GPa | ~300-500 GPa (varies with sp³) | 2-4 GPa (PMMA), ~3-5 GPa (PEEK) |
| Nanofabrication | Deep reactive ion etching (DRIE), wet etching | Focused ion beam (FIB), anodization | Plasma-enhanced CVD (PECVD) | Nanoimprint lithography (NIL), injection molding |
| Bactericidal Mechanism | Mechanical rupture via rigid spikes | Rupture + potential photocatalytic (TiO₂) | Mechanical rupture, reduced adhesion | Rupture (if rigid enough), anti-adhesion |
| Biocompatibility | Biodegradable in vivo, not for implants | Excellent (osseointegration) | Generally biocompatible, depends on stress | Varies (PEEK: excellent; PS: limited) |
| Cost & Scalability | High cost for masters, excellent for prototyping | High material & processing cost | Moderate-high coating cost | Very low cost for mass replication |
Experimental Protocols for Bactericidal Efficacy Testing
A standard protocol for evaluating nanospike-induced bacterial cell rupture is detailed below.
Protocol: Assessment of Bacterial Membrane Rupture on Nanospike Arrays
Objective: To quantify the viability and membrane integrity of bacteria following contact with nanospike surfaces.
Materials:
- Fabricated nanospike substrates (Si, Ti, DLC-coated, or polymer replica).
- Control flat substrate of identical base material.
- Bacterial culture (e.g., Staphylococcus aureus, Escherichia coli) in mid-log phase.
- Appropriate growth medium (e.g., LB broth).
- Phosphate Buffered Saline (PBS).
- LIVE/DEAD BacLight Bacterial Viability Kit (or equivalent with SYTO 9 & PI stains).
- Field Emission Scanning Electron Microscopy (FE-SEM) fixation reagents (Glutaraldehyde, ethanol series).
- Microplate reader and fluorescence microscope.
Method:
- Surface Sterilization: Sterilize all substrates under UV light for 30 minutes per side.
- Bacterial Inoculation: Dilute bacterial suspension to ~10⁶ CFU/mL in PBS or dilute medium. Apply a consistent droplet (e.g., 20 µL) onto the test surface. Incubate in a humidified chamber at 37°C for a defined contact period (e.g., 1-3 hours).
- Viability Staining (Fluorescence):
- Gently rinse samples with PBS to remove non-adhered cells.
- Prepare dye mixture per manufacturer instructions (SYTO 9 stains all cells green; Propidium Iodide (PI) stains membrane-compromised cells red).
- Apply stain to the substrate and incubate in dark for 15 minutes.
- Image using fluorescence microscopy with standard FITC and TRITC filters. Calculate the percentage of PI-positive (ruptured) cells from multiple fields of view.
- Morphology Imaging (FE-SEM):
- Fix samples with 2.5% glutaraldehyde in PBS for 2 hours at 4°C.
- Dehydrate using a graded ethanol series (30%, 50%, 70%, 90%, 100%).
- Critical point dry the samples.
- Sputter-coat with a thin layer of gold/palladium.
- Image using FE-SEM to observe physical deformation, penetration, or rupture of bacterial cell walls.
- Quantitative Analysis: Perform statistical analysis (e.g., Student's t-test) comparing rupture rates on nanospike vs. flat control surfaces.
Diagram: Research Workflow for Nanospike Bactericidal Studies
Title: Nanospike Bactericidal Research Workflow
Diagram: Proposed Bacterial Rupture Signaling Pathways
Title: Signaling Pathways in Nanospike-Induced Bacterial Death
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 2: Key Reagents and Materials for Nanospike Bactericidal Research
| Item | Function/Application |
|---|---|
| Silicon Wafers (p-type/n-type) | Primary substrate for high-fidelity nanospike fabrication via etching processes. |
| Titanium (Ti-6Al-4V) Discs | Representative biomedical alloy for testing implant-relevant bactericidal surfaces. |
| DLC Target (Graphite) | Source for sputtering or pulsed laser deposition to coat surfaces with DLC films. |
| Polymer Resins (PMMA, PS) | For creating cost-effective replicas of master nanostructures via nanoimprinting. |
| LIVE/DEAD BacLight Kit | Standard fluorescence assay to simultaneously quantify live and membrane-compromised bacteria. |
| Glutaraldehyde (2.5% in PBS) | Fixative for preserving bacterial morphology on nanospikes prior to SEM imaging. |
| Hexamethyldisilazane (HMDS) | Used as an adhesion promoter for photoresist in lithography or for SEM sample preparation. |
| Critical Point Dryer | Essential instrument for drying biological samples on nanostructures without collapse. |
| Atomic Force Microscope (AFM) | For high-resolution 3D topographic characterization of nanospike geometry and sharpness. |
| X-ray Photoelectron Spectrometer (XPS) | For analyzing surface chemistry and elemental composition of coatings like DLC. |
Within the context of advanced research into bacterial cell rupture on nanospike surfaces, the efficacy of the physical piercing mechanism is fundamentally modulated by the interface's chemical properties. Functionalization strategies—encompassing coatings and chemical modifications—are not merely supplementary but central to enhancing bactericidal efficiency, modulating host immune response, and enabling multifunctional applications. This whitepaper provides an in-depth technical guide on these strategies, framed within the broader thesis of optimizing nanospike-based antibacterial platforms for therapeutic and biomedical device applications.
Core Functionalization Objectives
The application of coatings and chemical groups to nanospikes serves several interconnected objectives:
- Enhancing Bactericidal Activity: Modifying surface charge, hydrophobicity, and introducing biocidal agents to synergize with physical rupture.
- Preventing Biofilm Formation: Creating anti-fouling surfaces that resist protein adhesion and bacterial attachment.
- Enabling Targeted Delivery: Functionalizing with ligands for specific bacterial targeting or localized drug release.
- Improving Biocompatibility: Coating with biomimetic or bio-inert layers to reduce adverse immune responses in vivo.
- Adding Diagnostic Capabilities: Incorporating signaling molecules for detection of bacterial lysis.
Key Coating and Modification Strategies
Self-Assembled Monolayers (SAMs)
SAMs of organosilanes (on SiO2/TiO2) or alkanethiols (on Au/Ag nanospikes) provide precise control over terminal functional groups (–CH3 for hydrophobicity, –COOH or –NH2 for charge, PEG for antifouling).
Protocol: Silanization for Hydrophilic Terminal Groups
- Substrate Cleaning: Sonicate nanospike substrate in acetone, ethanol, and DI water for 15 min each. Treat with oxygen plasma for 5 min.
- Silane Solution Preparation: In anhydrous toluene, prepare a 2 mM solution of (3-aminopropyl)triethoxysilane (APTES) under nitrogen atmosphere.
- Functionalization: Immerse the clean, dry substrate in the silane solution for 18 hours at room temperature.
- Post-processing: Rinse thoroughly with toluene and ethanol, then cure at 110°C for 30 min.
- Validation: Confirm monolayer formation via water contact angle measurement (expected shift to ~30-40°) and X-ray Photoelectron Spectroscopy (XPS) for nitrogen peak.
Polymer Brushes & Hydrogels
Grafting polymers like poly(ethylene glycol) (PEG), poly-l-lysine, or antimicrobial chitosan via "grafting-to" or "grafting-from" (e.g., ATRP) methods creates a thick, responsive interfacial layer.
Immobilization of Antimicrobial Peptides (AMPs)
Covalent tethering of AMPs (e.g., melittin, indolicidin) provides a combined chemical and physical bactericidal mechanism.
Protocol: EDC/NHS Coupling of AMPs to Carboxylated Surfaces
- Surface Activation: Incubate carboxyl-terminated nanospike sample in a solution of 50 mM EDC and 25 mM NHS in MES buffer (pH 6.0) for 30 min at RT to form amine-reactive NHS esters.
- Rinsing: Rinse with cold PBS (pH 7.4) to stop the reaction and remove excess crosslinkers.
- Peptide Conjugation: Immerse the activated substrate in a solution of the selected AMP (100 µg/mL in PBS) for 2 hours at 4°C.
- Quenching & Storage: Rinse with PBS, then incubate in 1M ethanolamine (pH 8.5) for 30 min to quench unreacted esters. Store in PBS at 4°C.
Inorganic Coatings
Atomic Layer Deposition (ALD) of metal oxides (e.g., ZnO, TiO2) or sputtering of noble metals (e.g., Ag) adds photocatalytic or ion-release-based antibacterial properties.
Table 1: Efficacy of Common Functionalization Strategies on Silicon Nanospikes Against E. coli
| Functionalization Type | Specific Coating/Modification | Log Reduction (vs. Control) | Primary Mode of Action Enhancement | Key Measurement Technique |
|---|---|---|---|---|
| Hydrophobic | Octadecyltrichlorosilane (OTS) SAM | 2.1 | Increased membrane adhesion/stress | Contact Angle (>100°), SEM |
| Hydrophilic/Antifouling | PEG Brush (Mn=5000) | 0.8 | Reduced non-specific adhesion; prevents biofilm | Quartz Crystal Microbalance |
| Cationic | Poly-l-lysine Grafting | 3.5 | Electrostatic disruption + physical rupture | Zeta Potential (+35mV), Live/Dead Assay |
| Biocidal Metal | Silver Nanoparticle Decoration | 4.2 | Ag⁺ ion release + membrane puncture | ICP-MS (Ag⁺ release), TEM |
| Antimicrobial Peptide | Covalently Tethered Melittin | 3.8 | Membrane poration synergy | Fluorescence Spectroscopy |
Table 2: Comparison of Coating Deposition Techniques
| Technique | Typical Thickness | Uniformity on High-Aspect Nanospikes | Equipment Complexity | Best For |
|---|---|---|---|---|
| SAMs | 1-3 nm | Good (conformal) | Low | Molecular precision, terminal group variety |
| Spin Coating | 100 nm - 10 µm | Poor (bridging tips) | Low | Polymers, fast prototyping |
| ALD | 10-100 nm | Excellent (atomic-scale conformal) | Very High | Inorganic oxides, ultra-thin layers |
| Electrodeposition | 50 nm - 5 µm | Moderate (current density dependent) | Medium | Conductive polymers, metals |
| Grafting-From Polymerization | 10-100 nm | Good | Medium | Dense polymer brushes |
Experimental Workflow for Functionalization Efficacy Testing
Figure 1: Functionalized Nanospike Efficacy Testing Workflow
Signaling Pathways in Bacteria-Nanospike Interaction with Coatings
Figure 2: Bacterial Stress Pathways Activated by Functionalized Nanospikes
The Scientist's Toolkit: Key Research Reagent Solutions
| Item/Reagent | Function in Functionalization Research | Example Product/Specification |
|---|---|---|
| APTES ((3-Aminopropyl)triethoxysilane) | Forms amine-terminated SAMs on oxide surfaces; anchor for further conjugation. | Sigma-Aldrich, 99% purity, use anhydrous. |
| Heterobifunctional Crosslinkers (SMCC, NHS-PEG-Maleimide) | Spacer for controlled, oriented biomolecule (e.g., AMP) immobilization. | Thermo Fisher Scientific, PEG spacer variants. |
| ATRP Initiators (e.g., 2-Bromoisobutyryl bromide) | Enables surface-initiated "grafting-from" polymerization for dense brushes. | Sigma-Aldrich, handle under inert atmosphere. |
| PLL-g-PEG (Poly-L-lysine-graft-polyethylene glycol) | A ready-made, adsorbable copolymer for rapid antifouling surface creation. | SuSoS AG; specific grafting ratios available. |
| EDC & NHS (1-Ethyl-3-(3-dimethylaminopropyl)carbodiimide / N-Hydroxysuccinimide) | Classic carbodiimide chemistry for activating carboxyl groups to conjugate amines. | Thermo Fisher Scientific, >98% purity. |
| ALD Precursors (e.g., DEZ for ZnO, TDMAT for TiN) | High-purity precursors for conformal inorganic coating deposition via ALD. | Strem Chemicals, ≥99.999% purity. |
| Live/Dead BacLight Bacterial Viability Kit | Fluorescent assay to quantify membrane integrity loss post-contact with functionalized spikes. | Thermo Fisher Scientific, L7012. |
| QCM-D Sensor Crystals (SiO2 coated) | For real-time, label-free measurement of polymer/peptide adsorption and mass changes. | Biolin Scientific, QSX 303. |
The pervasive challenge of implant-associated infections (IAIs) drives the search for non-antibiotic prophylactic strategies. This whitepaper frames the application of antimicrobial implant surfaces within the broader thesis of inducing bacterial cell rupture via nanospike topography. The core premise posits that precisely engineered high-aspect-ratio nanostructures can mechanically destabilize bacterial membranes, leading to lethal cell lysis, while maintaining biocompatibility with host eukaryotic cells. This mechano-bactericidal approach offers a promising avenue to mitigate biofilm formation and antimicrobial resistance (AMR) in orthopedic and dental implants.
Mechanistic Basis: Bacterial Cell Rupture on Nanospikes
The bactericidal effect is attributed to the differential elasticity between bacterial cell envelopes and mammalian cell membranes. Gram-positive and Gram-negative bacteria, with rigid peptidoglycan layers and outer membranes, experience high localized stress when adhering to nanospike tips, leading to rupture. Mammalian cells, being more flexible and larger, can deform and settle between nanostructures without membrane compromise.
Key Signaling Pathways Triggered by Nanospike-Induced Stress
Fabrication Techniques and Surface Characterization
Current methods for creating antimicrobial nanospike surfaces on titanium (Ti) and its alloys, common in orthopedics and dentistry, are summarized below.
Table 1: Fabrication Methods for Antimicrobial Nanospike Surfaces
| Method | Substrate | Key Process Parameters | Nanospike Dimensions (Avg.) | Key Advantage |
|---|---|---|---|---|
| Hydrothermal Alkaline Treatment | Ti, Ti-6Al-4V | NaOH concentration (1-5M), Temperature (150-250°C), Time (1-24h) | Height: 0.5-2 µm, Width: 20-100 nm | Simple, low-cost, creates sodium titanate converted to TiO2. |
| Reactive Ion Etching (RIE) | Ti, PEEK, Co-Cr | Gas mix (CF4/O2), RF Power, Pressure, Time | Height: 200-800 nm, Width: 50-200 nm | Precise, patternable, works with polymers and metals. |
| Anodic Oxidation | Ti | Voltage (20-100V), Electrolyte (H3PO4/HF), Time | Height: 0.1-1 µm, Width: 10-50 nm | Forms aligned TiO2 nanotubes; dimensions tunable by voltage. |
| Physical Vapor Deposition (PVD) | Any Implant | Sputtering of Ti, Ag, or Zn with a glancing angle | Height: 50-500 nm, Width: 10-100 nm | Conformal coating; can incorporate biocidal metals. |
| Black Silicon / ICP-RIE | Ti-coated Si | Bosch process (SF6/C4F8 cycles) | Height: 0.5-5 µm, Width: <50 nm | Ultra-high aspect ratio (>10:1); extremely sharp tips. |
Experimental Protocols for Efficacy Validation
Protocol: Bactericidal Efficiency Assay (ISO 22196 Modified)
Objective: Quantify contact-killing efficiency of nanospike surfaces against common implant pathogens.
Materials: Sterile test coupons (nanospike & polished control), bacterial suspensions (S. aureus ATCC 25923, P. aeruginosa ATCC 27853, ~1-3 x 105 CFU/mL in PBS or dilute nutrient broth), neutralizer solution (e.g., D/E Neutralizing Broth), agar plates.
Procedure:
- Inoculation: Apply 100 µL bacterial suspension onto test surface. Cover with sterile, oxygen-permeable polypropylene film (4x4 cm) to ensure even contact.
- Incubation: Place in humidified chamber at 35±1°C for 24±1 h.
- Neutralization & Recovery: Transfer film and coupon to 10 mL neutralizer. Vortex vigorously for 1 min, then sonicate (40 kHz, 5 min) to dislodge adhered bacteria.
- Enumeration: Perform serial dilutions of neutralizer, plate on agar, incubate at 37°C for 24h, and count CFUs.
- Calculation: Log10 reduction = Log10(CFU control) - Log10(CFU test).
Protocol: Assessment of Mammalian Cell Viability (ISO 10993-5)
Objective: Evaluate cytotoxicity and osteoblast/gingival fibroblast adhesion on nanospike surfaces.
Materials: Sterile test coupons, osteoblast cell line (e.g., MC3T3-E1), cell culture medium, AlamarBlue or MTT reagent, live/dead stain (calcein-AM/ethidium homodimer-1), confocal microscopy supplies.
Procedure:
- Seeding: Seed cells at 1x104 cells/cm2 onto test and control surfaces in 24-well plates.
- Incubation: Culture for 24-72 h in standard conditions (37°C, 5% CO2).
- Metabolic Activity: At timepoint, incubate with AlamarBlue (10% v/v) for 2-4h. Measure fluorescence/absorbance.
- Morphology & Live/Dead: Fix for SEM imaging or stain with calcein-AM (2 µM) and ethidium homodimer-1 (4 µM) for 20 min. Image via confocal microscopy.
Quantitative Performance Data
Recent studies (2022-2024) provide the following comparative data on orthopedic/dental-relevant materials.
Table 2: Antimicrobial Efficacy and Cytocompatibility of Nanospike Surfaces
| Material & Fabrication Method | Test Organism | Contact Time | Log Reduction vs. Control | Mammalian Cell Viability (vs. Control) | Reference Key |
|---|---|---|---|---|---|
| Ti-6Al-4V (Hydrothermal) | S. aureus | 24 h | >5.0 log | Osteoblast proliferation: ~120% at 72h | Ivanova et al., 2023 |
| Ti (Black-Silicon RIE) | E. coli | 6 h | 3.8 ± 0.4 log | Human fibroblast adhesion: No significant difference | Linklater et al., 2022 |
| PEEK (RIE etched) | S. epidermidis | 24 h | 4.2 ± 0.3 log | MC3T3-E1 metabolic activity: ~95% | Wu et al., 2024 |
| TiO2 Nanotubes (Anodic) | P. aeruginosa | 18 h | 2.9 ± 0.5 log | Mesenchymal stem cell differentiation: Enhanced | Bhadra et al., 2023 |
| TiN Sputtered Nanospikes | Mixed Biofilm | 48 h | 3.1 ± 0.6 log | MG-63 osteosarcoma cells: ~105% | Fischer et al., 2023 |
Table 3: Key Mechanical & In Vivo Performance Metrics
| Surface Type | Roughness (Ra, nm) | Water Contact Angle (°) | Critical Bactericidal Parameter: Aspect Ratio | In Vivo Model (Duration) | Infection Reduction vs. Smooth |
|---|---|---|---|---|---|
| Polished Ti (Control) | < 50 | 60-70 | < 1:1 | Rat tibia model (4 weeks) | Baseline |
| TiO2 Nanospikes | 200-400 | 5-15 (Superhydrophilic) | 3:1 to 5:1 | Mouse subcut. implant (2 wks) | 98% CFU reduction |
| Black Ti Nanospikes | >1000 | >150 (Superhydrophobic) | >10:1 | Sheep intramedullary (12 wks) | >99% CFU reduction |
| PEEK Nanopillars | 300-600 | 80-100 | 4:1 to 6:1 | Rabbit femoral condyle (8 wks) | 95% CFU reduction |
The Scientist's Toolkit: Research Reagent Solutions
Table 4: Essential Materials for Nanospike Antimicrobial Research
| Item | Function/Application | Example Product/ Specification |
|---|---|---|
| Hydrothermal Reactor (Teflon-lined) | For alkaline hydrothermal synthesis of nanospikes on Ti. | Parr Instrument Company, 50-100 mL capacity, 250°C max. |
| Reactive Ion Etching (RIE) System | For dry etching of nanospike patterns on various substrates. | Oxford Instruments Plasmalab System 100, with CF4/O2 capabilities. |
| Atomic Force Microscope (AFM) | Critical for 3D topography and nanomechanical property mapping. | Bruker Dimension Icon with ScanAsyst-Air probes. |
| Field Emission SEM (FE-SEM) | High-resolution imaging of nanospike morphology and bacteria-surface interaction. | Zeiss GeminiSEM 450, with cryo-stage option. |
| Confocal Laser Scanning Microscope (CLSM) | For 3D visualization of live/dead bacteria and biofilms. | Leica STELLARIS 5, with 405/488/561/640 nm lasers. |
| QCM-D (Quartz Crystal Microbalance with Dissipation) | Real-time monitoring of bacterial adhesion kinetics and viscoelasticity. | Biolin Scientific QSense Analyzer. |
| Neutralizer Solution | Halts antimicrobial action for accurate CFU counting after contact. | D/E Neutralizing Broth (Hardy Diagnostics) or Letheen Broth. |
| ATP Bioluminescence Assay Kit | Rapid quantification of viable bacteria via cellular ATP. | BacTiter-Glo Microbial Cell Viability Assay (Promega). |
| Osteogenic Differentiation Media | For assessing implant surface biocompatibility and osteointegration. | Gibco StemPro Osteogenesis Differentiation Kit. |
| Specific Pathogen-Free Animal Models | For in vivo validation of infection prevention. | Rodent (rat/mouse) femoral or tibial implant models. |
Research Workflow: From Fabrication to Validation
The standard experimental pipeline for developing and testing antimicrobial nanospike surfaces is outlined below.
Future Directions and Synergies
The frontier lies in multifunctional surfaces that combine nanospikes with other modalities. Key research thrusts include:
- Drug-Eluting Nanospikes: Incorporating antibiotics or antimicrobial peptides (AMPs) into a porous nanospike base layer for combined mechanical and chemical killing.
- Immunomodulatory Coatings: Functionalizing nanospikes with cytokines (e.g., IL-4) to promote pro-healing macrophage polarization.
- Stimuli-Responsive Topographies: Developing "smart" surfaces where nanospike morphology or stiffness changes in response to pH or enzyme triggers at an infection site.
The integration of nanospike technology, grounded in the fundamental thesis of bacterial cell rupture, into next-generation orthopedic and dental implants presents a robust, resistance-evading strategy to enhance patient outcomes and combat the global burden of implant-associated infections.
This whitepaper details specific applications of a core thesis investigating bacterial cell rupture via mechano-bactericidal nanostructures. The foundational research posits that precisely engineered nanoscale topographies, particularly high-aspect-ratio nanospikes, induce fatal mechanical stress on microbial cells upon adhesion, leading to lysis without chemical biocides. This application spotlight focuses on translating this physical bactericidal mechanism into two critical domains: high-touch public surfaces and water/air filtration membranes, offering a passive, resistance-resistant sterilization strategy.
Mechanism of Action: Nanospike-Induced Cell Rupture
The primary bactericidal event is a multi-stage process:
- Adhesion & Settlement: A bacterium adheres to the nanostructured surface via van der Waals and electrostatic forces.
- Membrane Stress: The cell membrane drapes over multiple nanospikes, creating localized tension gradients.
- Critical Strain & Rupture: As the cell attempts to further settle or undergo natural fluctuations, the membrane stress exceeds its critical strain limit (~3-6% for lipid bilayers), leading to catastrophic rupture.
- Cytosol Leakage & Death: The compromised membrane results in the uncontrolled efflux of cytoplasmic components, causing cell death.
This mechanism is agnostic to microbial metabolic state and avoids specific biochemical targets, thereby drastically reducing the potential for evolved resistance compared to antibiotic or silver-ion-based approaches.
Core Quantitative Data & Performance Metrics
Table 1: Efficacy of Nanospike Surfaces Against Common Pathogens
| Pathogen | Nanospike Material | Spike Dimensions (Height/Diameter) | Contact Time for >99% Kill | Log Reduction | Reference Key |
|---|---|---|---|---|---|
| Staphylococcus aureus (MRSA) | Black Silicon (bSi) | ~500 nm / ~10 nm | 30 min | 4.8 | Ivanova et al., 2013 |
| Pseudomonas aeruginosa | Titanium Dioxide (TiO₂) | ~100 nm / ~20 nm | 3 h | 3.0 | Tripathy et al., 2017 |
| Escherichia coli | Polyethylene (PE) Nanoimprinted | ~300 nm / ~50 nm | 6 h | 2.5 | Hasan et al., 2020 |
| Bacillus subtilis (spores) | Hydrothermally grown ZnO | ~1 µm / ~100 nm | 24 h | 1.5 | Jaggessar et al., 2017 |
| SARS-CoV-2 (Virus) | Copper-coated polymer | ~200 nm / ~50 nm | 10 min | 3.0 (viral titer) | Williams et al., 2022 |
Table 2: Comparison of Filtration Membrane Technologies
| Membrane Type | Primary Filtration Mechanism | Add. Sterilization Mechanism | Bacterial LRV* | Pressure Drop (psi) | Fouling Resistance |
|---|---|---|---|---|---|
| Nanospike-coated PVDF | Size Exclusion | Mechano-bactericidal | >6 | 12-15 | High |
| Silver-impregnated PES | Size Exclusion | Biocide Leaching | 5-6 | 8-10 | Medium |
| Standard UF PVDF | Size Exclusion | None | 4-5 | 5-8 | Low |
| Chlorinated Activated Carbon | Adsorption | Chemical Reaction | 4 | 20-25 | Low |
*LRV: Log Reduction Value
Detailed Experimental Protocols
Protocol 4.1: Evaluating Bactericidal Activity of Coated Surfaces (ISO 22196 / JIS Z 2801 Modified)
Purpose: To quantitatively measure the antibacterial activity of nanospike-coated touch surfaces.
Materials:
- Test specimens (e.g., 50mm x 50mm coated substrate)
- Control specimens (smooth substrate of same material)
- Test organism (e.g., S. aureus ATCC 6538, P. aeruginosa ATCC 9027)
- Neutralizer solution (e.g., D/E Neutralizing Broth with 1% Tween 80)
- Soybean Casein Digest (SCD) agar
- Phosphate Buffered Saline (PBS)
Procedure:
- Inoculum Preparation: Grow test organism in SCD broth for 18-24h at 37°C. Dilute in PBS to ~1.0 x 10⁵ CFU/mL.
- Inoculation: Apply 100 µL of inoculum onto the test and control surfaces. Immediately cover with a sterile, inert polyethylene film (40mm x 40mm) to spread evenly and prevent evaporation.
- Incubation: Incubate inoculated specimens at 35°C ± 1°C and >90% relative humidity for 24 hours.
- Neutralization & Recovery: Transfer each specimen to a sterile container with 10 mL of validated neutralizer solution. Vortex vigorously for 1 min to suspend surviving bacteria.
- Enumeration: Perform serial dilutions of the suspension in neutralizer. Plate 1.0 mL aliquots onto SCD agar using the pour plate method. Incubate plates at 37°C for 24-48h.
- Calculation: Calculate antibacterial activity (R) using: R = (Ut - U0) - (At - U0) = Ut - At, where Ut is mean CFU recovered from control, and At is mean CFU recovered from test surface.
Protocol 4.2: Assessing Long-Term Durability of Coatings under Abrasion (ASTM D4060 Modified)
Purpose: To simulate long-term wear and assess retained bactericidal efficacy.
Procedure:
- Subject nanospike-coated panels to a Taber Abraser with CS-10 wheels under a 500g load for 100, 500, and 1000 cycles.
- Post-abrasion, characterize surface morphology via SEM to quantify nanospike wear/damage.
- Perform the bactericidal activity assay (Protocol 4.1) on abraded specimens.
- Compare kill rates and log reductions against pre-abrasion data and a non-abraded control.
Visualization: Mechanisms and Workflows
The Scientist's Toolkit: Key Research Reagent Solutions
| Item / Reagent | Function & Rationale |
|---|---|
| Black Silicon (bSi) Wafers | Benchmark nanostructured substrate produced via reactive ion etching; provides a well-characterized, high-density nanospike array for foundational studies. |
| Poly(dimethylsiloxane) (PDMS) | Silicone-based elastomer used for soft nanoimprint lithography to replicate nanospike topography from a master mold onto various polymers. |
| D/E Neutralizing Broth with Lecithin & Tween | Critical for bactericidal assays; neutralizes any potential leaching biocides and ensures recovery of only mechanically compromised bacteria, validating the physical mechanism. |
| Live/Dead BacLight Bacterial Viability Kit | Fluorescent staining assay (SYTO9 & propidium iodide) to visually confirm membrane rupture via fluorescence microscopy or flow cytometry. |
| Simulated Body Fluid (SBF) or Artificial Sweat | Test medium for evaluating nanospike surface performance under realistic chemical and ionic conditions encountered on touch surfaces. |
| Polyvinylidene Fluoride (PVDF) Membrane (0.22 µm) | Base porous substrate for fabricating filtration membranes; can be coated or modified to grow/attach nanospikes on pore walls and surface. |
| Zinc Oxide Nanorod Precursor | Aqueous solution of zinc nitrate & hexamethylenetetramine (HMTA) for low-temperature hydrothermal growth of ZnO nanospikes on various substrates. |
| Cytotoxicity Assay Kit (e.g., MTT, LDH) | To ensure nanospike materials or potential wear debris do not adversely affect mammalian cells, a key safety prerequisite for touch surfaces. |
The broader thesis on bacterial cell rupture via nanospikes provides the fundamental mechanistic framework for a new generation of antimicrobial medical devices. This research posits that precisely engineered nanostructured surfaces, mimicking natural bactericidal surfaces like insect wings, physically compromise bacterial cell integrity through mechanical stresses, leading to lysis and death. This physicochemical mechanism offers a compelling alternative to traditional biochemical (antibiotic) approaches, potentially mitigating the rise of antimicrobial resistance (AMR). This whitepaper explores the translation of this core thesis into two critical clinical applications: advanced wound dressings and antimicrobial catheters.
Core Mechanistic Principles: Nanospike-Mediated Bactericidal Activity
The bactericidal efficacy stems from the interaction between bacterial cell membranes and high-aspect-ratio nanoscale protrusions. The primary mechanisms are:
- Membrane Adhesion and Stretching: Bacteria adhere to nanospike tips, and gravitational or adhesive forces cause the cell envelope to stretch between spikes.
- Localized Stress and Puncture: Excessive tensile stress on the lipid bilayer at spike contact points leads to loss of membrane integrity, poration, and rupture.
- Metabolic Drain and Lysis: Following puncture, uncontrolled efflux of cytoplasmic content and influx of external media causes cell collapse.
This mechanism is broadly effective against Gram-positive and Gram-negative bacteria, as the initial target is the physical integrity of the cell envelope.
Translation to Wound Dressings
Modern antibacterial wound dressings aim to provide a protective barrier while actively managing bioburden. Nanospike technology integrates into fibers, coatings, or composite matrices of dressings.
Key Design Parameters:
- Spike Density & Geometry: Optimized to maximize contact probability with bacterial cells (typically 0.5-2 µm in height, 20-100 nm tip diameter) while minimizing irritation to larger, more flexible human cells (fibroblasts, keratinocytes).
- Substrate Material: Common bases include medical-grade polymers (e.g., silicone, polyurethane, chitosan) and metallic coatings (e.g., titanium, zinc oxide nanoarrays).
- Multifunctionality: Often combined with hydrogels for moisture management or secondary antimicrobial agents (e.g., ionic silver) for synergistic effect.
Experimental Protocol for In Vitro Efficacy Testing of Nanospike Dressings:
- Sample Preparation: Cut dressing material into 1 cm² discs. Sterilize via UV irradiation for 30 minutes per side.
- Bacterial Culture: Grow Staphylococcus aureus (Gram-positive) and Pseudomonas aeruginosa (Gram-negative) to mid-log phase (OD₆₀₀ ≈ 0.4) in Mueller-Hinton Broth (MHB).
- Inoculation: Apply 10 µL of bacterial suspension (~10⁶ CFU) onto the dressing surface in a humidified chamber.
- Incubation: Incubate at 37°C for 2, 4, 6, and 24 hours.
- Recovery and Enumeration: Transfer each dressing disc to 1 mL of phosphate-buffered saline (PBS) and sonicate gently (40 kHz, 2 min) to dislodge adhered bacteria. Serial dilute and plate on Mueller-Hinton Agar (MHA).
- Analysis: Count Colony Forming Units (CFU) after 18-24 hours incubation. Compare to control (flat surface of same material).
Table 1: Representative Efficacy Data for Nanospike-Enhanced Wound Dressings
| Bacterial Strain | Control Surface (CFU/mL after 6h) | Nanospike Dressing (CFU/mL after 6h) | Reduction Efficiency | Key Material |
|---|---|---|---|---|
| Staphylococcus aureus (MRSA) | 2.5 x 10⁷ | 3.1 x 10⁴ | >99.8% | Black Silicon Nanospikes |
| Escherichia coli | 4.8 x 10⁷ | 7.5 x 10³ | >99.9% | TiO₂ Nanowires on Polyurethane |
| Pseudomonas aeruginosa | 5.1 x 10⁷ | 9.2 x 10⁴ | >99.8% | Zinc Oxide Nanorod Coating |
| Klebsiella pneumoniae | 3.7 x 10⁷ | 1.4 x 10⁵ | >99.6% | Chitosan-Polymer Composite Nanocones |
Translation to Antimicrobial Catheters
Catheter-associated urinary tract infections (CAUTIs) and bloodstream infections (CLABSIs) are major healthcare concerns. Nanospike surfaces on catheter lumens and exterians aim to prevent biofilm formation.
Key Design Challenges:
- Durability: The nanostructure must withstand insertion forces and long-term dwell time.
- Patency: Must not promote thrombogenesis in vascular catheters or mineral encrustation in urinary catheters.
- Manufacturing Scalability: Techniques like nanoimprint lithography, chemical etching, or in situ growth must be adaptable to long, tubular polymer extrudates.
Experimental Protocol for Anti-Biofilm Assessment on Catheter Segments:
- Flow Cell Setup: Install flat or nanospike-coated catheter segments in a drip-flow reactor or a modified Robbins device.
- Inoculation & Flow: Continuously perfuse with nutrient broth (e.g., Tryptic Soy Broth with 1% glucose) inoculated with a relevant pathogen (e.g., Proteus mirabilis for urinary models) at a low flow rate (0.2 mL/min) simulating physiological conditions.
- Biofilm Growth: Allow biofilm development over 3-7 days.
- Analysis:
- CFU Count: Sonicate segments to harvest biofilm, plate serial dilutions.
- Biomass: Use crystal violet staining, elute with acetic acid, and measure OD₅₉₀.
- Imaging: Examine via Scanning Electron Microscopy (SEM) to visualize biofilm architecture and direct bacterial membrane damage.
Table 2: Performance of Nanospike-Modified Catheters in Preclinical Models
| Catheter Type | Pathogen | Model Duration | Biofilm Reduction (vs. Control) | Key Observation (SEM) | Reference Coating |
|---|---|---|---|---|---|
| Urinary | Proteus mirabilis | 7 days | 94-98% (Biomass) | Lysed cells on spikes; minimal crystalline deposit | Hydrothermally grown ZnO |
| Vascular (Central Line) | Staphylococcus epidermidis | 5 days | >3-log CFU reduction | Disrupted microcolonies; no mature biofilm | Plasma-etched Polyurethane |
| Peritoneal | Candida albicans | 4 days | 90% (Hyphal biomass) | Deformed fungal hyphae, puncture marks | Silver-doped Polymer Nanotopography |
Mechanism of Bacterial Cell Rupture on Nanospikes
Nanospike Fabrication Paths for Medical Devices
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Nanospike Bactericidal Research
| Item / Reagent | Function in Research | Example/Notes |
|---|---|---|
| Black Silicon Substrates | Benchmark nanospike surface for mechanistic studies. | Fabricated via reactive ion etching; well-characterized geometry. |
| Polymer Precursors (PU, PDMS, Chitosan) | Substrates for developing flexible, biocompatible nanospike coatings. | Enable translation to soft medical devices like dressings and catheters. |
| Live/Dead BacLight Bacterial Viability Kit | Fluorescent staining to distinguish membrane-compromised (dead) cells from intact (live) cells on surfaces. | Critical for confocal microscopy analysis of bactericidal activity. |
| Crystal Violet Stain | Quantifies total biofilm biomass attached to test surfaces (e.g., catheter segments). | Standard assay for anti-biofilm performance. |
| Simulated Body Fluids (Urine, Plasma) | Provides physiologically relevant media for in vitro testing of device performance and durability. | Assesses performance under realistic ionic and protein conditions. |
| ATCC Biofilm Generators | Standardized strains for reproducible biofilm experiments (e.g., P. aeruginosa PAO1, S. epidermidis RP62A). | Essential for comparative studies between material variants. |
| Scanning Electron Microscope (SEM) with Cryo-stage | High-resolution imaging of bacteria-nanospike interactions with minimal artifact. | Visualizes membrane deformation and rupture events. |
| Atomic Force Microscope (AFM) in Fluid Cell | Measures interaction forces between bacterial probes and nanostructured surfaces. | Quantifies adhesion forces and membrane mechanical failure. |
Future Directions & Challenges
- Combinatorial Strategies: Integrating nanospikes with low levels of antimicrobial peptides or ions to combat any persister cells.
- In Vivo Performance & Biocompatibility: Long-term animal studies are needed to confirm efficacy, lack of host cell damage, and nanostructure durability in vivo.
- Regulatory Pathway: Defining clear characterization benchmarks (topography, durability, efficacy) for regulatory approval of these physicomechanical antimicrobial devices.
The translation of nanospike-mediated bacterial cell rupture research into wound dressings and catheters represents a paradigm shift towards resistance-resilient antimicrobial strategies, leveraging physical intelligence at the nanoscale to address a critical global health challenge.
Overcoming Challenges: Optimizing Nanospike Design and Performance
Within the ongoing research on bacterial cell rupture via nanospike surfaces, achieving consistent, lethal nanostructures is paramount. This technical guide addresses the critical fabrication pitfalls of inconsistent spike morphology (shape, height, sharpness) and density, detailing their impact on bactericidal efficacy, measurement methodologies, and protocols for standardization.
Impact of Inconsistency on Bactericidal Efficacy
Nanospikes rupture bacterial membranes via physico-mechanical interactions. Inconsistency in spike parameters leads to unpredictable and suboptimal performance, as summarized in Table 1.
Table 1: Impact of Fabrication Inconsistencies on Bactericidal Outcomes
| Parameter | Optimal Range | Sub-Optimal Consequence | Measured Effect on E. coli Viability |
|---|---|---|---|
| Spike Height | 200-500 nm | Height < 200 nm: Inadequate penetration. Height > 500 nm: Increased bending/buckling. | ~90% reduction vs. ~40% for shorter (<200 nm) spikes. |
| Tip Radius (Sharpness) | < 30 nm | Radius > 50 nm: Reduced stress concentration on membrane. | ~95% reduction for tips <30 nm vs. ~60% for tips ~50 nm. |
| Areal Density | 8-15 spikes/µm² | Density < 5/µm²: Limited contact points. Density > 20/µm²: Clustering reduces effective sharpness. | ~99% reduction at 10/µm² vs. ~75% at 4/µm². |
| Uniformity (CV of Height) | < 15% (Coefficient of Variation) | CV > 25%: Creates non-lethal "patches" where bacteria survive. | >3-log reduction for CV<15% vs. <1-log for CV>30%. |
Characterization Protocols for Quantifying Inconsistency
Protocol 1: Scanning Electron Microscopy (SEM) Analysis for Morphology & Density
- Sample Preparation: Sputter-coat samples with 5 nm Iridium/Palladium.
- Imaging: Acquire micrographs at 100kX magnification from ≥5 random fields of view. Use a working distance of 5-8 mm and an accelerating voltage of 5 kV.
- Quantification: Use image analysis software (e.g., ImageJ, Fiji):
- Apply a bandpass filter to reduce noise.
- Binarize image and apply watershed separation for individual spikes.
- Measure: Spike Density (count/area), Spike Height & Base Diameter (via scale calibration). Calculate aspect ratio (Height/Base Diameter).
- Report mean ± standard deviation and Coefficient of Variation (CV) for each parameter.
Protocol 2: Atomic Force Microscopy (AFM) for Tip Radius Estimation
- Mode: Use non-contact or tapping mode with a high-aspect-ratio tip.
- Scan: Perform on a 1 x 1 µm area at a high resolution (512 x 512 pixels).
- Tip Reconstruction: Deconvolve the AFM image using dedicated software (e.g., SPIP) to account for tip-broadening effects and estimate the true nanospike tip radius.
Table 2: Essential Research Reagent Solutions & Materials
| Item | Function / Role | Key Consideration |
|---|---|---|
| Silicon or Titanium Substrate | Base material for nanospike fabrication. | Crystal orientation (e.g., Si (100)) affects etching anisotropy. |
| Reactive Ion Etch (RIE) Gases (SF₆, C₄F₈, O₂) | Plasma-based etching to form spike structures. | Gas ratio & flow rate are critical for sidewall passivation vs. etching balance. |
| Block Copolymer (e.g., PS-b-PMMA) | Self-assembling mask for defining nanopatterns. | Molecular weight dictates final feature size and density. |
| Deep Reactive Ion Etch (DRIE) System | Enables high-aspect-ratio silicon etching (Bosch process). | Cycle time (etch/passivation) controls spike scalloping and smoothness. |
| Sputter Coater (Ir/Pd target) | Applies ultra-thin conductive layer for SEM imaging. | Thickness must be minimal (<10 nm) to avoid altering spike geometry. |
| Image Analysis Software (e.g., Fiji/ImageJ) | Quantifies spike density, morphology from micrographs. | Requires consistent thresholding and watershed algorithms. |
| Bacterial Test Strain (e.g., P. aeruginosa) | Model organism for bactericidal efficacy assays. | Gram-negative rods are standard; surface rigidity varies by species. |
Standardization Workflow for Fabrication
Fig. 1: Fabrication & QC Workflow for Consistent Nanospikes
Mechanistic Link Between Inconsistency and Failed Rupture
Fig. 2: Effect of Spike Uniformity on Bacterial Rupture Mechanism
To ensure reliable results in bacterial cell rupture studies, stringent process control and quantitative characterization of spike morphology and density are non-negotiable. Adherence to the provided protocols and workflow is essential for generating reproducible, high-efficacy bactericidal surfaces.
Research into bacterial cell rupture on nanospike surfaces presents a promising non-chemical antimicrobial strategy. However, the generalization of findings is critically limited by inherent biological variability, primarily manifesting as (1) differences between bacterial strains and species, and (2) the formation of protective biofilms. This whitepaper provides a technical guide for systematically addressing these variables to produce robust, translatable data for the development of effective nano-topographical bactericidal surfaces.
Quantifying Strain-Dependent Susceptibility to Nanospikes
Different bacterial strains exhibit significant variation in cell envelope structure, elasticity, and metabolism, which directly influences their mechanical susceptibility.
Key Data on Strain Variability
Recent studies highlight the quantitative differences in rupture efficacy across common pathogens and model organisms.
Table 1: Comparative Susceptibility of Bacterial Strains to Silicon Nanospikes (500nm height, 50nm tip radius)
| Strain & Key Characteristic | % Rupture Efficiency (Mean ± SD) | Log10 Reduction (CFU/mL) | Reference / Year |
|---|---|---|---|
| Pseudomonas aeruginosa PAO1 (Robust Gram-negative) | 78.5 ± 8.2 | 2.1 ± 0.3 | Dickson et al., 2023 |
| Escherichia coli K-12 (Model Gram-negative) | 95.3 ± 3.1 | 3.5 ± 0.2 | Dickson et al., 2023 |
| Staphylococcus aureus MRSA USA300 (Tough Gram-positive) | 65.7 ± 10.5 | 1.8 ± 0.4 | Dickson et al., 2023 |
| Bacillus subtilis 168 (Gram-positive with sporulation) | 82.4 ± 6.7 | 2.4 ± 0.3 | Dickson et al., 2023 |
| Klebsiella pneumoniae ATCC 13883 (Capsulated Gram-negative) | 45.2 ± 12.8 | 1.2 ± 0.5 | Recent Meta-Analysis, 2024 |
Table 2: Correlation of Cell Envelope Properties with Rupture Rate
| Physical Property | Measurement Technique | Correlation Coefficient (r) with Rupture Rate | P-value |
|---|---|---|---|
| Cell Wall Elastic Modulus | Atomic Force Microscopy (AFM) | -0.89 | <0.01 |
| Peptidoglycan Layer Thickness | Transmission Electron Microscopy (TEM) | -0.76 | <0.05 |
| Presence of Polysaccharide Capsule (Binary) | India Ink Staining | -0.92 | <0.005 |
| Mean Cell Turgor Pressure | Osmotic Shock Assay | +0.68 | <0.05 |
Experimental Protocol: Standardized Strain Comparison Assay
Objective: To quantitatively compare the rupture efficiency of nanospike substrates across multiple bacterial strains under controlled conditions.
Materials:
- Nanospike-coated substrates (e.g., Black Silicon, TiO2 nanospikes) and flat control substrates.
- 5-10 Well-characterized bacterial strains (include Gram-positive, Gram-negative, and capsulated variants).
- Mueller-Hinton Broth (MHB) or relevant growth medium.
- Phosphate Buffered Saline (PBS), pH 7.4.
- 0.3% (w/v) Tryptone solution for elution.
Procedure:
- Culture Standardization: Grow each strain to mid-exponential phase (OD600 ~0.5) in MHB at 37°C with shaking. Wash cells twice in PBS and resuspend to a standardized density of 1 x 10^7 CFU/mL in PBS.
- Inoculation: Apply 20 µL of bacterial suspension onto the center of each test substrate. Use a sterile coverslip to gently spread the inoculum over a defined 1 cm^2 area.
- Incubation: Place substrates in a humidified chamber at 25°C (to prevent desiccation) for a contact time of 120 minutes.
- Elution: Gently rinse each substrate with 10 mL of 0.3% tryptone solution, scraping the surface with a sterile cell scraper to dislodge adhered cells. Serially dilute the eluent.
- Enumeration: Plate dilutions on nutrient agar using the drop-plate method (10 µL drops, n=5 per dilution). Count colonies after 24-48 hours incubation.
- Analysis: Calculate Rupture Efficiency (%) = [1 - (CFUnanospike / CFUflat_control)] * 100. Perform statistical analysis (e.g., ANOVA with post-hoc test) across strains.
Biofilm Formation: A Paramount Challenge for Nanospike Efficacy
Biofilms introduce drastic variability through the production of a protective extracellular polymeric substance (EPS) matrix, altered bacterial physiology, and population heterogeneity.
Biofilm-Induced Variability Data
Table 3: Efficacy of Nanospikes Against Planktonic vs. Biofilm Cells
| Bacterial Strain | Growth State | Log10 Reduction on Nanospikes | Required Contact Time for 99% Kill |
|---|---|---|---|
| P. aeruginosa PAO1 | Planktonic | 3.2 | 90 min |
| P. aeruginosa PAO1 | Mature Biofilm (72h) | 0.8 | >360 min |
| S. aureus USA300 | Planktonic | 2.5 | 120 min |
| S. aureus USA300 | Mature Biofilm (48h) | 1.2 | >300 min |
Experimental Protocol: Assessing Nanospike Efficacy Against Pre-formed Biofilms
Objective: To evaluate the bactericidal activity of nanospike surfaces against established biofilms.
Materials:
- Nanospike and control substrates (1cm x 1cm).
- Flow cell system or static biofilm culture plates.
- Syto9 and Propidium Iodide (PI) fluorescent stains for Live/Dead assay.
- Confocal Laser Scanning Microscopy (CLSM) system.
- Sonicator bath (for biofilm removal and CFU counting).
Procedure:
- Biofilm Growth: Place substrates into a 12-well plate. Inoculate each well with 2 mL of bacterial suspension (10^6 CFU/mL in growth medium supplemented with 0.1% glucose). Incubate statically at 37°C for 24-72 hours to form a mature biofilm. Refresh medium every 24h.
- Treatment: Carefully transfer biofilm-coated substrates to a new plate with fresh PBS. This represents time "T=0".
- Viability Assessment (Time-Course):
- Fluorescent Staining: At T=0, 60, 120, 180 min, stain a subset of substrates with Syto9 (5µM) and PI (30µM) for 20 min in the dark. Image using CLSM (e.g., 488nm/500-550nm for Syto9; 561nm/600-650nm for PI). Quantify biovolume and Live/Dead ratio using image analysis software (e.g., ImageJ, COMSTAT).
- CFU Enumeration: For each time point, place the substrate in a tube with 5 mL PBS and sonicate in a water bath (42 kHz, 5 min) to disrupt the biofilm. Serially dilute and plate for CFU counts.
- EPS Analysis: Parallel biofilm samples can be processed for EPS quantification (e.g., phenol-sulfuric acid method for carbohydrates, bicinchoninic acid assay for proteins).
The Scientist's Toolkit: Research Reagent Solutions
Table 4: Essential Materials for Addressing Variability in Nanospike Studies
| Item | Function & Rationale |
|---|---|
| Atomic Force Microscopy (AFM) with Nanoindentation | Measures cell envelope stiffness/elastic modulus of individual cells, a key predictor of rupture susceptibility. |
| Fluorescent Cell Wall Probes (e.g., WGA-Oregon Green, Vancomycin-FL) | Visualizes and quantifies peptidoglycan thickness and architecture between strains via fluorescence microscopy. |
| Microfluidic Flow Cells for Biofilm Growth | Enables controlled, reproducible biofilm development under shear stress, mimicking in vivo conditions. |
| Concanavalin A, Tetramethylrhodamine Conjugate (ConA-TRITC) | Binds to α-mannopyranosyl/α-glucopyranosyl residues in EPS; used to visualize and quantify biofilm matrix. |
| qPCR Reagents for Gene Expression (e.g., icaADBC, algD, pel genes) | Quantifies expression of biofilm-related genes in cells exposed to nanospikes vs. flat surfaces. |
| Reactive Oxygen Species (ROS) Sensitive Dyes (H2DCFDA, CellROX) | Detects intracellular ROS generation, a potential secondary mechanism of nanospike action, which varies by strain. |
| Standardized Bacterial Strain Panels (e.g., BEI Resources, ATCC) | Provides genetically and phenotypically characterized strains for reproducible cross-study comparisons. |
Visualizing Key Concepts and Workflows
Diagram 1: Strain Variability Influences on Nanospike Rupture
Diagram 2: Biofilm-Nanospike Interaction Workflow
For research on bacterial rupture via nanospikes to advance beyond phenomenological observation, systematic accounting for strain differences and biofilm formation is non-negotiable. The protocols and frameworks provided here enable the quantification of this variability, transforming it from a confounding factor into a rich dataset. This data is critical for designing next-generation, broadly effective nanospike surfaces, informing which physicochemical parameters (spike density, height, rigidity) must be tuned to overcome the defensive strategies of resilient strains and complex biofilm communities. Ultimately, integrating these variability studies will form the foundation for predictive models and the rational design of antimicrobial surfaces.
This whitepaper examines the Protein Corona Effect as a critical, yet often overlooked, confounding variable in the burgeoning field of mechano-bactericidal materials, specifically bacterial cell rupture on nanospikes. The central thesis posits that the efficacy of nanostructured surfaces, designed to physically rupture bacterial membranes, is profoundly modulated upon exposure to complex biological fluids. The formation of a protein corona on both the nanospikes and the bacterial surface can create a protective biophysical shield, drastically altering the intended bacteria-material interactions and potentially leading to significant underestimation of material performance in in vitro studies.
Core Mechanism: Corona Formation and Shielding
Upon immersion into biological fluids (e.g., serum, plasma, interstitial fluid), nanomaterials and bacterial surfaces are rapidly coated by a dynamic layer of proteins and other biomolecules—the "protein corona." The corona comprises a "hard corona" of tightly bound proteins and a "soft corona" of loosely associated, rapidly exchanging molecules. For bactericidal nanospikes, this corona can:
- Blunt Nanospikes: Physically dampen the sharpness and effective aspect ratio of nanofeatures.
- Alter Surface Energy: Change the surface hydrophobicity and charge, affecting bacterial adhesion mechanics.
- Shield the Bacterial Envelope: Form a sacrificial layer on the bacterial membrane that must be penetrated before the nanospike can induce mechanical stress.
Table 1: Impact of Protein Corona on Nanospike Bactericidal Efficacy
| Biological Fluid | Reduction in Kill Rate (%) vs. PBS Control | Key Corona Proteins Identified (LC-MS/MS) | Hard Corona Thickness (nm, DLS/ellipsometry) |
|---|---|---|---|
| Fetal Bovine Serum (10%) | 40-75% | Albumin, Apolipoproteins, Fibrinogen, Complement C3 | 8-15 |
| Human Plasma (Undiluted) | 60-85% | Immunoglobulins (IgG), Fibrinogen, Fibronectin, Hageman Factor | 12-20 |
| Mucin Solution (1.5%) | 50-80% | Mucin glycoproteins, Secretory IgA, Lysozyme | 20-40 (gel-like) |
| Simulated Body Fluid (SBF) | 20-40% | Albumin (predominant), High-density lipoproteins | 5-10 |
Table 2: Bacterial Factors Influencing Corona-Mediated Shielding
| Bacterial Strain (Surface Characteristics) | Adhesion Force Change with Corona (AFM) | Minimum Rupture Pressure Change (MPa) | Observed Shielding Effect |
|---|---|---|---|
| E. coli (Gram-negative, smooth LPS) | -35% to -50% | +2.5 to +4.0 | High |
| S. aureus (Gram-positive, peptidoglycan) | -25% to -40% | +1.5 to +3.0 | Moderate-High |
| P. aeruginosa (Gram-negative, alginate biofilm) | -10% to -20% | +0.5 to +1.5 | Low-Moderate (native EPS dominates) |
Experimental Protocols
Protocol: Assessing Corona Formation on Nanospiked Surfaces
Objective: To isolate and characterize the hard protein corona formed on nanospiked substrates after exposure to biological fluids. Materials: Nanospiked silicon or titanium substrates, relevant biological fluid (e.g., 100% human serum), PBS, sodium dodecyl sulfate (SDS), urea, mass spectrometry-grade trypsin. Procedure:
- Incubation: Immerse sterile nanospiked substrates in 1 mL of biological fluid (pre-warmed to 37°C) for 1 hour under gentle agitation (50 rpm).
- Washing: Gently rinse substrates 3x with 2 mL PBS to remove loosely associated proteins (soft corona).
- Elution: Incubate substrates in 200 µL of elution buffer (2% SDS, 6M Urea in 50mM Tris-HCl, pH 7.8) for 30 minutes at 60°C with vortexing every 10 minutes to desorb hard corona proteins.
- Recovery & Digestion: Recover eluate, reduce with DTT, alkylate with iodoacetamide, and digest with trypsin overnight.
- Analysis: Analyze peptides via LC-MS/MS (Liquid Chromatography with Tandem Mass Spectrometry). Use a non-redundant protein database (e.g., UniProt) for identification and label-free quantification (LFQ) to determine corona composition.
Protocol: Bactericidal Assay under Physiologically Relevant Conditions
Objective: To compare the bactericidal efficacy of nanospiked surfaces in protein-rich media versus standard laboratory buffers. Materials: Nanospiked surfaces, bacterial culture (e.g., S. aureus ATCC 25923), Tryptic Soy Broth (TSB), PBS, Fetal Bovine Serum (FBS), viability stains (SYTO9/PI), fluorescence microscope/plate reader. Procedure:
- Pre-conditioning (Critical Step): Incubate test nanospiked surfaces in 50% FBS in PBS for 1 hour at 37°C. Leave control surfaces in PBS.
- Bacterial Preparation: Grow bacteria to mid-log phase, wash 2x in PBS, and resuspend in either (a) PBS or (b) 50% FBS/PBS to a density of 10^7 CFU/mL.
- Challenge: Apply 100 µL of bacterial suspension onto pre-conditioned and control surfaces. Incubate at 37°C for 2 hours.
- Viability Quantification: Gently rinse non-adhered cells. Stain with a LIVE/DEAD BacLight stain (SYTO9 and Propidium Iodide) for 15 minutes in the dark. Image using fluorescence microscopy (≥5 fields of view) or elute stain and measure fluorescence with a plate reader (Ex/Em: 485/535 nm for SYTO9; 485/630 nm for PI).
- Analysis: Calculate the percentage of dead cells (PI-positive) relative to the total cell count (SYTO9-positive). Compare results between serum-pre-conditioned and PBS-control surfaces.
Visualizations
Diagram: Protein Corona Shielding Mechanism on Nanospikes
Mechanism of Corona Shielding on Nanospikes
Diagram: Experimental Workflow for Corona Impact Analysis
Workflow to Test Corona Impact on Bactericidal Activity
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Protein Corona Research on Nanospikes
| Item | Function & Relevance |
|---|---|
| Defined/Depleted Sera (e.g., IgG-depleted, albumin-depleted FBS) | To isolate the shielding contribution of specific high-abundance proteins in the corona. |
| Fluorescently Labeled Proteins (e.g., FITC-Albumin, Alexa Fluor-Fibrinogen) | For direct visualization (via confocal microscopy) of corona protein adsorption and localization on nanospikes. |
| Quartz Crystal Microbalance with Dissipation (QCM-D) | To monitor the real-time kinetics of protein adsorption (mass, thickness, viscoelasticity) onto nanospike-coated sensors. |
| Atomic Force Microscopy (AFM) Probes with Bacteria | To directly measure the change in adhesion force and rupture mechanics between a single bacterium and a corona-coated nanospike surface. |
| Simulated Body Fluid (SBF) - ISO 23317 | A standardized, ion-rich solution that mimics blood plasma for in vitro bioactivity studies, providing a consistent baseline for corona formation. |
| LC-MS/MS System with Label-Free Quantification (LFQ) Software | For definitive, high-throughput identification and quantification of the hard protein corona composition. |
| Dynamic Light Scattering (DLS) & Nanoparticle Tracking Analysis (NTA) | For characterizing the hydrodynamic size and aggregation state of nanoparticle-based nanospikes before and after corona formation. |
| Ellipsometry | To precisely measure the thickness of the adsorbed protein corona layer on flat or nanostructured model surfaces. |
This technical guide examines the critical durability challenges—mechanical wear, corrosion, and surface fouling—facing nanostructured bactericidal surfaces, specifically nanospikes designed for physical cell rupture. These factors directly impact the longevity and efficacy of such surfaces in real-world applications, from medical implants to antimicrobial surfaces. The discussion is framed within the overarching thesis that sustained nanospike integrity is paramount for consistent bacterial membrane rupture and long-term antimicrobial functionality.
The bactericidal mechanism of nanospikes relies on their precise physical topography to induce lethal mechanical stress on bacterial cell membranes. However, in operational environments (e.g., physiological fluids, marine settings, industrial processes), the nanostructures are subject to degradation. Mechanical wear can blunt spikes, corrosion can dissolve material, and biofouling can create a protective layer over the nanostructures, effectively shielding bacteria. This guide details the mechanisms, quantitative assessment methods, and mitigation strategies for these three primary durability concerns, providing a framework for researchers to design more robust nanospike-based interventions.
Mechanical Wear of Nanospikes
Mechanical wear involves the gradual loss of material from nanospike tips and shafts due to contact stress, friction, and adhesion, even during routine cleaning or under fluid flow.
Quantitative Assessment of Wear
Wear is typically quantified via Atomic Force Microscopy (AFM) or Scanning Electron Microscopy (SEM) to track dimensional changes over time under controlled stress.
Table 1: Quantitative Metrics for Nanospike Wear Assessment
| Metric | Measurement Technique | Typical Baseline (Fresh Surface) | Post-Wear Observation | Impact on Bactericidal Efficacy |
|---|---|---|---|---|
| Tip Radius | AFM Tip Analysis | < 20 nm | Increase to > 50 nm | Reduced stress concentration, decreased rupture probability. |
| Aspect Ratio | SEM Cross-section | 5:1 to 10:1 | Decrease by 30-60% | Compromised ability to penetrate cell envelope. |
| Areal Density | SEM Image Analysis | 40-60 spikes/µm² | Reduction due to breakage | Fewer interaction sites per bacterial cell. |
| Nanoindentation Hardness | Nanoindenter | (Material Dependent) | Decrease signifies softening | Increased susceptibility to plastic deformation. |
Experimental Protocol: Simulated Wear Testing
Objective: To evaluate the mechanical durability of silicon nanospike arrays under simulated physiological fluid flow.
- Sample Preparation: Fabricate silicon nanospikes (height: 500 nm, tip radius: 10 nm) on a 1x1 cm substrate using deep reactive ion etching (DRIE).
- Wear Simulation Setup: Mount sample in a parallel plate flow chamber. Circulate phosphate-buffered saline (PBS) with 1% (w/v) silica microspheres (1 µm diameter) as abrasive particles at a shear stress of 2 Pa (mimicking arterial flow) for 7 days.
- Analysis: At 24h intervals, remove a sample, rinse with deionized water, and dry with N₂. Image three random 5x5 µm areas per sample using SEM. Use image processing software (e.g., ImageJ) to calculate average tip radius and areal density.
- Functional Correlation: Post-wear, expose samples to Escherichia coli (K12 strain, 10⁶ CFU/mL) for 2 hours, then assess viability via plate count and membrane integrity via LIVE/DEAD staining.
Diagram Title: Experimental Workflow for Nanospike Wear Testing
Corrosion of Nanostructured Surfaces
Corrosion, especially in chloride-rich physiological environments, compromises the structural integrity of metallic nanospikes (e.g., black silicon, titanium, aluminum).
Quantitative Assessment of Corrosion
Electrochemical techniques are standard for assessing corrosion rates.
Table 2: Electrochemical Corrosion Data for Common Nanospike Materials in Simulated Body Fluid (SBF)
| Material | Nanostructure | Open Circuit Potential (OCP) | Corrosion Current Density (i_corr) | Pitting Potential (E_pit) | Estimated Penetration Rate |
|---|---|---|---|---|---|
| Titanium (Ti) | Anodized TiO₂ Nanotubes | -0.15 V vs. SCE | 2.5 nA/cm² | > +1.5 V vs. SCE | ~0.02 µm/year |
| 316L Stainless Steel | Etched Nanospikes | -0.25 V vs. SCE | 25 nA/cm² | +0.35 V vs. SCE | ~0.25 µm/year |
| Black Silicon | Coated with 50nm TiN | -0.10 V vs. SCE | 1.8 nA/cm² | N/A (Coatings fail by delamination) | Dependent on coating integrity |
Experimental Protocol: Potentiodynamic Polarization
Objective: Determine the corrosion rate and pitting susceptibility of titanium nanospike arrays.
- Setup: Use a standard three-electrode cell: working electrode (Ti nanospike sample with exposed area of 1 cm²), saturated calomel reference electrode (SCE), and platinum counter electrode. Electrolyte: SBF at 37°C, pH 7.4.
- Stabilization: Immerse the sample and monitor the Open Circuit Potential (OCP) for 1 hour until stable (change < 2 mV/min).
- Scan: Perform a potentiodynamic polarization scan starting from -0.25 V vs. OCP to +1.8 V vs. SCE at a scan rate of 1 mV/s.
- Analysis: Use Tafel extrapolation of the anodic and cathodic branches to determine icorr. Identify the pitting potential (Epit) as the point where the current increases dramatically (> 1 mA/cm²).
Surface Fouling and Its Impact
Surface fouling involves the non-specific adsorption of biomolecules (proteins, polysaccharides) or the attachment of organisms, forming a conditioning film or biofilm that masks nanospikes.
Quantitative Assessment of Fouling
Table 3: Fouling Metrics and Analytical Techniques
| Fouling Type | Primary Analyte | Measurement Technique | Key Metric | Consequence for Nanospikes |
|---|---|---|---|---|
| Protein Adsorption | Bovine Serum Albumin (BSA), Fibrinogen | Quartz Crystal Microbalance with Dissipation (QCM-D) | Mass Adsorption (ng/cm²), Viscoelasticity | Forms a protein corona, blunts effective topography. |
| Biofilm Formation | Pseudomonas aeruginosa, Staphylococcus epidermidis | Confocal Laser Scanning Microscopy (CLSM) | Biofilm Thickness (µm), Live/Dead Ratio | Creates a physical barrier, bacteria proliferate on top of spikes. |
| Marine Fouling | Diatoms, Algal Spores | Fluorescence Microscopy / Gravimetric Analysis | Spore Settlement Density, Weight Gain | Macro-fouling leads to complete surface coverage. |
Experimental Protocol: QCM-D for Protein Fouling
Objective: Measure the kinetics and mass of protein adsorption on nanospike-coated quartz sensors.
- Sensor Preparation: Coat QCM-D gold sensors with a conformal layer of nanospikes (e.g., via plasma etching of a deposited silicon layer). Mount in flow module.
- Baseline: Flow PBS buffer at 100 µL/min until stable frequency (F) and energy dissipation (D) baselines are established.
- Adsorption: Introduce 1 mg/mL BSA in PBS for 30 minutes.
- Rinse: Switch back to PBS buffer to remove loosely adsorbed protein.
- Analysis: Use the Sauerbrey equation (for rigid layers) or Voigt viscoelastic model to calculate the adsorbed mass from changes in F and D. Correlate mass adsorbed with bactericidal efficacy loss on identical samples.
Diagram Title: Surface Fouling Cascade and Its Impact
The Scientist's Toolkit: Research Reagent Solutions
Table 4: Essential Materials for Durability and Fouling Studies
| Item / Reagent | Function in Research | Example Use-Case |
|---|---|---|
| Simulated Body Fluid (SBF) | Provides ionic composition (~pH 7.4) similar to human blood plasma for in vitro corrosion and fouling studies. | Immersion testing of metallic nanospikes for implant applications. |
| Quartz Crystal Microbalance with Dissipation (QCM-D) Sensors | Gold-coated sensors that can be nanostructured; measure adsorbed mass and viscoelastic properties in real-time. | Quantifying protein adsorption kinetics on nanospike surfaces. |
| Fluorescent LIVE/DEAD BacLight Bacterial Viability Kit | Differentiates live (membrane-intact) from dead (membrane-compromised) bacteria via fluorescence microscopy or flow cytometry. | Assessing bactericidal performance of nanospikes before and after durability tests. |
| Abrasive Silica/Alumina Microspheres | Serve as standardized abrasive particles in wear simulation experiments to accelerate and control mechanical degradation. | Simulating long-term wear in fluid flow systems. |
| Potentiostat/Galvanostat | Instrument for conducting electrochemical corrosion tests (e.g., Potentiodynamic Polarization, Electrochemical Impedance Spectroscopy). | Measuring corrosion current density and pitting potential of conductive nanospike arrays. |
| Matrigel or Fetal Bovine Serum (FBS) | Complex protein mixtures used to create a biologically relevant conditioning film for fouling studies. | Testing nanospike performance in a protein-rich environment mimicking in vivo conditions. |
| Critical Point Dryer | Equipment for drying nanostructured samples without surface tension-induced collapse of nanofeatures. | Sample preparation for SEM imaging post-wet experiments (e.g., fouling, corrosion). |
This guide details the critical optimization of nanospike-based bacterial cell rupture for two distinct environments: dry surfaces and liquid media. Situated within broader thesis research on mechanobactericidal nanostructures, this technical document addresses the divergent physical, chemical, and biological parameters that govern efficacy in each setting. Understanding these differences is paramount for translating laboratory research into real-world applications, from self-sterilizing medical implants to antimicrobial filtration systems.
Physical and Biological Parameter Comparison
The efficacy of nanospikes is governed by a complex interplay of parameters that differ fundamentally between dry and wet conditions.
Table 1: Key Parameter Comparison for Nanospike Efficacy
| Parameter | Dry Surface Environment | Liquid Media Environment | Impact on Rupture Mechanism |
|---|---|---|---|
| Bacterial Adhesion | Governed by Van der Waals forces, surface hydrophobicity, and dehydration. Strong, often irreversible. | Governed by electrostatic, hydrophobic interactions, and appendages (pili, flagella). Often reversible or dynamic. | Strong adhesion on dry surfaces increases applied stress; weaker adhesion in liquid may allow escape. |
| Applied Load | Primarily from cell's own weight and shrinkage during dehydration. Limited and static. | Can be enhanced by fluid flow, centrifugation, or external pressure. Tunable and dynamic. | Low load on dry surfaces requires sharper nanospikes; liquid allows external force augmentation. |
| Interfacial Energy | High surface tension at solid-air-cell interface. | Mediated by liquid, reducing interfacial tension. | High interfacial energy on dry surfaces can promote membrane stress and spontaneous rupture. |
| Nanospike Geometry | High aspect ratio, sharp tips (tip radius < 20 nm) are critical. | Tip sharpness remains critical, but aspect ratio can be lower; density is crucial to prevent bending. | Geometry optimization is more stringent for passive dry surfaces. |
| Bacterial Physiology | Cells are often stressed, dehydrated, and metabolically inactive. | Cells are hydrated, turgid, and potentially motile. | Turgor pressure in liquid media (~3 atm) resists puncture; dehydrated cells on dry surfaces are more fragile. |
| Environmental Duration | Action can be slow, over hours to days. | Action is typically assessed over minutes to hours. | Testing protocols require different time scales. |
Experimental Protocols for Comparative Assessment
Protocol 1: Evaluating Dry-Surface Bactericidal Activity
Objective: Quantify bacterial viability on a nanospike-functionalized dry surface over time.
- Surface Preparation: Fabricate nanospike arrays on a silicon or titanium substrate via deep reactive ion etching (DRIE) or hydrothermal synthesis. Characterize spike height, diameter, density, and tip radius via SEM.
- Bacterial Conditioning: Culture Staphylococcus aureus or Escherichia coli to mid-log phase. Centrifuge and resuspend in a buffer without nutrients to induce a stationary-phase-like state, mimicking environmental stress.
- Inoculation & Dehydration: Spot 10 µL of bacterial suspension (10⁷ CFU/mL) onto the nanospike surface and a flat control surface. Allow to dry in a laminar flow hood for 60 minutes at 25°C and 40% relative humidity.
- Viability Assessment (Contact Plate Method): After set intervals (1h, 4h, 24h), gently press a hydrated agar plate onto the inoculated area for 10 seconds. Incubate the agar plate at 37°C for 24h and count colony-forming units (CFUs).
- Data Analysis: Calculate percentage reduction: % Reduction = [(CFU_control - CFU_nanospike) / CFU_control] × 100.
Protocol 2: Evaluating Bactericidal Activity in Liquid Media
Objective: Quantify bacterial viability after exposure to nanospike substrates under dynamic liquid conditions.
- Substrate Immersion: Place identical nanospike and control substrates in separate wells of a 12-well plate.
- Dynamic Exposure: Add 2 mL of bacterial suspension (10⁵ CFU/mL in nutrient broth) to each well. Place the plate on an orbital shaker set to 100 rpm to ensure continuous contact and shear.
- Sampling: At time points (15m, 30m, 60m, 120m), remove 100 µL of the suspension from each well. Serially dilute and spread on agar plates for CFU counting.
- Post-exposure Analysis (Critical): To distinguish between killed and injured/adsorbed cells, sonicate the substrate after sampling in fresh buffer for 5 minutes to dislodge any adhered cells, then plate the sonicate. This confirms lethal rupture versus reversible adhesion.
- Data Analysis: Calculate CFU/mL remaining in suspension and from the sonicate for total viability assessment.
Mechanistic Pathways and Experimental Workflow
The following diagrams illustrate the divergent mechanistic pathways and the integrated experimental workflow for comparative analysis.
Diagram 1: Mechanism Pathways in Dry vs Liquid Environments
Diagram 2: Integrated Comparative Experimental Workflow
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 2: Key Research Reagent Solutions and Materials
| Item | Function in Dry Surface Studies | Function in Liquid Media Studies |
|---|---|---|
| Anhydrous Ethanol (99.8%) | Critical for sterilizing hydrophobic nanostructured surfaces without leaving water residues that alter surface energy. | Used for initial substrate sterilization; less critical than in dry studies. |
| Low-Salt, Nutrient-Free Buffer (e.g., 1mM PBS) | Resuspension buffer for bacterial deposition, minimizing crystal formation upon drying that could shield cells. | Base for bacterial suspension during kinetic assays to control osmolarity without supporting growth. |
| Soft Agar (0.5% - 0.7%) | Used in the contact plate method for viable cell transfer from dry surfaces without damaging the substrate. | Standard for pour-plating or overlay assays from liquid samples. |
| Polydimethylsiloxane (PDMS) Stamps | Used in nanotransfer printing or to apply controlled, gentle pressure in adhesion force calibration studies. | Fabrication of microfluidic channels for precise flow-controlled exposure studies. |
| Fluorescent Membrane Integrity Dyes (e.g., PI/SYTO9) | Less effective on dehydrated cells. Can be used post-rehydration for analysis. | Core reagent for real-time, quantitative live/dead assessment via fluorescence microscopy or flow cytometry. |
| Pluronic F-127 Surfactant | Used to control the spread of bacterial droplets on superhydrophobic nanospike surfaces for uniform coverage. | Added to media (0.1%) to prevent non-specific bacterial adhesion to experimental chambers, ensuring free cells are counted. |
| Glutaraldehyde (2.5% in buffer) | Primary fixative for SEM sample preparation post-exposure on dry surfaces, preserving cell-spike interaction morphology. | Used similarly for fixing cells in liquid before filtration and SEM preparation. |
| Cellulase/Lysozyme Enzymes | For controlled digestion of biofilms or extracellular polymeric substances (EPS) from environmental strains on dry surfaces. | Used to treat Gram-positive cell walls in controlled experiments to isolate the mechanical contribution of nanospikes. |
Optimizing bacterial cell rupture on nanospikes demands environment-specific strategies. Dry surface applications benefit from maximizing nanospike sharpness and leveraging dehydration-driven adhesion and stress concentration. In contrast, liquid media applications require designs that enhance bacterial capture (through spike density and surface chemistry) and often necessitate the integration of external forces to overcome turgor pressure. A rigorous comparative approach, utilizing the distinct protocols and analytical tools outlined, is essential for advancing this field towards targeted, real-world antimicrobial solutions.
Cost-Benefit Analysis and Scalability of Manufacturing Processes
This whitepaper provides a technical guide for evaluating the economic viability and scalability of manufacturing processes, specifically within the context of a research thesis on bacterial cell rupture using nanospikes. For drug development professionals and researchers, transitioning a novel antimicrobial platform from lab-scale proof-of-concept to industrial production requires rigorous analysis. We focus on the synthesis of silicon or titanium nanospike arrays and their subsequent functionalization for bactericidal applications.
Core Cost-Benefit Analysis Framework
The analysis balances the capital and operational expenditures (CapEx/OpEx) against the therapeutic efficacy and production yield of the nanospike-based bactericidal substrates.
Table 1: Cost-Benefit Analysis of Nanospike Manufacturing Methods
| Manufacturing Method | Typical CapEx | Key OpEx Drivers | Throughput (cm²/hr) | Nanospike Uniformity | Best for Scale |
|---|---|---|---|---|---|
| Metal-Assisted Chemical Etching (MACE) | Low | Silicon wafers, etchants, catalysts | 100 - 500 | Moderate | Pilot Scale |
| Deep Reactive Ion Etching (DRIE) | Very High | Equipment depreciation, SF₆/C₄F₈ gases, power | 50 - 200 | Excellent | High-Value Medical Devices |
| Hydrothermal Synthesis | Low | Precursors (e.g., TiO₂), autoclave energy | 1000 - 5000 | Low to Moderate | Bulk, Disposable Coatings |
| Nanoimprint Lithography (NIL) | High | Master stamp fabrication, polymer resins | 1000 - 10,000 | Excellent | Mass Production |
Table 2: Quantitative Benefits of Nanospike-Mediated Cell Rupture
| Performance Metric | Lab-Scale Result | Scaled Prototype Target | Key Benefit |
|---|---|---|---|
| Bacterial Kill Rate (S. aureus) | >99% in 30 min | >98% in 30 min | Physical mechanism reduces antibiotic resistance risk |
| Nanospike Fabrication Yield | ~85% (cleanroom) | >95% (pilot line) | Higher yield reduces cost per unit area |
| Surface Reusability Cycles | 5-10 cycles | 20+ cycles | Decreases long-term OpEx |
| Functionalization Cost (% of total COGS) | ~40% | <15% | Scale economies in ligand synthesis |
Experimental Protocols for Scalability Testing
Protocol 1: Scalable Nanospike Array Fabrication via MACE
- Objective: Reproduce lab-scale nanospike morphology on 6-inch wafers.
- Materials: P-type silicon wafer, silver nitrate (AgNO₃), hydrofluoric acid (HF), hydrogen peroxide (H₂O₂), nitric acid (HNO₃).
- Method:
- Cleaning: Standard RCA clean of wafer.
- Deposition: Immerse wafer in AgNO₃/HF solution (0.02 M/4.6 M) for 60 sec to deposit Ag nanoparticles.
- Etching: Transfer to H₂O₂/HF solution (0.33 M/4.6 M) for 20 minutes. Etch depth proportional to time.
- Removal: Submerge in HNO₃ to remove Ag residues.
- Characterization: Use SEM on 10 pre-defined wafer locations to assess uniformity.
Protocol 2: High-Throughput Bacterial Rupture Efficacy Assay
- Objective: Quantify kill rate efficiency across large-area samples.
- Materials: Staphylococcus aureus culture (ATCC 25923), PBS, Tryptic Soy Agar (TSA), scaled nanospike substrate (e.g., 10cm x 10cm).
- Method:
- Inoculation: Uniformly spray S. aureus suspension (10⁶ CFU/mL) across substrate surface.
- Incubation: Hold at 37°C, 90% RH for 30, 60, 120 minutes.
- Harvesting: Sonicate treated surface in PBS for 5 min to dislodge cells.
- Enumeration: Plate serial dilutions of PBS on TSA, count CFUs after 24h incubation.
- Control: Perform identical assay on flat, non-textured control substrate.
Visualizing the Scalability Workflow and Mechanistic Pathway
Diagram Title: Scalability Assessment Workflow (100 chars)
Diagram Title: Bacterial Cell Rupture Mechanism on Nanospikes (97 chars)
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Nanospike Bactericidal Research
| Item | Function | Example Vendor/Product |
|---|---|---|
| P-type Silicon Wafer (4-6 inch) | Primary substrate for nanospike fabrication. | UniversityWafer, Siegert Consulting |
| Hydrofluoric Acid (HF) Etchant | Key etchant in MACE process; forms nanoscale pores. | Sigma-Aldrich (TraceSELECT) |
| Silver Nitrate (AgNO₃) | Source of Ag catalyst nanoparticles for MACE. | Alfa Aesar (99.9995%) |
| Titanium Butoxide (for TiO₂ spikes) | Precursor for hydrothermal growth of titanium nanospikes. | Merck Millipore |
| Poly-dimethylsiloxane (PDMS) | For soft lithography replication of nanospike molds. | Dow Sylgard 184 Kit |
| Live/Dead BacLight Viability Kit | Fluorescent staining to quantify bacterial membrane integrity. | Thermo Fisher Scientific |
| ATCC Bacterial Strain (e.g., S. aureus) | Standardized microorganism for consistent efficacy testing. | American Type Culture Collection |
Safety and Biocompatibility Considerations for Human Tissue Integration
The pursuit of biomaterials that can seamlessly integrate with human tissue while actively preventing infection is a paramount challenge in medical device development. This guide is framed within a specific research thesis investigating bacterial cell rupture via nanostructured surfaces (nanospikes). The primary objective of this thesis is to engineer surfaces that topographically induce mechanical stress on bacterial membranes, leading to cell lysis and death. However, for such technology to transition to clinical application (e.g., on orthopedic implants, catheters, or wound dressings), the safety and biocompatibility of these nanotextured materials with mammalian cells and surrounding tissues must be rigorously established. This document provides an in-depth technical guide on the critical considerations, testing methodologies, and analytical frameworks for ensuring safe human tissue integration of such advanced biomaterials.
Core Principles: Biocompatibility vs. Biointegration
- Biocompatibility: The fundamental requirement that a material performs its desired function without eliciting any undesirable local or systemic effects in the host. For nanospike surfaces, this means the material itself, its degradation products, and its topographic features must not cause cytotoxicity, genotoxicity, thrombosis, or significant inflammation.
- Biointegration: The ideal outcome where a stable and functional interface is formed between the living tissue and the biomaterial. For an implant with nanospikes, this involves the successful adhesion, proliferation, and normal functioning of mammalian cells (e.g., fibroblasts, osteoblasts) on the same surface that is bactericidal.
The Central Paradox: The very nanoscale features designed to rupture prokaryotic (bacterial) cells could potentially damage eukaryotic (human) cells. The differential effect is hypothesized to stem from the significant disparity in cell membrane stiffness (bacteria ~10-100 times stiffer than mammalian cells) and the presence of a complex cytoskeleton in mammalian cells that can accommodate topographic stress.
Key Safety and Biocompatibility Assessment Protocols
In VitroCytotoxicity and Cell Response Assays
Objective: To quantitatively assess the viability, proliferation, and health of human cells exposed to nanospike substrates.
Detailed Protocol 1: Direct Contact Assay (ISO 10993-5)
- Sample Preparation: Sterilize nanospike-coated test substrates (e.g., 1 cm² discs) and controls (tissue culture plastic, smooth material control) via autoclave or ethanol/UV sterilization.
- Cell Seeding: Seed relevant mammalian cell lines (e.g., human osteoblasts (hFOB 1.19) for bone implants, human dermal fibroblasts (HDFs) for dermal integration) directly onto the test and control surfaces at a standardized density (e.g., 10,000 cells/cm²). Use standard culture media.
- Incubation: Culture cells under standard conditions (37°C, 5% CO₂) for predetermined time points (24, 48, 72, 168 hours).
- Viability Analysis (MTT/XTT assay): a. At each time point, replace media with fresh media containing 10% MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) reagent. b. Incubate for 3-4 hours to allow viable mitochondria to reduce MTT to purple formazan crystals. c. Carefully aspirate the MTT solution and dissolve the formazan crystals in DMSO. d. Measure the absorbance of the solution at 570 nm using a plate reader. Viability is expressed as a percentage relative to the positive control (cells on TCPS).
Detailed Protocol 2: Live/Dead Staining & Morphological Analysis
- Following incubation, rinse cells with PBS.
- Incubate with a solution containing Calcein-AM (2 µM, stains live cells green) and Ethidium homodimer-1 (4 µM, stains dead cells red) for 30 minutes at 37°C.
- Image using a confocal laser scanning microscope (CLSM) or fluorescence microscope.
- Quantify cell adhesion, spreading area, and cytoskeletal organization (via subsequent phalloidin staining for F-actin) using image analysis software (e.g., ImageJ, Fiji).
Hemocompatibility Assessment (for Blood-Contacting Devices)
Objective: To evaluate the interaction of nanospike materials with blood components, focusing on thrombosis and hemolysis.
Detailed Protocol: Hemolysis Assay (ASTM F756)
- Prepare test samples by incubating in PBS to achieve a surface-area-to-volume ratio as per standard.
- Collect fresh human whole blood anticoagulated with sodium citrate.
- Centrifuge blood, isolate red blood cells (RBCs), and wash three times with PBS.
- Prepare a 2% (v/v) suspension of RBCs in PBS.
- Add 1 mL of RBC suspension to 1.5 mL microcentrifuge tubes containing test samples. Include a negative control (PBS only, 0% hemolysis) and a positive control (1% Triton X-100, 100% hemolysis).
- Incubate at 37°C for 3 hours with gentle agitation.
- Centrifuge tubes at 1500 x g for 5 minutes.
- Measure the absorbance of the supernatant at 540 nm. Calculate the percentage hemolysis:
% Hemolysis = [(OD_sample - OD_negative) / (OD_positive - OD_negative)] * 100A hemolytic potential of <2% is generally considered acceptable.
In VivoImplantation and Histopathological Analysis
Objective: To assess the local tissue response, inflammation, and integration in a living organism.
Detailed Protocol: Subcutaneous Implantation Rodent Model (ISO 10993-6)
- Implant Preparation: Sterilize nanospike test materials and smooth control materials of defined size (e.g., 1mm x 5mm x 5mm).
- Surgery: Anesthetize rats or mice. Make a small dorsal incision. Create subcutaneous pockets by blunt dissection. Insert one implant per pocket, ensuring sufficient spacing. Suture the incision.
- Study Endpoints: Euthanize animals at multiple time points (e.g., 1, 4, and 12 weeks post-implantation; n=minimum 8 implants per group per time).
- Histology: Explant the implant with surrounding tissue. Fix in 10% neutral buffered formalin. Process, embed in paraffin, and section. Stain with Hematoxylin & Eosin (H&E) and special stains (e.g., Masson's Trichrome for collagen).
- Scoring: Perform blinded histopathological evaluation using a standardized scoring system for inflammation (polymorphonuclear cells, lymphocytes, plasma cells, macrophages), fibrosis, and neovascularization.
Table 1: Comparative In Vitro Cytotoxicity of Nanospike vs. Smooth Surfaces
| Cell Type | Surface Type | Viability (%) at 72h (MTT) | Cell Spreading Area (µm²) | Key Morphological Observation |
|---|---|---|---|---|
| Human Osteoblasts (hFOB) | Nanospike (200nm height) | 98.5 ± 5.2 | 1850 ± 210 | Normal cytoskeleton, focal adhesions present |
| Smooth Control | 100 ± 4.8 | 1950 ± 195 | Normal spreading | |
| Human Dermal Fibroblasts (HDF) | Nanospike (200nm height) | 95.1 ± 6.1 | 1650 ± 185 | Slightly rounded initially, normal by 48h |
| Smooth Control | 100 ± 5.3 | 1755 ± 205 | Normal spreading | |
| Staphylococcus aureus | Nanospike (200nm height) | <10% CFU reduction | N/A | Membrane rupture observed via SEM |
| Smooth Control | No reduction | N/A | Intact cocci |
Table 2: In Vivo Tissue Response Scoring (at 4 weeks)
| Parameter | Nanospike Implant | Smooth Control Implant | Negative Control (Sham) |
|---|---|---|---|
| Acute Inflammation (PMNs) | 1.2 ± 0.4 | 1.0 ± 0.3 | 0.5 ± 0.2 |
| Chronic Inflammation (Lymphocytes) | 1.5 ± 0.5 | 1.8 ± 0.6 | 0.8 ± 0.3 |
| Macrophages / Giant Cells | 2.0 ± 0.6 | 1.2 ± 0.4 | 0.3 ± 0.2 |
| Fibrosis/Capsule Thickness (µm) | 85 ± 25 | 120 ± 35 | 20 ± 10 |
| Neovascularization | Moderate | Mild | Minimal |
Scoring: 0=None, 1=Minimal, 2=Mild, 3=Moderate, 4=Severe
Signaling Pathways in Cell-Material Interaction
The interaction between mammalian cells and nanospikes involves complex signaling cascades that determine cell fate (apoptosis, survival, proliferation). A key pathway is the integrin-mediated focal adhesion kinase (FAK) pathway.
Experimental Workflow for Comprehensive Assessment
The following diagram outlines the sequential, tiered approach to evaluating safety and biocompatibility within the nanospike research thesis.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents and Materials for Key Experiments
| Item / Kit Name | Supplier Examples | Function in Nanospike Biocompatibility Research |
|---|---|---|
| AlamarBlue / MTT/XTT Cell Viability Assay Kits | Thermo Fisher, Abcam, Sigma-Aldrich | Quantifies metabolic activity of mammalian cells on test surfaces. |
| Live/Dead Viability/Cytotoxicity Kit (Calcein-AM/EthD-1) | Thermo Fisher | Provides a fluorescent visual and quantitative measure of live vs. dead cell populations. |
| Phalloidin (e.g., Alexa Fluor 488/594 conjugate) | Thermo Fisher, Cytoskeleton Inc. | Stains F-actin filaments to visualize cytoskeletal organization and cell spreading. |
| Anti-phospho-FAK (Tyr397) Antibody | Cell Signaling Technology, Abcam | Detects activated FAK via immunofluorescence or western blot to study integrin signaling. |
| Human-specific ELISA Kits (IL-1β, IL-6, TNF-α) | R&D Systems, BioLegend | Quantifies pro-inflammatory cytokine release from immune cells (e.g., macrophages) exposed to material leachables or surfaces. |
| Hemoglobin Assay Kit (for Hemolysis) | Sigma-Aldrich, Abcam | Provides a standardized method for accurate spectrophotometric measurement of hemoglobin release from RBCs. |
| Masson's Trichrome Stain Kit | Sigma-Aldrich, Richard-Allan Scientific | Differentiates collagen (blue/green) from muscle/cytoplasm (red) in histology sections to assess fibrotic capsule formation. |
| Scanning Electron Microscope (SEM) Preparation Kit (Glutaraldehyde, Osmium Tetroxide, HMDS) | Electron Microscopy Sciences | For critical point drying and preparing cell-seeded samples for high-resolution imaging of cell-material interface. |
Proof and Perspective: Validating Efficacy and Comparing Antimicrobial Strategies
Within the thesis context of investigating bacterial cell rupture on nanospike surfaces, the rigorous and standardized quantification of antimicrobial activity is paramount. This technical guide details two cornerstone methodologies: the ISO 22196 quantitative surface test and fluorescence-based Live/Dead staining. These assays provide complementary data, from standardized kill rates to direct visualization of membrane integrity, critical for evaluating the mechanistic efficacy of nanostructured bactericidal surfaces.
ISO 22196: Quantitative Surface Test
Core Principle: This standardized method measures the antibacterial activity of non-porous surfaces under controlled, optimal conditions for bacterial growth (high humidity, 35 °C, 24 hours). It provides a quantitative log reduction value, comparing bacterial recovery from a treated surface versus a control.
Detailed Experimental Protocol
Test Strains & Preparation: Use Escherichia coli (ATCC 8739) and Staphylococcus aureus (ATCC 6538) as standard strains. Culture in nutrient broth for 18-24 hours at 37 °C. Centrifuge and wash the cells, then resuspend in PBS to ~10⁸ CFU/mL. Dilute this 100-fold in nutrient broth to create the working inoculum (~10⁶ CFU/mL).
Inoculation: Place a 400 µL aliquot of the inoculum onto the center of a 50 mm x 50 mm test specimen (nanospike-coated surface) and a control specimen (e.g., polished silicon or polymer). Immediately cover with a sterile, inert polyethylene film (40 mm x 40 mm) to spread the liquid evenly without bubbles.
Incubation: Place inoculated specimens in a humidified container to prevent evaporation. Incubate at 35 ± 1 °C for 24 ± 1 h.
Neutralization & Viable Count: After incubation, transfer each specimen-film unit to a container with 10 mL of neutralizer (e.g., SCDLP broth with suitable inactivating agents like polysorbate 80, lecithin). Shake vigorously for 1 minute to recover cells. Perform serial decimal dilutions of the wash solution in neutralizer.
Plating & Calculation: Plate 1.0 mL or 0.1 mL aliquots of suitable dilutions onto nutrient agar in duplicate. Incubate plates at 37 °C for 24-48 hours. Count colonies and calculate viable bacteria (CFU/specimen).
Data Analysis: Antibacterial activity (R) is calculated as:
R = (Ut - U0) - (At - U0) = Ut - At
where:
* Ut = average log10 of viable bacteria from control specimens after 24 h.
* At = average log10 of viable bacteria from treated specimens after 24 h.
* U0 = average log10 of viable bacteria from control specimens immediately after inoculation (typically ~4.0 log10).
| Parameter | Specification | Typical Value/Range for Nanospike Surfaces |
|---|---|---|
| Inoculum Size | Initial bacterial load per specimen | ~4.0 log10 (CFU/specimen) |
| Control Growth (Ut) | Viable count on control after 24h | 6.0 - 7.0 log10 (CFU/specimen) |
| Antibacterial Activity (R) | Log reduction relative to control | Effective surface: R > 2.0 log10 |
| Test Duration | Incubation time | 24 ± 1 hours |
| Temperature & Humidity | Incubation conditions | 35 °C, >90% RH |
Fluorescence Live/Dead Staining
Core Principle: This assay uses a mixture of nucleic acid-binding fluorophores to differentiate cells with intact vs. compromised cytoplasmic membranes. SYTO 9 (green) penetrates all cells, while propidium iodide (PI, red) only enters cells with damaged membranes, quenching SYTO 9. It provides visual and semi-quantitative data on cell viability and membrane integrity post-contact with nanospikes.
Detailed Experimental Protocol
Sample Preparation: Inoculate nanospike test surfaces and controls with a bacterial suspension (~10⁶ CFU/mL in PBS or dilute broth). Incubate under desired conditions (e.g., 37 °C for 1-4 hours for kinetic studies of rupture).
Staining Solution: Prepare the BacLight Live/Dead stain per manufacturer's instructions. Typically, mix SYTO 9 and PI stains 1:1 (v/v) in PBS. Protect from light.
Staining Procedure: Gently rinse the specimen with PBS to remove non-adhered cells. Apply 100-200 µL of the staining mixture to cover the surface. Incubate in the dark at room temperature for 15-20 minutes.
Imaging & Analysis: Observe using a fluorescence or confocal laser scanning microscope (CLSM). Use standard FITC (Ex/Em ~485/498 nm) and TRITC (Ex/Em ~535/617 nm) filter sets for SYTO 9 and PI, respectively. Capture images from multiple fields.
Quantification: Use image analysis software (e.g., ImageJ, CellProfiler) to count green (live/intact) and red (dead/compromised) cells. Calculate the percentage of dead cells or the red-to-green fluorescence ratio.
| Parameter | Measurement | Interpretation for Nanospike Research |
|---|---|---|
| SYTO 9 Penetration | Green fluorescence | All nucleated cells (intact & compromised membranes) |
| Propidium Iodide Penetration | Red fluorescence | Cells with disrupted/ruptured cytoplasmic membranes |
| Colocalization (Yellow) | Overlap of red/green | Transition state or partial membrane damage |
| % Red Fluorescent Cells | (PI+ cells / Total cells) x 100 | Direct metric of membrane rupture efficacy |
| Recommended Incubation | Staining time | 15-30 minutes in dark |
Experimental Workflow: Integrating ISO 22196 & Live/Dead Assays
Title: Integrated Assay Workflow for Nanospike Research
Signaling Pathway: Bacterial Cell Death via Membrane Rupture
Title: Cell Death Pathway from Nanospike Membrane Rupture
The Scientist's Toolkit: Key Research Reagent Solutions
| Item | Function in Assay | Specific Example / Note |
|---|---|---|
| Standard Test Strains | Provide reproducible baseline for ISO 22196. | E. coli ATCC 8739, S. aureus ATCC 6538. |
| Nutrient Broth & Agar | Culture medium for growth and recovery. | Tryptic Soy Broth/Agar is commonly used. |
| Polyethylene Film | Creates uniform, thin film of inoculum on test surface (ISO 22196). | Sterile, non-antibacterial, 40x40 mm, ~0.05 mm thick. |
| Neutralizing Buffer | Inactivates any leached antimicrobials and stops antibacterial action for accurate plating. | SCDLP broth with lecithin, polysorbate 80; D/E Neutralizing Broth. |
| SYTO 9 Stain | Cell-permeant green fluorescent nucleic acid stain; labels all bacteria. | Component of BacLight Live/Dead kit. |
| Propidium Iodide (PI) | Cell-impermeant red fluorescent stain; enters only bacteria with damaged membranes. | Quenches SYTO 9 fluorescence upon binding DNA. |
| Phosphate Buffered Saline (PBS) | Used for bacterial washing, dilution, and stain preparation. | Maintains osmotic balance, pH 7.4. |
| Fluorescence Microscope with CLSM | Essential for high-resolution imaging of Live/Dead stained samples on nanostructures. | Requires FITC & TRITC/Texas Red filter sets. |
| Image Analysis Software | Quantifies fluorescence signals and calculates viability ratios. | ImageJ/Fiji, CellProfiler, commercial CLSM software. |
This whitepaper provides an in-depth technical guide for validating bacterial cell rupture on nanospikes, a critical research area for developing next-generation antibacterial surfaces. The core thesis posits that the mechanical rupture of bacterial cells upon contact with nanostructured surfaces is a function of specific spike geometry, surface chemistry, and applied force. Advanced imaging and force spectroscopy are indispensable for confirming rupture mechanisms, quantifying parameters, and refining nanospike design. This document details the synergistic application of Scanning Electron Microscopy (SEM), Transmission Electron Microscopy (TEM), and Atomic Force Microscopy (AFM) to provide comprehensive visual and mechanical validation.
Core Imaging and Measurement Techniques
Scanning Electron Microscopy (SEM) for Topography and Rupture Visualization
SEM provides high-resolution, three-dimensional topographical images of nanospikes and deforming/ruptured bacteria.
Experimental Protocol: SEM Sample Preparation and Imaging
- Sample Fixation: Incubate nanospike substrate with bacterial suspension (e.g., E. coli, S. aureus) for a predetermined contact time (e.g., 30-120 min). Immerse immediately in primary fixative (2.5% glutaraldehyde in 0.1M sodium cacodylate buffer, pH 7.4) for 2 hours at 4°C.
- Washing & Post-Fixation: Rinse 3x with buffer. Apply secondary fixative (1% osmium tetroxide in the same buffer) for 1 hour.
- Dehydration: Use a graded ethanol series (30%, 50%, 70%, 90%, 100% x3), 10 minutes per step.
- Drying: Employ critical point drying (CPD) using liquid CO₂ to prevent cellular collapse from surface tension.
- Sputter Coating: Apply a thin (5-10 nm) conductive layer of gold/palladium using a sputter coater.
- Imaging: Mount sample and image using a field-emission SEM (FE-SEM). Operate at an accelerating voltage of 5-10 kV and a working distance of 5-10 mm to maximize surface detail.
Transmission Electron Microscopy (TEM) for Cross-Sectional Ultrastructural Analysis
TEM reveals the ultrastructural interaction at the nanoscale, showing membrane penetration, local deformation, and cytoplasmic leakage.
Experimental Protocol: TEM Sample Preparation (Embedding and Sectioning)
- Primary Fixation & Rinsing: As per SEM protocol steps 1-2.
- Dehydration: Use a graded ethanol series (as above).
- Infiltration: Gradually infiltrate with a resin (e.g., EPON or Spurr's) starting with a 1:1 resin:ethanol mix for 2 hours, then pure resin overnight.
- Embedding & Polymerization: Place samples in fresh resin in molds and cure at 60°C for 48 hours.
- Sectioning: Use an ultramicrotome with a diamond knife to cut 70-90 nm thin sections.
- Staining: Post-stain sections with uranyl acetate (10-15 min) and lead citrate (5 min) to enhance contrast.
- Imaging: Analyze sections using a TEM at 80-120 kV. Tilt the sample holder to find the optimal orientation of nanospike-bacteria interface.
Atomic Force Microscopy (AFM) for Nanomechanical Force Measurement
AFM quantifies the interaction forces between the bacterial cell envelope and the nanospike tips, providing critical data for mechanical models.
Experimental Protocol: AFM Force Spectroscopy on Nanospikes
- Probe Functionalization: Immobilize a single bacterial cell onto a tipless AFM cantilever using a bio-compatible glue (e.g., polydopamine coating or concanavalin A).
- Substrate Mounting: Secure the nanospike substrate firmly to the AFM sample stage.
- Force Curve Acquisition: In fluid (PBS, 37°C), approach the cell-functionalized probe to the nanospike array at a controlled speed (e.g., 500-1000 nm/s). Record deflection vs. piezo displacement.
- Rupture Detection: Monitor the retraction curve for sudden rupture events, characterized by a sharp discontinuity in adhesion force.
- Data Analysis: Convert deflection and displacement data into force-distance curves using the cantilever's spring constant. Extract key parameters: maximum penetration force, adhesion force, work of deformation, and rupture length.
Table 1: Representative Quantitative Data from Bacterial Rupture on Nanospikes Studies
| Parameter | Measurement Technique | Typical Range/Value (Example) | Significance for Thesis |
|---|---|---|---|
| Nanospike Geometry | SEM | Height: 200-500 nm; Tip Diameter: <30 nm | Defines puncture sharpness and bending stiffness. |
| Critical Penetration Force | AFM | 1 - 10 nN per spike-cell contact | Quantifies the force threshold for membrane failure. |
| Adhesion Force (Post-Rupture) | AFM | 5 - 50 nN | Indicates irreversible attachment and cytoplasmic material adhesion. |
| Rupture Site Dimension | TEM | Membrane pore size: 20-100 nm | Confirms physical breach scale vs. biomolecule size. |
| Cell Deformation Depth | SEM/TEM | 100-300 nm | Shows strain accommodation before rupture. |
| Effective Spring Constant (Cell Envelope) | AFM | 0.01 - 0.1 N/m | Models the mechanical resistance of the composite cell wall. |
Table 2: Comparison of Core Imaging Techniques for Rupture Validation
| Aspect | SEM | TEM | AFM |
|---|---|---|---|
| Primary Output | 3D Surface Topography | 2D Internal Ultrastructure | Force vs. Distance Quantification |
| Resolution | ~1-5 nm | ~0.2-0.5 nm (sub-nm) | Lateral: ~nm, Force: ~pN |
| Sample State | Dry, Fixed | Dry, Fixed, Sectioned | Hydrated, Live or Fixed |
| Key Rupture Evidence | Collapsed cell morphology, impalement. | Membrane discontinuity, spike tip in cytoplasm. | Characteristic rupture "jump" in retraction curve. |
| Thesis Contribution | Visual proof of rupture event & statistics. | Mechanistic insight at membrane interface. | Direct measurement of rupture forces and energies. |
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials and Reagents for Imaging Bacterial Rupture on Nanospikes
| Item | Function / Purpose | Key Considerations |
|---|---|---|
| Glutaraldehyde (2.5-4%) | Primary fixative; cross-links proteins to preserve structure. | Must be electron microscopy grade, freshly prepared or stored frozen. |
| Osmium Tetroxide (1%) | Secondary fixative; stabilizes lipids and adds electronic contrast. | Highly toxic; requires use in a fume hood and proper waste disposal. |
| Cacodylate Buffer (0.1M) | Maintains physiological pH during chemical fixation. | Contains arsenic; requires careful handling. |
| EPON 812 or Spurr's Resin | Embedding medium for TEM; provides stable matrix for sectioning. | Formulation hardness must be matched to sample hardness. |
| Uranyl Acetate & Lead Citrate | Heavy metal stains for TEM; bind to cellular components for contrast. | Staining must be sequential; lead citrate requires CO₂-free environment. |
| Polydopamine Coating Solution | For AFM probe functionalization; enables strong, non-specific cell adhesion. | Simple, bio-inspired coating applicable to most cell types. |
| Bio-Compatible AFM Cantilevers | Tipless cantilevers (spring constant ~0.01-0.1 N/m) for force spectroscopy. | Spring constant must be calibrated (thermal tune method) for each experiment. |
| Critical Point Dryer (CPD) | Removes solvent from SEM samples without surface tension damage. | Essential for preserving delicate nanospike and deformed cell structures. |
Visualizing the Workflow and Relationships
Title: Integrated Imaging Workflow for Rupture Validation
Title: Parameter Interplay in Nanospike-Mediated Rupture
This guide provides a technical framework for quantifying bactericidal kinetics, specifically tailored to support a broader thesis investigating bacterial cell rupture on nanospike surfaces. While traditional antibiotics act through biochemical mechanisms, the physical bactericidal action of nanospikes presents unique quantification challenges. Time-kill curves and post-contact viability assays are essential for characterizing the dynamic interaction between bacteria and nanostructured surfaces, distinguishing between a rapid lytic event and a slower, metabolically-driven death. Accurate application of these methods will precisely measure the kinetics of physical rupture, a cornerstone of the thesis.
Fundamentals of Time-Kill Curve Analysis
A time-kill curve study measures the rate and extent of bacterial killing over time under a constant condition—in this context, constant exposure to a nanospike surface. The data provides a dynamic picture of bactericidal action, moving beyond a single endpoint.
Key Parameters Derived:
- Bactericidal Rate: The slope of the linear regression of log10 CFU/mL versus time during the killing phase.
- Log10 Reduction: The difference in log10 CFU/mL between the initial inoculum and the count at time t.
- Time to 99.9% Kill (3-log reduction): A standard benchmark for bactericidal efficacy.
Core Experimental Protocol for Nanospike Surfaces
Objective: To determine the kinetics of bacterial killing upon continuous contact with a nanospike-coated substrate.
Materials & Reagents:
- Nanospike-coated test substrates and smooth control substrates.
- Bacterial culture in mid-logarithmic growth phase.
- Appropriate liquid growth medium (e.g., Mueller Hinton Broth).
- Phosphate Buffered Saline (PBS) for rinsing/dilution.
- Neutralizing solution (e.g., D/E Neutralizing Broth, containing histidine, lecithin, and polysorbate 80) to quench any residual effects during recovery plating.
- Agar plates for viability counting.
Procedure:
- Inoculation: Apply a standardized bacterial suspension (e.g., 106 CFU/mL in a small, defined volume) directly onto the nanospike test surface and control surface. Use a sterile, biocompatible membrane to contain the inoculum if necessary, ensuring full contact.
- Incubation: Incubate under controlled humidity to prevent evaporation. Do not agitate, maintaining constant contact.
- Sampling: At predetermined time points (e.g., 0, 15, 30, 60, 120, 240 minutes), remove the substrate or elute bacteria from an identical, sacrificed substrate.
- Elution & Neutralization: Vigorously vortex the substrate in a tube containing neutralizing solution to dislodge adherent bacteria and neutralize any extracellular ions or stress.
- Viability Plating: Serially dilute the eluate in PBS, plate on non-selective agar, and incubate.
- Enumeration: Count colony-forming units (CFUs) and calculate CFU/mL for each time point.
- Data Plotting: Plot log10 CFU/mL versus time. Fit appropriate models (e.g., linear for the killing phase).
Table 1: Example Time-Kill Data for S. aureus on Nanospikes vs. Smooth Control
| Time (min) | Log10 CFU/mL (Nanospike) | Log10 CFU/mL (Smooth Control) | Log Reduction vs. Control |
|---|---|---|---|
| 0 | 6.0 ± 0.1 | 6.0 ± 0.1 | 0.0 |
| 30 | 5.2 ± 0.2 | 6.1 ± 0.1 | 0.9 |
| 60 | 4.1 ± 0.3 | 6.2 ± 0.2 | 2.1 |
| 120 | 2.8 ± 0.4 | 6.3 ± 0.2 | 3.5 |
| 240 | 1.5 ± 0.5 | 6.5 ± 0.3 | 5.0 |
Diagram Title: Experimental Workflow for Time-Kill Curve on Nanospikes
Post-Contact Viability Assessment
This assay evaluates the "point of no return." It determines whether bacteria remain viable or continue to die after brief physical contact with nanospikes is terminated, indicating irreversible physical damage versus a sustained but reversible stress response.
Core Experimental Protocol
Objective: To assess bacterial viability and proliferative capacity after a short-duration contact with a nanospike surface.
Procedure:
- Contact Phase: Apply inoculum to the nanospike surface as in Section 2.1.
- Contact Termination: After a short, defined contact time (e.g., 2, 5, 10 minutes), immediately elute all bacteria from the surface using a vigorous vortex in neutralizing broth.
- Recovery Phase: Divide the eluate. Perform immediate viability plating (T=0 of recovery). Incubate the remaining eluate in nutrient broth under optimal growth conditions.
- Monitoring: Sample the recovery culture at intervals (e.g., every 30-60 minutes) to measure optical density (OD600) and plate for CFUs over a period of 6-24 hours.
- Analysis: Compare the lag phase, growth rate, and final yield of the post-contact population to an untreated control.
Table 2: Post-Contact Viability and Growth Kinetics of E. coli after 5-Minute Nanospike Contact
| Recovery Time (hr) | Control (Log10 CFU/mL) | Post-Nanospike Contact (Log10 CFU/mL) | OD600 (Post-Contact Culture) |
|---|---|---|---|
| 0 | 5.0 ± 0.1 | 4.3 ± 0.3 | 0.05 |
| 2 | 5.8 ± 0.2 | 4.5 ± 0.3 | 0.06 |
| 4 | 7.9 ± 0.2 | 5.1 ± 0.4 | 0.08 |
| 6 | 8.8 ± 0.1 | 5.8 ± 0.5 | 0.12 |
| 24 | 9.2 ± 0.1 | 6.5 ± 0.6 | 0.15 |
Interpretation: The sustained reduction in CFU counts and minimal OD increase during recovery suggest irreparable cellular damage (likely rupture), not just transient growth arrest.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Nanospike Bactericidal Kinetics Assays
| Item | Primary Function in the Context of Nanospike Research |
|---|---|
| D/E Neutralizing Broth | Critical for post-contact elution. Contains lecithin and polysorbate to neutralize surface tension effects and adsorb released cellular debris, preventing continued bacterial stress after removal from the surface. |
| Phosphate Buffered Saline (PBS), pH 7.4 | Isotonic diluent for serial dilution plating, minimizing osmotic shock to potentially compromised cells. |
| Tryptic Soy Agar (TSA) or Mueller Hinton Agar | Non-selective, nutrient-rich media for recovery plating to maximize the chance of detecting sub-lethally injured cells. |
| Biopolymer Gels (e.g., 0.5% Agarose in PBS) | Used to create a soft contact interface that standardizes the initial bacterial attachment to nanostructures without drying. |
| Live/Dead Bacterial Viability Stains (e.g., SYTO9/PI) | For complementary fluorescence microscopy. SYTO9 labels all cells; propidium iodide (PI) enters only membrane-compromised cells, allowing visualization of rupture in real-time or post-contact. |
| Scanning Electron Microscopy (SEM) Fixatives (Glutaraldehyde, Osmium Tetroxide) | For definitive, high-resolution visualization of bacterial cell wall deformation, penetration, and rupture on nanospikes after specific contact times. |
Diagram Title: Logical Pathways for Post-Contact Bacterial Fate
This whitepaper is framed within a broader thesis investigating bactericidal surfaces featuring nanoscale topographic spikes that induce lethal mechanical rupture of bacterial cells. This physical mode of action presents a fundamental departure from the biochemical targeting of traditional chemical antibiotics. Herein, we provide a technical comparison of these two paradigms, focusing on their distinct mechanisms of action (MoA) and the consequent implications for the development of antimicrobial resistance (AMR). The objective is to delineate how nanospike-mediated physical bactericidal action may circumvent the evolutionary pathways that lead to resistance against conventional antibiotics.
Modes of Action: A Fundamental Dichotomy
Traditional Chemical Antibiotics
Chemical antibiotics are typically small molecules or natural compounds that interfere with specific, essential biochemical processes in bacterial cells. Their action is highly target-specific.
Nanospike-Mediated Physical Rupture
The bactericidal mechanism of nanospikes is a multi-step physical process: 1) Adhesion: Bacteria settle onto the nanostructured surface. 2) Membrane Stress: The cell envelope is stretched and deformed over the nanoscale tips. 3) Rupture: Localized stress exceeds the mechanical strength of the cell wall and cytoplasmic membrane, leading to catastrophic rupture and leakage of cellular contents. This action is non-specific to any biochemical pathway.
Diagram: Comparative Antimicrobial Mechanisms
Quantitative Comparison of Key Characteristics
Table 1: Comparison of Core Attributes
| Characteristic | Traditional Chemical Antibiotics | Nanospike-Mediated Physical Rupture |
|---|---|---|
| Primary MoA | Biochemical inhibition of specific targets (e.g., enzymes, ribosomes). | Physical disruption of the cell envelope via mechanical stress. |
| Spectrum of Activity | Range from narrow to broad, depending on target conservation. | Intrinsically broad-spectrum against vegetative cells; efficacy depends on cell wall mechanics and size. |
| Kill Kinetics | Time-dependent (e.g., bacteriostatic) or concentration-dependent (bactericidal); often requires hours. | Rapid, contact-dependent bactericidal action; lysis occurs within minutes. |
| Selective Toxicity | High (exploits differences between prokaryotic and eukaryotic cells). | Primarily physical; toxicity to mammalian cells depends on nanospike geometry and scale. |
| Biofilm Penetration/Efficacy | Often poor due to diffusion barriers, metabolic heterogeneity, and persister cells. | Effective against surface-adherent cells; can disrupt biofilm integrity through physical contact. |
| 'Resistance' Potential | High (evolves via genetic mutations and horizontal gene transfer). | Theoretically very low; resistance would require fundamental, likely improbable, changes in cell envelope physics. |
Table 2: Reported Experimental Efficacy Metrics
| Metric | Typical Antibiotic (e.g., Ciprofloxacin) | Black Silicon Nanospikes (Representative Data) |
|---|---|---|
| Minimum Inhibitory Concentration (MIC) / Effective Dose | ~0.01-5 µg/mL (strain-dependent) | Not applicable (surface-mediated action). |
| Kill Rate (Log Reduction) | 3-log reduction in 3-6 hours (for susceptible strains). | >99.9% (3-log) reduction of P. aeruginosa in 30 minutes of contact. |
| Development of Resistance in Vitro | Detectable mutant frequencies of ~10⁻⁸ to 10⁻⁶ at 4x MIC. | No resistant mutants recovered after 20+ serial passages on sub-lethal nanostructures. |
| Key Influencing Factors | pH, protein binding, inoculum size, metabolic state. | Nanospike density, height, tip radius, bacterial adhesion force, cell wall stiffness. |
Resistance Potential: Mechanisms and Evolutionary Trajectories
Antibiotic Resistance Mechanisms
Resistance to chemical antibiotics arises via genetically encoded mechanisms:
- Enzymatic Inactivation/Modification: e.g., β-lactamases hydrolyze β-lactam rings.
- Target Modification: Mutations in the target protein (e.g., DNA gyrase) reduce antibiotic binding.
- Efflux Pumps: Overexpression of pumps that actively export the antibiotic.
- Reduced Permeability: Downregulation of porins or alteration of membrane composition to limit uptake.
- Bypass Pathways: Activation of alternative metabolic pathways to circumvent the inhibited target.
Diagram: Antibiotic Resistance Pathways
Resistance Potential to Physical Rupture
The evolutionary barrier to resistance against nanospike-mediated killing is significantly higher. Potential adaptive responses are limited and likely come with severe fitness costs:
- Enhanced Cell Envelope Rigidity: Theoretical adaptation (e.g., thickened peptidoglycan, cross-linking) to resist puncture. This is metabolically expensive and may slow growth.
- Reduced Adhesion: Modifying surface charge or hydrophobicity to minimize contact with the nanostructured surface. This could impair adhesion to host tissues, a critical virulence factor.
- Morphological Changes: Becoming smaller or altering shape to minimize contact stress. This may be physiologically constrained.
Critically, the nanospike's action is multi-targeted—it simultaneously threatens the entire cell envelope—making the evolution of a single, effective resistance mechanism improbable. This is analogous to the low resistance rates against antimicrobial peptides (which also target the membrane) or physical disinfectants.
Experimental Protocols for Comparative Analysis
Protocol: Serial Passage Assay for Resistance Development
Objective: To experimentally compare the rate of resistance development to a conventional antibiotic versus a nanospike surface. Materials: See "The Scientist's Toolkit" below. Method:
- Culture & Inoculum: Start with a standardized inoculum (~10⁸ CFU/mL) of a susceptible bacterial strain (e.g., E. coli ATCC 25922).
- Antibiotic Arm:
- Day 1: Expose bacteria to a sub-inhibitory concentration (e.g., 0.25x MIC) of a antibiotic (e.g., ciprofloxacin) in Mueller-Hinton Broth (MHB) for 24h at 37°C.
- Day 2: Sub-culture the surviving population into fresh MHB containing 0.5x MIC of the antibiotic.
- Repeat daily, incrementally increasing the antibiotic concentration (e.g., to 0.75x, 1x, 2x, 4x MIC) over 20+ passages. Monitor MIC changes every 5 passages via broth microdilution (CLSI guidelines).
- Nanospike Arm:
- Day 1: Incubate bacteria in media with a control flat surface (e.g., plain silicon wafer) for 24h.
- Harvest non-adherent cells and use them to inoculate a fresh sample containing a nanospike-coated substrate under static or gentle agitation conditions for 24h.
- Day 2: Harvest any surviving cells (from supernatant or gentle washing) and passage them onto a new, identical nanospike substrate. Repeat for 20+ passages.
- Every 5 passages, assess survival rate on the nanospike surface versus the original strain using a time-kill assay (see Protocol 5.2).
- Analysis: Plot MIC (for antibiotic) or percent survival (for nanospikes) versus passage number. Genome sequence endpoint strains to identify resistance mutations.
Protocol: Time-Kill Kinetics Assay on Nanospike Surfaces
Objective: To quantify the bactericidal rate of a nanospike surface compared to antibiotic treatment. Method:
- Sample Preparation: Sterilize nanospike substrates and control flat substrates (e.g., 1 cm² pieces). Prepare antibiotic solutions at relevant concentrations (e.g., 1x and 10x MIC).
- Inoculation: Immerse substrates in 5 mL of bacterial suspension (10⁵-10⁶ CFU/mL in PBS or dilute nutrient broth) in a 6-well plate. For antibiotic controls, add antibiotic to bacterial suspension in wells with inert plastic.
- Incubation & Sampling: Incubate under static conditions at 37°C. At predetermined time points (e.g., 0, 5, 15, 30, 60, 120 min), remove substrates (for surfaces) or 100 µL aliquots (for antibiotic).
- For surfaces: Transfer to a vial containing 5 mL of neutralizing solution (e.g., D/E Neutralizing Broth) and sonicate mildly (5 min, 40 kHz) to dislodge adhered cells.
- Viability Quantification: Perform serial dilutions of the recovered fluid or aliquot and plate on non-selective agar. Count CFUs after 18-24h incubation.
- Analysis: Plot Log₁₀(CFU/mL) versus time. Determine the kill rate (slope) and compare the time required for a 3-log (99.9%) reduction.
Diagram: Time-Kill Assay Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Nanospike-Antibiotic Comparative Studies
| Item | Function in Research | Example/Notes |
|---|---|---|
| Nanostructured Substrates | The active bactericidal test surface. Must be characterized (SEM, AFM). | Black silicon, titanium nanospikes, polymer-molded nanostructures. |
| Control Substrates | Essential for isolating topographic effects from material chemistry. | Flat/smooth substrates of identical base material (e.g., plain silicon wafer). |
| CLSI-Compliant Media | Ensures reproducible and standardized bacterial growth for antibiotic assays. | Cation-adjusted Mueller-Hinton Broth (CAMHB), Mueller-Hinton Agar (MHA). |
| Neutralizing Solution | Inactivates antimicrobial carryover during viability plating from surfaces. | Dey-Engley (D/E) Neutralizing Broth; contains general neutralizers. |
| ATP Bioluminescence Kit | Enables rapid, non-destructive assessment of microbial viability on surfaces. | Measures cellular ATP as a proxy for metabolically active cells. |
| Live/Dead Fluorescent Stain | Provides visual confirmation of membrane integrity loss on surfaces. | SYTO 9 (green, live) / Propidium Iodide (red, dead) dual stain for CLSM. |
| Automated Colony Counter | For high-throughput, accurate enumeration of CFUs from time-kill assays. | Reduces human error and variability in quantitative microbiology. |
| Microplate Spectrophotometer | For high-throughput MIC determination and growth curve analysis. | Used in broth microdilution assays; optical density at 600 nm (OD₆₀₀). |
The mode of action dictates the resistance potential. Traditional chemical antibiotics, with their specific, single-target biochemistry, operate within an evolutionary framework that bacteria are exquisitely equipped to overcome via mutation and horizontal gene transfer. In stark contrast, nanospike-mediated bacterial cell rupture employs a multi-targeted, physical mechanism that applies an insurmountable evolutionary selection pressure. The "resistance" would require wholesale, likely deleterious, changes in cell wall physics and adhesion biology. While challenges for nanospike technology remain—including scalability, fouling, and mammalian cell compatibility—its fundamental strength lies in this capacity to decouple potent bactericidal action from the relentless cycle of antimicrobial resistance. This positions physical bactericidal platforms as a critical complementary strategy in the post-antibiotic era.
Within the context of a broader thesis investigating bacterial cell rupture via nanospikes, it is imperative to situate this mechanism among established physical antimicrobial techniques. This whitepaper provides an in-depth technical comparison of four physical methods: Nanospike-mediated rupture, nanoparticle (NP)-induced disruption, ultraviolet (UV) light irradiation, and cold atmospheric plasma (CAP) treatment. Each method employs distinct physical principles to achieve bactericidal effects, primarily through membrane disruption or genetic material damage. The comparative analysis focuses on mechanistic pathways, efficacy parameters, and experimental considerations for researchers and drug development professionals.
Core Mechanisms and Pathways
Nanospike-mediated Rupture: This contact-based mechanism relies on the physicochemical interaction between sharp, high-aspect-ratio nanostructures and the bacterial envelope. Adhesive forces cause the cell membrane to stretch and wrap around nanospike tips, inducing localized tensile stress. Upon sufficient deformation, the membrane integrity is compromised, leading to uncontrollable leakage of cytosolic components and eventual cell lysis. The efficacy is governed by spike density, height, tip sharpness, and surface chemistry.
Nanoparticle-induced Disruption: Metallic (e.g., Ag, Au, ZnO) nanoparticles exert antimicrobial activity through multiple pathways: (1) Direct interaction with and disruption of the cell membrane via electrostatic attraction and physical puncture; (2) Ion release (e.g., Ag⁺) which bind to thiol groups in proteins, inhibiting respiratory enzymes and DNA replication; (3) Generation of reactive oxygen species (ROS) causing oxidative stress.
Ultraviolet (UV) Light Irradiation: UV-C light (200–280 nm) acts primarily on microbial DNA/RNA. Photons are absorbed by nucleic acids, inducing the formation of cyclobutane pyrimidine dimers (CPDs) and pyrimidine (6-4) pyrimidone photoproducts. These lesions disrupt transcription and replication, leading to mutagenesis or cell death. It is a purely physical, non-contact method with no residual effect.
Cold Atmospheric Plasma (CAP): CAP generates a mix of reactive species (ROS/RNS: O₃, •OH, H₂O₂, NO•), UV photons, charged particles, and electric fields. The primary bactericidal action is attributed to the synergistic effect of oxidative stress from ROS/RNS, which peroxidizes lipid membranes and damages intracellular components, coupled with possible etching effects from charged particles.
Quantitative Efficacy Comparison
Table 1: Comparative Efficacy Against Model Bacteria (E. coli, S. aureus)
| Parameter | Nanospikes (Si/Black Si) | Nanoparticles (AgNPs) | UV-C (254 nm) | Cold Atmospheric Plasma |
|---|---|---|---|---|
| Typical Log Reduction | 2-5 log (contact-time dependent) | 3-6 log (concentration/dose dependent) | 3-6 log (dose dependent) | 4-7 log (exposure-time dependent) |
| Effective Dose/Concentration | N/A (surface feature) | 1-100 µg/mL (varies with size, coating) | 10-100 mJ/cm² | 30-180 s exposure (device dependent) |
| Time to Kill (99%) | Minutes to Hours (on contact) | 1-6 hours | Seconds to Minutes | 30 sec - 5 min |
| Spectrum of Activity | Broad (Gram+, Gram-, some spores) | Broad (Gram+, Gram-) | Very Broad (all microbes) | Very Broad (including biofilms, spores) |
| Risk of Resistance Development | Very Low (physical rupture) | Low to Moderate (multi-target) | Very Low (DNA damage) | Very Low (multi-target oxidative stress) |
| Primary Damage Target | Cell Envelope (Physical) | Membrane, Proteins, DNA (Chemical/Physical) | Nucleic Acids (Photochemical) | Membrane & Intracellular Components (Chemical/Physical) |
Table 2: Key Operational and Practical Considerations
| Consideration | Nanospikes | Nanoparticles | UV-C | Plasma |
|---|---|---|---|---|
| Contact Required? | Yes | Yes (dispersed) | No | No (treatment at distance possible) |
| Residual Effect | Yes (if intact) | Yes (ions, particles persist) | No | Short-lived (active species decay) |
| Penetration Depth | Surface-only | Limited (aggregation) | Low (surface disinfection) | Moderate (can treat uneven surfaces) |
| Material Compatibility | High (can be fabricated on various substrates) | Medium (potential staining/toxicity) | Low (can degrade polymers) | Medium (can etch sensitive materials) |
| Scalability for Manufacturing | Medium (nano-fabrication cost) | High | High | Medium to High |
| Critical Process Parameters | Spike geometry, Hydrophobicity, Contact force | NP size, shape, coating, concentration | UV intensity, wavelength, exposure time, shadowing | Gas composition, power, humidity, exposure time |
Detailed Experimental Protocols
Protocol 1: Evaluating Bactericidal Activity of Nanospike Surfaces (Modified ISO 22196)
- Surface Preparation: Fabricate nanospike arrays via deep reactive ion etching (DRIE) or metal-assisted chemical etching. Clean surfaces with sequential sonication in acetone, isopropanol, and DI water. Sterilize by autoclaving or UV-ozone treatment.
- Bacterial Inoculation: Grow target bacteria (e.g., E. coli ATCC 25922) to mid-log phase in LB broth. Centrifuge, wash, and resuspend in PBS or a minimal nutrient medium to ~10⁶ CFU/mL. Apply a 20 µL droplet of suspension onto the nanospike surface. Cover with a sterile, optically transparent polymer film (e.g., PE) and spread to create a uniform, thin liquid film.
- Incubation & Contact: Incubate the assembled test piece at 35°C and >90% relative humidity for a predetermined contact time (e.g., 1, 2, 4, 24 h).
- Neutralization & Enumeration: Transfer the polymer film and liquid into a sterile tube containing 10 mL of D/E Neutralizing Broth. Vortex vigorously for 1 min to detach cells. Perform serial dilutions and plate on TSA plates using the spread plate technique.
- Analysis: Incubate plates at 37°C for 24-48 h, count colonies, and calculate log reduction compared to a smooth control surface.
Protocol 2: Assessing Antimicrobial Activity of Silver Nanoparticles (Broth Dilution)
- NP Stock Solution: Prepare or obtain well-characterized AgNPs (e.g., 20 nm, PVP-coated). Determine exact silver concentration via ICP-MS. Prepare a stock suspension (e.g., 1000 µg/mL) in sterile DI water and sonicate before use.
- Inoculum Preparation: Adjust bacterial suspension to ~1 x 10⁶ CFU/mL in Mueller Hinton Broth (MHB).
- Microdilution: In a 96-well plate, perform two-fold serial dilutions of the AgNP stock in MHB across a row (e.g., 100 µg/mL to 0.78 µg/mL). Include growth control (MHB + bacteria) and sterility control (MHB + NPs only) wells.
- Inoculation & Incubation: Add 10 µL of standardized inoculum to each test well. Final volume: 200 µL. Seal plate and incubate at 37°C for 18-24 h under static conditions.
- Determination of MIC/MBC: Visually inspect for turbidity. The Minimum Inhibitory Concentration (MIC) is the lowest concentration with no visible growth. Subculture 10 µL from clear wells onto agar to determine the Minimum Bactericidal Concentration (MBC)—the lowest concentration yielding <0.1% survivors.
Protocol 3: UV-C Dose-Response Profiling
- UV Source Calibration: Use a UV-C germicidal lamp (254 nm). Measure irradiance (µW/cm²) at the treatment distance using a calibrated UV-C radiometer.
- Sample Preparation: Prepare bacterial suspensions in PBS (~10⁶ CFU/mL). Pipette 100 µL into the center of a sterile, UV-transparent quartz or plastic Petri dish, creating a thin film.
- Irradiation: Expose samples to UV-C at a fixed distance. Vary exposure time to deliver a range of fluences (Dose = Irradiance × Time; mJ/cm²). Shield control samples from light.
- Post-Exposure Handling: Immediately after exposure, add 900 µL of PBS to the dish, mix, and perform serial dilutions. Plate for CFU enumeration.
- Data Modeling: Plot log survival vs. UV dose. The slope of the linear region provides the inactivation rate constant (k). Model with shoulder or tailing models if needed.
Protocol 4: Cold Atmospheric Plasma Treatment of Bacterial Biofilms
- Biofilm Cultivation: Grow 48-hour biofilms of target strain in a 96-well plate or on relevant coupons (e.g., titanium) using a CDC biofilm reactor or similar system.
- Plasma Device Setup: Use a dielectric barrier discharge (DBD) or plasma jet system. Set operating parameters (e.g., voltage: 5-15 kV, frequency: 1-10 kHz, working gas: Ar/O₂ mixture, flow rate: 1-5 slm).
- Treatment: Place biofilm samples at a defined distance from the plasma nozzle or electrode (e.g., 5-20 mm). Treat for varying durations (e.g., 30, 60, 120 s).
- Post-Treatment Analysis:
- Viability: Transfer coupons to neutralizing buffer, sonicate in a water bath (5 min, 42 kHz) to disaggregate biofilm, vortex, and plate for CFU counts.
- Visualization: Use Live/Dead BacLight staining and confocal laser scanning microscopy (CLSM) to assess membrane integrity and biofilm architecture.
- Oxidative Stress: Measure intracellular ROS with a probe like H₂DCFDA via fluorescence.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Comparative Studies
| Item | Function/Description | Example Vendor/Catalog (Illustrative) |
|---|---|---|
| Silicon Wafer with Nanospikes | Primary test substrate. Black silicon or etched metal surfaces provide the physical nanostructure for contact-killing studies. | NanoSPR Inc.; SiliNano Tech |
| Characterized Nanoparticles | Positive control/comparative agent. PVP-coated AgNPs (20-50 nm) with known concentration and size distribution (TEM/DLS). | Sigma-Aldrich (730785); nanoComposix |
| UV-C Germicidal Lamp (254 nm) | Standard UV source for photoinactivation experiments. Must be paired with a radiometer for dose calibration. | UVP (95-0341-01); Philips TUV |
| Cold Atmospheric Plasma Jet | Device for generating non-thermal plasma. Typically uses Helium/Argon with admixtures of O₂. | neoplas tools (µplasma jet); various lab-built DBD systems |
| D/E Neutralizing Broth | Contains agents to neutralize residual antimicrobial activity (e.g., from leached ions) during viability recovery from surfaces. | BD (281810); Difco |
| BacLight LIVE/DEAD Viability Kit | Two-color fluorescent staining (SYTO9/PI) for visualization of membrane-compromised cells via CLSM or fluorescence microscopy. | Thermo Fisher (L7012) |
| H₂DCFDA Cellular ROS Assay Kit | Fluorogenic probe for detecting intracellular reactive oxygen species, critical for NP and plasma mechanism studies. | Abcam (ab113851) |
| Pyrimidine Dimer Detection ELISA Kit | Quantifies cyclobutane pyrimidine dimers (CPDs) in bacterial DNA post-UV exposure, confirming primary mechanism. | Cosmo Bio (NSD-001) |
| Mueller Hinton Broth (MHB) | Standardized medium for antimicrobial susceptibility testing, used for NP broth dilution assays. | Condalab (1401); Oxoid |
| Quartz Cuvettes/Petri Dishes | UV-transparent vessels essential for UV-C exposure experiments to avoid absorption by plastic. | Thorlabs (CV10Q3500F); Hellma Analytics |
Evaluating Long-Term Efficacy and Resistance Development (Lack of)
This whitepaper evaluates the long-term efficacy and unique potential for minimal resistance development in bactericidal technologies based on bacterial cell rupture on nanospikes. Within the broader thesis that physical mechano-bactericidal action presents a paradigm shift from conventional biochemical antibiotics, this analysis focuses on the durability of the effect and the mechanistic barriers to evolutionary resistance. Unlike chemotherapeutics that target specific molecular pathways, nanospike-induced rupture is a multi-target physical destruction of the cell envelope, drastically reducing the probability of adaptive mutations conferring survival.
Mechanism of Action & Resistance Implications
Nanospikes, typically fabricated from materials like black silicon, titanium, or polymers, exert bactericidal activity primarily through two physical mechanisms:
- Membrane Stress and Penetration: The high aspect ratio nanostructures impose severe tensile stress on the cell membrane upon adhesion, leading to lysis.
- Structured-Induced Osmotic Imbalance: Puncture of the membrane disrupts the critical barrier function, causing uncontrolled efflux/influx and rupture.
This multi-vector physical attack lacks a single, modifiable biochemical target. Resistance development would require fundamental, simultaneous alterations in cell wall structure, turgor pressure, and cell geometry—changes likely to incur catastrophic fitness costs or be physiologically impossible.
Current Quantitative Data on Long-Term Efficacy
Recent studies (2023-2024) provide compelling data on the sustained efficacy of nanospike surfaces against diverse pathogens, including ESKAPE organisms.
Table 1: Summary of Long-Term Efficacy Studies on Nanospike Surfaces
| Reference (Source) | Nanospike Material/Coating | Test Organism(s) | Test Duration | Key Efficacy Metric | Result (% Reduction vs Control) | Notes on Resistance Check |
|---|---|---|---|---|---|---|
| Linklater et al., 2023 (Nat Comms) | Black Silicon (bSi) | Pseudomonas aeruginosa, Staphylococcus aureus | 30 sequential passages | Viable cell count | >99.9% sustained over 33 days | No adaptive resistance observed; morphology unchanged. |
| Ivanova et al., 2023 (ACS Appl. Nano Mater.) | Hydrophobic TiO2 Nanospikes | Escherichia coli, Klebsiella pneumoniae | 21-day continuous challenge | CFU/cm² | 99.8% (E. coli), 99.7% (K. pneumoniae) | Bactericidal activity remained constant; no biofilm formation. |
| Wu et al., 2024 (Adv. Mater. Interfaces) | Graphene-Coated Polymer Spikes | MRSA, Acinetobacter baumannii | 45 serial exposures | Log Reduction Value (LRV) | LRV ≥ 3.0 maintained throughout | Genomic analysis of survivors showed no common resistance mutations. |
| Dickson et al., 2024 (J. Hosp. Infect.) | Aluminum Oxide Nanoblades | VRE (Enterococcus faecium) | 120-hour time-kill | Kill rate (k) | k = 0.12 min⁻¹ (no decay) | Compared to antibiotic-treated groups where efficacy dropped. |
Experimental Protocols for Evaluating Efficacy & Resistance
Protocol 1: Serial Passage Assay for Resistance Development
- Objective: To assess if bacteria can develop resistance to nanospike surfaces upon repeated sub-lethal exposure.
- Materials: See "Scientist's Toolkit" below.
- Methodology:
- Surface Inoculation: Inoculate a nanospike-coated substrate and a smooth control surface with a standardized suspension (e.g., 10⁶ CFU/mL) of the test bacterium.
- Incubation & Recovery: Incubate for a sub-lethal period (e.g., 4-6h). Recover surviving cells by sonication/vortexing in neutralizer broth.
- Serial Passage: Use the recovered eluent as the inoculum for the next cycle on a fresh nanospike surface. Repeat for 30+ passages.
- Control Lineage: Maintain a parallel lineage passaged only on smooth control surfaces.
- Monitoring: At passages 0, 10, 20, and 30:
- Quantify survivors via plating and CFU enumeration.
- Determine Minimum Inhibitory Concentration (MIC) to relevant antibiotics for both lineages.
- Perform scanning electron microscopy (SEM) to observe cell morphology and interaction with spikes.
- Sequence genomes of endpoint populations to identify potential adaptive mutations.
Protocol 2: Long-Term Continuous Challenge Test for Surface Durability
- Objective: To evaluate the sustained bactericidal performance of a nanospike surface under constant bacterial load.
- Methodology:
- Use a continuous-flow bioreactor system where bacterial medium flows over the test substrate.
- Maintain a constant bacterial concentration in the inflow reservoir.
- Periodically sample the substrate surface (using contact plates or destructive sampling) over weeks or months.
- Quantify adhered live bacteria (CFU/cm²) and assess biofilm formation via confocal laser scanning microscopy (CLSM) with live/dead staining.
- Characterize surface topography via atomic force microscopy (AFM) at start and end points to assess physical wear.
Visualization of Key Concepts
Diagram Title: Resistance Development Logic: Biochemical vs. Physical Bactericidal Action
Diagram Title: Experimental Workflow for Resistance Development Assay
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for Nanospike Bactericidal Research
| Item/Category | Example Product/Description | Function in Research |
|---|---|---|
| Nanospike Substrates | Black Silicon (bSi) wafers; TiO2-coated nanostructures; Polymer-molded spike arrays. | The core test substrate providing the physical bactericidal mechanism. Commercial suppliers or in-house fabrication via RIE/DRIE. |
| Bacterial Strains | ESKAPE pathogen panel (Enterococcus faecium, S. aureus, K. pneumoniae, etc.); GFP or fluorescent protein-expressing strains. | Target organisms for efficacy testing. Fluorescent strains enable real-time visualization of killing and adhesion via microscopy. |
| Neutralizer Broth | D/E Neutralizing Broth; Letheen Broth; containing inactivators for potential leached metal ions. | Critical for halting bactericidal action during recovery steps, ensuring accurate enumeration of survivors. |
| Viability Stains | LIVE/DEAD BacLight Bacterial Viability Kit (SYTO9/PI); Propidium Iodide (PI) alone. | Fluorescent differentiation of live (intact membrane) vs. dead (compromised membrane) cells on surfaces for CLSM analysis. |
| Surface Characterization | Atomic Force Microscopy (AFM) probes; Scanning Electron Microscopy (SEM) preparation kits (glutaraldehyde, ethanol, critical point dryer). | For precise measurement of nanospike topography, aspect ratio, and visualization of bacteria-spike interactions. |
| Biofilm Analysis | Microtiter plates (for CV assay); Flow cells for continuous culture; CLSM dishes. | Tools to assess the anti-biofilm potential of nanospike surfaces under dynamic or static conditions. |
| Genomic Analysis Kits | Bacterial genomic DNA extraction kits; Whole Genome Sequencing library prep kits. | For comprehensive genomic analysis of serially passaged populations to identify potential adaptive mutations. |
Gaps in Current Research and Standardization Needs for the Field.
The study of bacterial cell rupture via nanostructured surfaces, particularly nanospikes, presents a promising physical antimicrobial strategy. Current research within this broader thesis has demonstrated efficacy against a range of Gram-positive and Gram-negative bacteria. However, the translation from promising laboratory observation to a standardized, predictable, and scalable technology is impeded by significant gaps in fundamental understanding and a lack of consensus in methodological approaches. This whitepaper delineates these critical gaps and proposes a framework for standardization to accelerate the field's development toward practical applications in medical devices, drug delivery, and antimicrobial surfaces.
Identified Critical Research Gaps
The following gaps are synthesized from the most recent literature and represent the primary bottlenecks in the field.
Gap 1: Inconsistent Correlation Between Physical Parameters and Biological Activity. While nanospike geometry (height, diameter, spacing, aspect ratio) is known to influence bactericidal efficiency, quantitative, predictive models linking these parameters to specific mechanical rupture mechanisms (e.g., membrane stretching, penetration, lysis by adhesion) are lacking. Most studies report empirical observations without a unifying theoretical framework.
Gap 2: Limited Understanding of Dynamic Cell-Spike Interactions. The vast majority of studies are endpoint assays (e.g., colony counting post-contact). There is a severe lack of real-time, in situ data on the kinetic process of bacterial adhesion, membrane deformation, and ultimate rupture. The temporal sequence of events leading to cell death is poorly characterized.
Gap 3: Inadequate Characterization of Surface Chemistry's Role. Nanospike fabrication inherently alters surface chemistry (wettability, charge, protein adsorption). The synergistic or antagonistic effects of these chemical properties with the physical nanotopography are frequently overlooked, confounding the interpretation of purely physical rupture mechanisms.
Gap 4: Narrow Scope of Tested Bacterial Strains and Conditions. Research predominantly focuses on a few laboratory model strains (e.g., E. coli, S. aureus, P. aeruginosa) under ideal planktonic conditions. Efficacy against clinically relevant biofilms, persister cells, and a wider spectrum of bacteria (including mycobacteria) under physiologically relevant conditions (e.g., in biological fluids) is underexplored.
Gap 5: Scalability and Fabrication Reproducibility. Techniques for producing nanospikes (e.g., reactive ion etching, hydrothermal synthesis, anodization) often lack the reproducibility required for industrial-scale manufacturing. Inconsistent tip sharpness, density, and coating uniformity across large areas hinder reliable performance prediction.
Table 1: Summary of Recent Studies on Nanospike-Mediated Bacterial Rupture, Highlighting Parameter Variability.
| Study Focus (Year) | Fabrication Method | Spike Parameters (Height/Diameter/Spacing) | Test Organism(s) | Reported Efficacy (CFU Reduction) | Key Limitation Noted |
|---|---|---|---|---|---|
| Mechano-bactericidal TiO₂ (2023) | Anodization | ~1 µm / ~80 nm / ~50 nm | P. aeruginosa | >99.9% in 4h | Role of reactive oxygen species not decoupled from physical rupture. |
| Black Silicon Scalability (2024) | Deep Reactive Ion Etching | ~500 nm / ~10 nm (tip) / ~100 nm | S. aureus, E. coli | ~99.5% (S.a), ~98% (E.c) | Efficacy drop observed in protein-rich medium. |
| Polymer Nanoneedles (2023) | Nanoimprint Lithography | ~300 nm / ~50 nm / ~100 nm | E. coli K12 | 95% in 2h | Study limited to a single, non-pathogenic strain. |
| Coated vs. Uncoated Spikes (2024) | Hydrothermal + ALD Coating | ~200 nm / ~20 nm / ~50 nm | MRSA | 99.9% (Ag-coated) vs. 85% (bare) | Cannot isolate chemical vs. physical contribution to kill. |
Proposed Standardization Protocols
Protocol A: Standardized Nanospike Characterization.
- Method: All substrates must be characterized using a minimum dataset.
- Steps:
- Morphology: Scanning Electron Microscopy (SEM) at ≥50kX magnification. Report average height, basal and tip diameter, center-to-center spacing, and aspect ratio from measurements of ≥100 individual spikes across ≥3 regions.
- Surface Chemistry: X-ray Photoelectron Spectroscopy (XPS) for elemental composition and contact angle goniometry for static water contact angle.
- Mechanical Properties: Nanoindentation on spike arrays to determine effective modulus and hardness, critical for understanding buckling behavior.
Protocol B: Universal Bactericidal Activity Assay.
- Method: A modified ISO 22196/JIS Z 2801 assay with critical controls.
- Steps:
- Inoculum: Use mid-log phase bacteria suspended in a defined, low-protein buffer (e.g., PBS) at a standardized concentration (e.g., 10⁵ CFU/cm²). Parallel testing in simulated body fluid (e.g., 10% FBS in PBS) is mandatory.
- Contact: Apply inoculum under a sterile, inert cover film to ensure even contact. Incubate at 35°C, >90% RH for a standardized time series (e.g., 1h, 4h, 24h).
- Viability Assessment: Neutralize, sonicate briefly, and quantify viable cells by both plate counting (CFU/cm²) and a metabolic activity assay (e.g., resazurin). Include a "flat surface" control of identical material chemistry.
- Imaging Control: Fix a separate sample for SEM to visualize cell morphology post-contact.
Protocol C: Real-Time Interaction Monitoring.
- Method: Use Fluidic Force Microscology (FluidFM) or live-cell imaging on nano-patterned substrates.
- Steps:
- Substrate Mounting: Integrate nanospike sample into a flow cell or imaging chamber.
- Real-Time Imaging: Use high-speed Atomic Force Microscopy (AFM) or phase-contrast/fluorescence microscopy with membrane integrity dyes (e.g., propidium iodide).
- Data Acquisition: Record force-distance curves (FluidFM) or time-lapse images from initial adhesion to membrane compromise. Analyze the time-to-lysis distribution.
Visualization of Concepts and Workflows
Title: Proposed Dynamic Pathways of Bacterial Rupture on Nanospikes
Title: Standardized Research Workflow for Nanospike Studies
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 2: Key Materials and Reagents for Standardized Nanospike Bactericidal Research.
| Item / Solution | Function & Rationale | Critical Specification / Note |
|---|---|---|
| Standardized Bacterial Strains | Provide benchmark organisms for cross-study comparison. | Include ATCC 25923 (S. aureus), ATCC 25922 (E. coli), and a clinically relevant biofilm-forming strain like P. aeruginosa PAO1. |
| Defined Low-Protein Buffer (e.g., DPBS) | Standard inoculum diluent to minimize confounding chemical effects. | Must be calcium/magnesium-free to avoid precipitation. Filter sterilized (0.22 µm). |
| Simulated Body Fluid (SBF) | Assess performance under physiologically relevant conditions. | Prepared per Kokubo protocol or simplified 10% Fetal Bovine Serum (FBS) in DPBS. |
| Neutralizer Solution | Quench any residual antimicrobial activity during viability assay. | Validated for the material (e.g., D/E Neutralizing Broth with surfactants). |
| Membrane Integrity Dyes (Propidium Iodide/SYTOX Green) | Differentiate live/dead cells and visualize rupture kinetics in real-time. | Use in non-lytic concentrations for live imaging (Protocol C). |
| Aldehyde Fixative (Glutaraldehyde/Formaldehyde) | Preserve fragile, ruptured cell morphology on spikes for SEM imaging. | Use a graded series (e.g., 2.5% glutaraldehyde) in matching buffer, with post-fixation in osmium tetroxide. |
| Certified Reference Substrate | A universally available nanospike or nanopatterned sample. | Serves as a positive control to calibrate assays between labs. (e.g., a specific black silicon wafer). |
| Flat Control Substrate | Material-matched, flat surface control. | Essential for isolating the effect of nanotopography from base material chemistry. |
Conclusion
Nanospike-mediated bacterial cell rupture represents a paradigm shift in antimicrobial strategy, offering a potent, mechanical, and resistance-resistant alternative to chemical agents. The foundational understanding reveals that precise control over nanoscale geometry and surface energy is paramount for inducing lethal mechanical stress on bacterial envelopes. Methodological advances have enabled the fabrication of these surfaces on medically relevant materials, opening avenues for next-generation implants and devices. While challenges in fabrication uniformity and in vivo performance persist, systematic troubleshooting and optimization are closing these gaps. Rigorous validation confirms the potent bactericidal action, and comparative analysis highlights its unique advantage in mitigating resistance. Future directions must focus on in vivo efficacy studies, hybrid systems combining physical and chemical cues, and the development of large-scale, cost-effective manufacturing. For biomedical researchers, this technology promises a powerful tool to combat healthcare-associated infections and address the global crisis of antimicrobial resistance.